Introduction
Materials and Methods
Strains
Sample collection
Isolation of Campylobacter
Confirmation of Campylobacter
Motility assay
Statistical analysis
Results and Discussion
Introduction
Campylobacter is one of major foodborne pathogenic bacteria worldwide, and its symptoms after human infection include diarrhea, abdominal cramping, and headache (Habib et al., 2013). It is mainly associated with poultry including chickens, turkeys, and ducks (Moffatt et al., 2020). Poultry’s body temperature at 40-43°C and intestinal conditions of low oxygen tension correspond well with the optimal growth temperature, 42°C, and microaerophilic nature of Campylobacter, suggesting that the intestine of poultry is an ideal niche for Campylobacter (Park, 2002). Even though such unique growth conditions are unlikely to occur in food-related environments and Campylobacter is vulnerable to oxygen at the level of normal atmosphere, foodborne illnesses by Campylobacter remain high around the world.
Foodborne illness by Campylobacter occurs frequently around the world although the symptoms are generally not severe. World Health Organization estimated that 96 out of approximately 600 million cases of global foodbone illness were attributed to Campylobacter spp. in 2010 (Havelaar et al., 2015). In US, Campylobacter was the second most common foodborne bacterial pathogen in 2016 (Marder et al., 2017). In Canada, Campylobacter spp. were also one of the leading microbial agents causing foodborne illness, accounting for 8.4% cases among all the 30 foodborne pathogens (Thomas et al., 2013). In France, Campylobacter, Salmonella, and norovirus were responsible for more than 70% of all illnesses associated with foodborne pathogens (Van Cauteren et al., 2017). Consistently, in EU, Campylobacter was the most common bacterial agent causing gastroenteritis in 2017 (EFSA and ECDC, 2018). In South Korea, not different from these developed countries, Campylobacter was the third most common bacterial agent responsible for foodborne illness in 2019 (Food Safety Nation, 2020).
Chicken consumption is a major source for campylobacteriosis, a human infection by Campylobacter (Rosner et al., 2017). Despite the increasing trend in chicken consumption, the knowledge regarding prevalence of Campylobacter in retail chickens and their virulence potential is still limited. Therefore, in our study, the prevalence of C. jejuni and C. coli, two main foodborne pathogens among Campylobacter spp. in retail raw chickens was studied and their virulence potential was assessed by studying motility.
Materials and Methods
Strains
Campylobacter jejuni 11168 and 81-176 were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). C. jejuni 11168 was originally isolated from feces of human patient with diarrhea (Skirrow, 1977). C. jejuni 81-176 was originally isolated from human patient infected after consumption of raw milk (Korlath et al., 1985).
Sample collection
Sixty raw chicken samples were collected from retail stores located in GyeongGi-Do province in South Korea during 2015. Among 60, 30 samples were whole chickens from 3 different manufacturers. The other 30 samples were chicken cuts from 4 different manufacturers. After purchase, they were immediately transported to our laboratory on ice pack and processed within 4 h.
Isolation of Campylobacter
Campylobacter was isolated from raw chicken samples using modified Korean Food Standards Codex (Ministry of Food and Drug Safety, 2020). The sample was rinsed with 400 mL of buffered peptone water (Difco, Sparks, MD, USA) in a sterile chicken rinse bag (39 × 52 cm, Nasco, Fort Atkinson, WI, USA) by shaking manually for 1 min. The chicken rinsate of 25 mL was added to a 2x bolton broth (Oxoid, Basingstoke, UK) of 25 mL, supplemented with laked horse blood (Oxoid, Basingstoke, UK) to a final concentration of 2.5% and bolton broth selective supplement (Oxoid, Basingstoke, UK) to a final concentration of 20 mg/mL for cefoperazone, 20 mg/mL for vancomycin, 20 mg/mL for trimethoprim and 50 mg/mL for cycloheximide in a Whirl-Pak bag of 710 mL capacity (Nasco, Fort Atkinson, WI, USA). The samples were placed in a rectangular jar in which microaerobic atmosphere (6-12% O2, 5-8% CO2) was generated by Anaero Pack-MicroAero (Mitsubishi Gas Chemical Co., Tokyo, Japan). Then, they were incubated at 37°C for 4 h followed by at 42°C for 44 h under microaerobic conditions. After incubation, the enrichment was streaked on preston agar prepared from campylobacter agar base (Oxoid), supplemented with laked horse blood to a final concentration of 5%, modified preston campylobacter selective supplement (Oxoid), and campylobacter growth supplement (Oxoid). The plates were incubated at 42°C for 48 h under microaerobic conditions. Among the isolated colonies, those of semitranslucent, gray, and sleek morphology were presumed to be Campylobacter.
Confirmation of Campylobacter
The presumptive colonies were individually subcultured onto two sets of preston agar plates. One set was incubated at 42°C under microaerobic conditions, and the other set was incubated at 25°C under aerobic conditions. The isolates growing at 42°C under microaerobic conditions, but not at 25°C under aerobic conditions were further confirmed by spiral shape under microscopic analysis, and oxidase positive. To differentiate between C. jejuni and C. coli, hippurate hydrolysis test (Hardy Diagnostics, Santa Maria, CA, USA) was conducted following the manufacturer’s protocol.
Motility assay
Campylobacter was grown on tryptic soy agar supplemented with 5% (v/v) sheep blood at 37°C for about 28 h under microaerobic conditions (6-12% O2, 5-8% CO2) using Anaero Pack-MicroAero (Mitsubishi Gas Chemical Co.) as described previously with a minor modification (Jung et al., 2017). The culture was suspended in Mueller-Hinton broth using sterile cotton swabs at OD600 0.1. The cell suspension was inoculated onto a soft agar, Mueller-Hinton containing 0.4% agar, by stabbing the center of the agar to the bottom with a pipette tip loaded with 2 µL of cell suspension, followed by injecting the suspension. The inoculated plates were incubated at 37°C under microaerobic conditions with the lids facing upward. The motility halo was measured in diameter at 48 h. Motility index was calculated as the ratio of the diameter of motility halo of test strains to the mean of the triplicate measurements of a reference strain, 11168, which is a clinical isolate from human diarrhea (Champion et al., 2008).
Statistical analysis
Statistical processing was performed with Tukey’s Honest Significant Difference (HSD) test at significance level of 0.05 by Minitab 17 (Minitab Inc., State College, PA, USA).
Results and Discussion
Retail raw chickens were analyzed for the presence of Campylobacter by a selective enrichment in bolton broth followed by selective plating on preston agar, and subsequent confirmation tests. Campylobacter was commonly found in both whole chickens (23 out of 30) and chicken cuts (22 out of 30) (Table 1). It was found in 45 (75%) out of 60 tested chicken samples overall. There was no difference between whole chickens and chicken cuts in terms of prevalence of Campylobacter. C. jejuni was more common than C. coli in tested chicken samples as many previous studies had shown (García-Sánchez et al., 2018; Kim et al., 2019). In some studies, however, C. coli was more common than C. jejuni in chicken meat (Walker et al., 2019).
Table 1.
Prevalence of Campylobacter in retail raw chickens in this study
| Samples | Number of samples tested | Positive | Total | |
| C. jejuni | C. coli | |||
| Whole chickens | 30 | 12 (40%) | 11 (37%) | 23 (77%) |
| Chicken cuts | 30 | 18 (60%) | 4 (13%) | 22 (73%) |
| Total | 60 | 30 (50%) | 15 (25%) | 45 (75%) |
Our results were compared to other studies from different countries (Table 2). Campylobacter was common among the commercial chickens and always detected in every country studied. The detection rates of Campylobacter from commercial chickens greatly varied and ranged between 17% and 92% depending on countries (Table 2).
Table 2.
Prevalence of Campylobacter in retail raw chickens in other studies
| Countries |
Samples (parts) |
Number of samples tested |
Number (%) of positive samples |
Study periods | References |
| Australia |
Breast, drumstick, thigh, wing, and whole chicken | 89 | 73 (82%) | 2016-2018 | Walker et al., 2019 |
| China |
Wing, leg, and breast Not specified; fresh and frozen |
227 302 |
42 (19%) 52 (17%) |
2015-2016 2014-2015 | Ma et al., 2017 Zhang et al., 2016 |
| Korea | Whole chicken | 133 | 67 (50%) | 2016-2017 | Kim et al., 2019 |
| Pakistan | Not specified | 200 | 80 (40%) | 2016 | Samad et al., 2019 |
| Canada |
Breast, wing, thigh, leg, and drumstick | 480 | 312 (65%) | 2012 | Hodges et al., 2019 |
| Romania | Unclear | 34 | 10 (29%) | 2016-2018 | Tîrziu et al., 2020 |
| Brazil |
Whole chickens and chicken cuts | 36 | 33 (92%) | Unspecified | Würfel et al., 2019 |
| Spain |
Chicken cuts, minced, marinated | 512 | 202 (39%) | 2015 | García-Sánchez et al., 2018 |
Virulence potential of Campylobacter is determined by many aspects of virulence characteristics including motility, chemotaxis, and host cell adhesion/invasion (Bolton, 2015). Motility is one of the important virulence characteristics among many foodborne pathogenic bacteria including Campylobacter and plays an import role in colonization of host intestines (Bolton, 2015; Kamar et al., 2013). Therefore, we attempted to understand a part of the pathogenic potential of Campylobacter isolates by measuring the diameter of their motility halo of 45 isolates (one isolate from each sample) in soft agar and comparing to the motility of two human clinical strains, C. jejuni 11168 and 81-176, which had been extensively studied (Fig. 1). The motility index was calculated as the ratio of the diameter of motility halo of test strains compared to the one of C. jejuni 11168. Compared to the motility index of 1.0 for strain 11168 as a control, both strain 81-176 and the group of 45 chicken isolates had significantly lower average motility indices of 0.74 and 0.75, respectively (P < 0.05) (Fig. 1). The motility index of each measurement in the chicken isolates group ranged between 0.11 and 1.77. Most measurements of the group (78%) fell into the motility index range of 0.34 to 0.93 and 18% had motility index more than 1.0. Of interest, the high motility index (> 1.0) was found only in C. jejuni. Based on that the average motility index of chicken isolates group is similar to strain 81-176 which is highly virulent and 18% of the chicken isolates group have motility index more than 1.0, this study shows that substantial population of Campylobacter contaminating retail chickens in Korea could be highly virulent and could pose a high risk for human infection. Several studies investigated the motility of Campylobacter isolates from animals or environments to evaluate their virulence characteristics (Shyaka et al., 2015; Stef et al., 2013). In Japan, Shyaka et al. (2015) found that only one out of 7 Campylobacter isolates from wild birds was more motile compared to strain 11168 and the rest were non-motile. In Romania, five C. jejuni chicken isolates showed a higher motility compared to the highly virulent strain 81-176 (Stef et al., 2013).
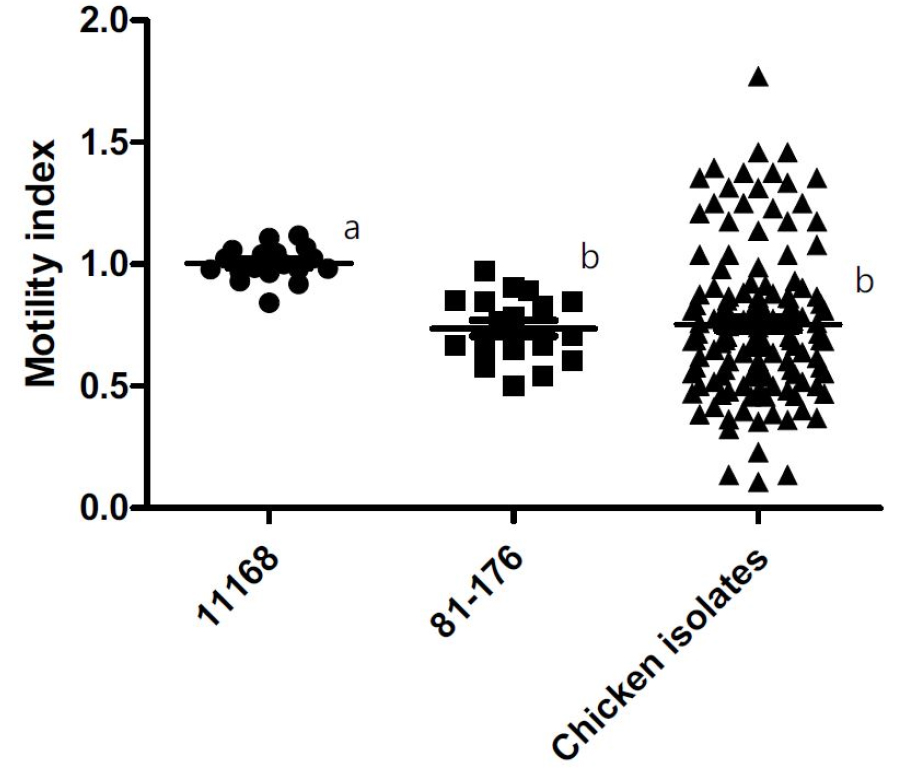
Fig. 1
Motility indices of Campylobacter isolates from retail raw chickens compared to two human clinical strains, C. jejuni 11168 and 81-176. The diameter of motility halo was measured in Mueller-Hinton soft agar (0.4%) after 48-h incubation at 37ºC under microaerobic conditions after inoculation. Forty-five isolates were tested in triplicate, totaling 134 data points. Each dot represents the motility index of each measurement. The motility index was calculated as the ratio of the diameter of the motility halo of each strain to the mean of the triplicate measurements of the control strain 11168. Each motility index of strain 11168 was calculated as the ratio of each measurement to the mean of the triplicate measurements of itself. Different lowercase letters next to the clusters of dots indicate significant differences (P < 0.05).
In conclusion, such high prevalence and motility of Campylobacter in retail chickens suggest that they pose a significant threat to public health in Korea.




