Introduction
Materials and Methods
Experimental materials
General component analysis
Dietary fiber content analysis
Mineral composition analysis
Measurement of total phenolic and total favonoid content
Measurement of DPPH radical scavenging activity
ABTS radical scavenging activity evaluation
Cell toxicity measurement used in the MTT assay
Tyrosinase inhibitory activity
Antibacterial test
Statistical analysis
Results and Discussion
Yield of S. tamariscina leaf extract
General ingredients and dietary fiber content
Mineral content
Total polyphenol and total flavonoid content
Measurement of DPPH radical scavenging activity
ABTS
Measurement of cell viability
Verification of tyrosinase inhibitory activity
Antibacterial test
Summary
Introduction
Oxidation continuously occurs within the body to supply energy. Reactive oxygen species (ROS) produced from oxygen are a subset of free radicals. Due to stress, smoking, alcohol consumption, and other factors, the amount of ROS reacting with lipids, DNA, and proteins gradually increases, which accelerates aging and contributes to conditions such as cancer, heart disease, and atherosclerosis (Crack and Aylor, 2005; Moskovitz et al., 2002). The most significant characteristic of modern bio-industry consumers is their emphasis on product safety and the objective efficacy of ingredients (Park and Kim, 2020). Therefore, the production of cosmetic ingredients using natural substances is actively being investigated. The use of herbal medicines, which include safe and effective natural substances that have been used for a long time for therapeutic purposes, is steadily increasing. Recently, antioxidant and bioactivity evaluations have been performed to identify functional chemical substances in herbal medicines through advances in biotechnology, and such substances are gaining attention as materials for use in various fields, such as the functional food and cosmetics industries (Kim and Lee, 2021). Due to heightened interest in health, research on natural functional substances, including functional components or bioactive substances, present in medicinal plants and crude drugs is increasing (Joung et al., 2007). Bioactive substances are high-value-added materials with increasing applications across numerous fields, and new substances are being researched and developed (Tabance et al., 2001).
Selaginella tamariscina is a fern belonging to the Selaginellaceae family. The entire plant, known as “gwonbaek,” has been used medicinally to treat menstrual pain, chronic hepatitis, hematuria, and uterine bleeding. Recent studies have confirmed that methanol extracts improve dementia in scopolamine-induced dementia model rats by protecting against oxidative stress (Chu et al., 2016) and exhibit antioxidant and anti-hyperlipidemic effects in diabetic model rats (Zheng et al., 2013). Its antibacterial activity against pathogenic bacteria such as gram-negative Escherichia coli and gram-positive Staphylococcus aureus (Choi et al., 2019; Hwang et al., 2013), and efficacy in inhibiting the proliferation of gastric cancer cells HT-29 have been reported (Lee et al., 1999). Additionally, among the species belonging to Selaginella, S. tamariscina exhibits the highest drought tolerance, shows resistance to desiccation under extreme drought conditions, and readily recovers upon water supply (Gu et al., 2019). The active ingredient in S. tamariscina is amentoflavone, which has been reported to inhibit amyloid-β in PC-12 nerve cells, exhibit anti-inflammatory and antioxidant effects, and inhibit apoptosis in hippocampal tissue (Zhang et al., 2015).
Therefore, this study analyzed the components contained in S. tamariscina leaves to identify those capable of exerting physiological activity within the body. S. tamariscina leaves has been traditionally used as a medicinal plant, and our study sought to determine whether it could be used as a new functional material. The findings will serve as a basis for evaluating the efficacy of S. tamariscina leaves in the future.
Materials and Methods
Experimental materials
S. tamariscina leaves cultivated in 2024 were purchased as a sample from the Herb Market located in Jegi-dong, Seoul. They were washed with distilled water, dried with hot air, and then ground to a particle size of 120 mesh or smaller for use in the general component analysis. Additionally, the prepared S. tamariscina leaves in a triangular flask were extracted by refluxing with 9 parts distilled water and 80% methanol for 4 h. The extract was filtered through cotton cloth and concentrated under reduced pressure (CCA-1100, Eyela, Tokyo, Japan) and subjected to rapid freeze-drying (-70°C, PVTFA 10AT, ILSIN, Suwon, Korea) to prepare a powder. This powder was used for various bioactive compound content analysis experiments.
General component analysis
General components were analyzed according to the AOAC method (AOAC, 1990). Specifically, the moisture content was analyzed using the 105°C atmospheric pressure drying method and the ash content was analyzed using direct combustion method at 550°C. The crude protein content was analyzed using a protein analyzer (Kjeltec protein analyzer, Tecator Co, Hoeganaes, Sweden) employing the micro-Kjeldahl method, while the crude fat content was analyzed using the Soxhlet method. The total carbohydrate content was calculated as the sum of the above measurements minus 100.
Dietary fiber content analysis
Total dietary fiber (TDF) content was analyzed using the enzymatic-gravimetric method according to the AOAC method (AOAC, 1995). Specifically, the dry powder sample was liquefied using heat-stable thermo-α-amylase and then reacted sequentially with protease and amyloglucosidase to hydrolyze proteins and starch. The soluble dietary fiber in the solution was subsequently precipitated with ethanol. The solution was vacuum filtered into a pre-weighed crucible. The residue was washed with ethanol and acetone and dried, and the total dietary fiber content was determined by the weight difference before and after drying, excluding the protein and ash contents in the dry residue.
Mineral composition analysis
The mineral content (Ca, P, Mg, K, Na, Fe, Zn, and Mn) was analyzed according to the AOAC method (AOAC, 1984). Specifically, 1 g of powdered S. tamariscina leaves was placed in a calcination vessel, pre-calcined, and then calcined at 550°C for 2 h. After adding approximately 10 mL of distilled water to moisten the sample, 3–4 mL of 50% nitric acid was added. Heat was applied to evaporate the excess nitric acid, after which the mixture was heated for an additional hour in the retort. After heating, hydrochloric acid was added in a 1:1 ratio to dissolve the substance. The solution was then transferred to a 50 mL volumetric flask and diluted to volume with distilled water. The inorganic composition of this solution was analyzed using an inductively coupled plasma emission spectrometer (Atom Scan 25, Thermo Jarrell Ash Co, Franklin, MA, USA). The analysis conditions were as follows: approximate RF power was 1,150 W, analysis pump rate was 100 rpm, nebulizer pressure was 30 psi, and observation height was 15 mm.
Measurement of total phenolic and total favonoid content
Total phenols were measured according to the Folin-Denis method (Folin and Denis, 1912). After adding 100 µL of sample solution diluted at various concentrations to 100 µL of Folin-Ciocalteu phenol reagent, the mixture was reacted at room temperature for 3 min. Subsequently, 100 µL of Na2CO3 solution was added, and the absorbance was measured at 760 nm using an ELISA reader, followed by calculating the average polyphenol content for each concentration. For quantitative analysis, gallic acid was used as the standard substance for the calibration curve. The total flavonoid content was measured using a method partially modified from Kim et al. (2011). To 0.5 mL of S. tamariscina leaf extract, 1.5 mL of 95% ethanol, 0.1 mL of 10% aluminum nitrate, 1 M potassium acetate 0.1 mL, and distilled water 2.8 mL were added. The mixture was reacted at room temperature for 30 min. The absorbance was measured at 415 nm using a microplate reader. The total flavonoid content was determined using a rutin standard curve.
Measurement of DPPH radical scavenging activity
The DPPH radical scavenging activity is a method for measuring antioxidant activity through electron transfer. It utilizes the principle that when the DPPH radical is reduced by receiving electrons or hydrogen from aromatic compounds or aromatic amines to form a stable molecule, the purple color is decolorized. This method is widely used to screen for antioxidants from various natural materials and is a simple way to measure antioxidant capacity in a short time. Free radical scavenging activity using 2,2-diphenyl-1-picrylhydrazyl (DPPH) was measured by the Blois (1958) method. To this end, 0.1 mL of a 0.2 mM DPPH solution was added to 0.1 mL of the sample, reacted at 37°C for 30 min, and then the absorbance was measured at 517 nm.
ABTS radical scavenging activity evaluation
The ABTS radical cation is a deep blue-green radical that decolorizes to a light green color upon reacting with antioxidants. ABTS is an antioxidant analysis method that measures hydrogen ion donating capacity, and it quantifies antioxidant effects by measuring the degree to which the ABTS free radical—generated by the reaction of potassium persulfate—is quenched by antioxidants, causing the radical’s characteristic blue-green color to fade to a pale green (Natella et al., 2014). The measurement of antioxidant capacity using the ABTS radical is based on the principle that the ABTS free radical is scavenged by antioxidants in the sample, causing the blue-green color to fade (Cho et al., 2013). A 7 mM ABTS solution and a 2.45 mM potassium persulfate solution were reacted in the dark for 16 h to generate radicals. Subsequently, 10 µL of sample and 290 µL of ABTS solution were added to a 96-well microplate, mixed, and reacted in the dark for 6 min. The absorbance was measured at 734 nm, and the scavenging activity was calculated (Re et al., 1999).
ABTS Radical Scavenging Ability (%) = (1 - Absorbance of sample group / Absorbance of control group) × 100
Cell toxicity measurement used in the MTT assay
In this experiment, RAW264.7 cells were seeded at 4 × 104 cells per well in a 96-well plate and cultured for 24 h. After replacing the plates, they were treated with LPS at 100 ng/mL and S. tamariscina leaf extracts at various concentrations. After incubating for 24 h and removing the medium, 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT, 0.5 mg/mL, Sigma Aldrich, St. Louise, MO, USA) was added, and the mixture was further cultured for 4 h. The medium was removed, and dimethyl sulfoxide (DMSO) was added to each well to dissolve the MTT formazan, followed by measuring the absorbance at 570 nm (Mosmann, 1983).
Tyrosinase inhibitory activity
Mushroom tyrosinase-150 unit (Sigma-Aldrich) at 150 µL, L-tyrosinase at 225 µL (2.5 mM), 0.4 M HEPES buffer at 225 µL (pH 6.8), and ethanol at 300 µL were combined. After mixing the solution, the absorbance at 475 nm was measured before and after the reaction (Kim et al., 2006). The degree of tyrosinase inhibition was calculated as follows.
where A and B represent the absorbance of solutions containing each sample before and after incubation and C and D represent the absorbance of the blank solution (reference solution) before and after incubation.
Antibacterial test
Antibacterial activity was tested using the paper disc method (Davidson and Parish, 1989). The microbial strains used for antimicrobial activity were obtained from the Korean Collection for Type Cultures Biological Resource Center (KCTC/BRC, Jeongeup, Korea) and the Korean Culture Center of Microorganisms (KCCM, Seoul, Korea). The strains and numbers of each microbial strain used in the experiment are presented in Table 1. The cultured strain was adjusted to 1.0 × 106 CFU/mL before use in this experiment. Each strain cultured on agar plates was prepared by streaking 100 µL of each culture. Extracts were applied at concentrations of 0.5 to 10 mg/mL, 40 µL each, onto paper discs (diameter 8 mm, Toyo Roshi Kaisha Ltd., Tokyo, Japan), followed by culture at 37°C for 24 h. Ethanol was used as the control group, and the clear zone (mm) formed around the disc after incubation was measured to compare antimicrobial activity.
Table 1
List of strains and cultivation condition used for antimicrobial experiments
Statistical analysis
Results obtained from repeated measurements (three or more times) were analyzed using one-way ANOVA with SPSS (Statistical Package for the Social Sciences, Version 26.0, IBM-SPSS Inc., Chicago, IL, USA). The significance between each measured mean was verified at the p < 0.05 level using Duncan’s multiple range test.
Results and Discussion
Yield of S. tamariscina leaf extract
The extraction yield was calculated by measuring the weight of the final powder obtained after extracting 100 g of dry sample with hot water and 80% methanol, concentrating under reduced pressure, and freeze-drying. The results are shown in Table 2. The hot-water extract (WST) yielded 8.6954%, while the 80% methanol extract (MST) yielded 12.3692%, indicating that the extraction conditions using 80% MST produced a higher yield. According to the findings of Zheng et al. (2011), the yield of WST was 12.13%, while the yield of the 70% MST was 8.17%. Although these results showed differences in yield depending on the extraction solvent compared to this study, the highest yield was obtained by using methanol as the solvent. Therefore, the final yield varied depending on the solvent and extraction method used for the same material.
Table 2
Extraction yields of hot-water and 80% methanol extracts from Selaginella tamariscina
| Sample | Yield (%) |
| WST1) | 8.695 ± 0.51a,4) |
| MST3) | 12.369 ± 0.42b) |
General ingredients and dietary fiber content
The results of the general component analysis of S. tamariscina leaves are shown in Table 3. Per 100 g of dried leaves, the composition was as follows: moisture 7.21%, crude fat 1.32%, crude ash 12.24%, crude protein 13.20%, and carbohydrates 66.03%. The main component of S. tamariscina leaves was identified as carbohydrates, a constituent of plant bodies, while the lowest component was neutral fat. Compared to the findings of Shin (2017) for Hippophae rhamnoides leaves, the component analysis of S. tamariscina leaves yielded results similar to the nutritional components of H. rhamnoides leaves. Furthermore, the total dietary fiber content was 31.25% and primarily consisted of insoluble dietary fiber—a structural polysaccharide and structural non-sugar present in the cell walls of plants or seeds, which are the main components of the cell walls—and soluble dietary fiber present in the cell membranes or the multilayer between the cell walls and cell membranes. These result indicate that further research on the composition of insoluble and soluble dietary fiber should be conducted to investigate the functionality of food materials.
Table 3
Proximate compositions of the Selaginella tamariscina leaves
| Nutrients | Selaginella tamariscina leaves | |
|
General Nutrients (%) | Moisture | 7.21 ± 0.721) |
| Carbohydrate2) | 66.03 ± 0.62 | |
| Crude protein | 13.20 ± 1.36 | |
| Crude fat | 1.32 ± 0.95 | |
| Crude ash | 12.24 ± 1.74 | |
| Dietary fiber | 31.25 ± 0.32 | |
Mineral content
The mineral content results for S. tamariscina leaves are shown in Table 4. The potassium content was highest at approximately 856.32 mg, followed by the calcium (214.49 mg), magnesium (73.25 mg), phosphorus (80.35 mg), and sodium (41.32 mg) contents. The micronutrient iron, manganese, and zinc contents were 3.28 mg, 0.83 mg, and 2.69 mg, respectively, which was similar to the nutritional component results for Adenophora triphylla leaves (Cha et al., 2025). Potassium, in particular, is responsible for important physiological functions, including energy metabolism, transport across cell membranes, maintaining voltage differences across cell membranes, nerve stimulation through interaction with sodium, skeletal muscle contraction and relaxation, blood pressure maintenance, and acid-base balance (Suter, 1998). Potassium intake has been reported to be effective in the prevention and treatment of hypertension (Cappuccio and MacGregor, 1991).
Table 4
Mineral contents in Selaginella tamariscina leaves (mg/kg)
| Mineral | Selaginella tamariscina leaves |
| Ca | 214.49 ± 1.57 |
| Mg | 73.25 ± 1.39 |
| Na | 41.32 ± 0.88 |
| K | 856.32 ± 0.92 |
| P | 80.35 ± 2.61 |
| Fe | 3.28 ± 0.85 |
| Zn | 2.69 ± 0.78 |
| Mn | 0.83 ± 0.33 |
Total polyphenol and total flavonoid content
Polyphenolic compounds, which are secondary metabolites found in natural plants, act as antioxidants and exhibit effects against cancer, bacteria, diabetes, and obesity (Sinha, 2019). The results of measuring the total polyphenol and total flavonoid contents of the WST and MST of S. tamariscina leaves are shown in Table 5. The total polyphenol content of the WST and MST was 43.25 mg GAE/g and 67.32 mg GAE/g, respectively, indicating that the MST yielded a higher polyphenol content. Furthermore, the total flavonoid content was also higher in the MST at 16.80 mg RE/g compared to the WST. Differences in the cultivation environment of the samples or variations in the methods used to measure total flavonoids and flavonoid content likely resulted in these differing values.
Table 5
Total polyphenol and flavonoid content of Selaginella tamariscina leaves
| Sample | WST1) | MST2) |
| Total Phenol contents (GAE3) mg/g) | 43.25 ± 8.32a) | 67.32 ± 5.20a,5) |
| Total Flavonoid contents (RE4) mg/g) | 12.33 ± 0.09 b) | 16.80 ± 3.64b) |
Measurement of DPPH radical scavenging activity
To measure DPPH radical scavenging activity, the DPPH scavenging activity of the WST at concentrations of 50.0, 100.0, 200.0, and 500.0 µg/mL was 10.32, 18.39, 38.32, and 80.21%, respectively, while that of MST was 20.24, 39.44, 78.95, and 93.34%, respectively. DPPH radical scavenging activity increased with increasing concentration (Fig. 1). The antioxidant efficacy results were similar to that of Centella asiatica extract under extraction conditions (Kim et al., 2019). Thus, S. tamariscina leaves may also exhibit excellent anti-aging effects against free radicals.
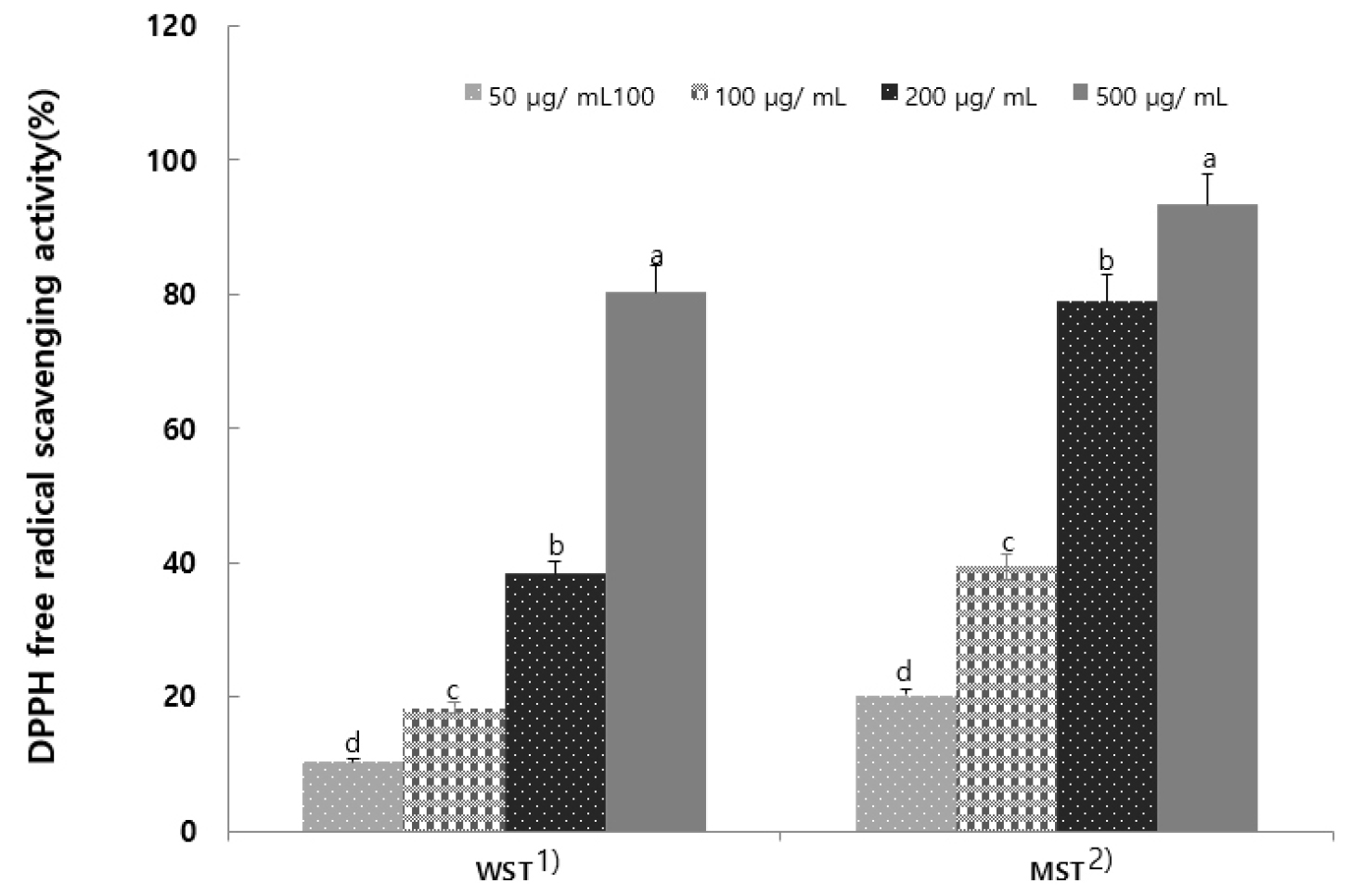
Fig. 1.
DPPH radical scavenging activity of hot-water and 80% methanol extracts from Selaginella tamariscina (Results are presented as the mean ± SD of 3 independent in triplicate. Means with different letters on the same kind of bars are significantly different at p < 0.05 by Duncan’s multiple range test. 1) Selaginella tamariscina hot water extract; 2) Selaginella tamariscina 80% methanol extract).
ABTS
Measuring antioxidants using the ABTS cation radical is an effective method for assessing the antioxidant capacity of hydrophilic and hydrophobic substances because it relies on the characteristic decolorization of the blue-green color. The DPPH radical scavenging activity utilizes the elimination of free radicals, whereas the ABTS radical scavenging activity utilizes the elimination of cationic radicals (Miller et al., 1993). The ABTS radical scavenging activity of S. tamariscina leaf extract is shown in Fig. 2. Both the WST and MST showed increased ABTS radical scavenging activity with increasing extract concentration, similar to the DPPH scavenging results. The MST demonstrated superior antioxidant activity compared to the WST.
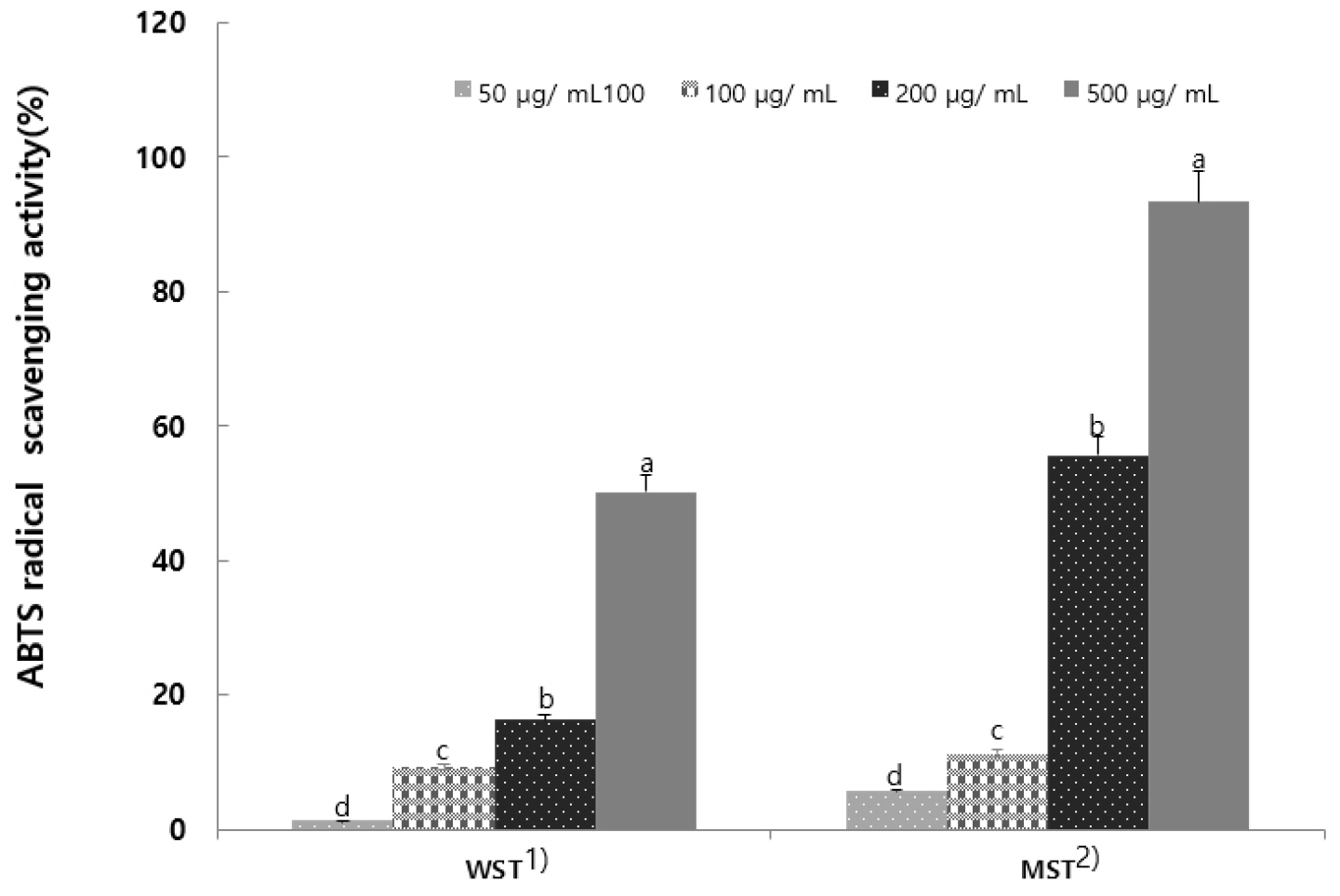
Fig. 2.
ABTS radical scavenging activity of hot-water and 80% methanol extract from Selaginella tamariscina (Results are presented as the mean ± SD of 3 independent in triplicate. Means with different letters on the same kind of bars are significantly different at p < 0.05 by Duncan’s multiple range test. 1) Selaginella tamariscina hot water extract; 2) Selaginella tamariscina 80% methanol extract).
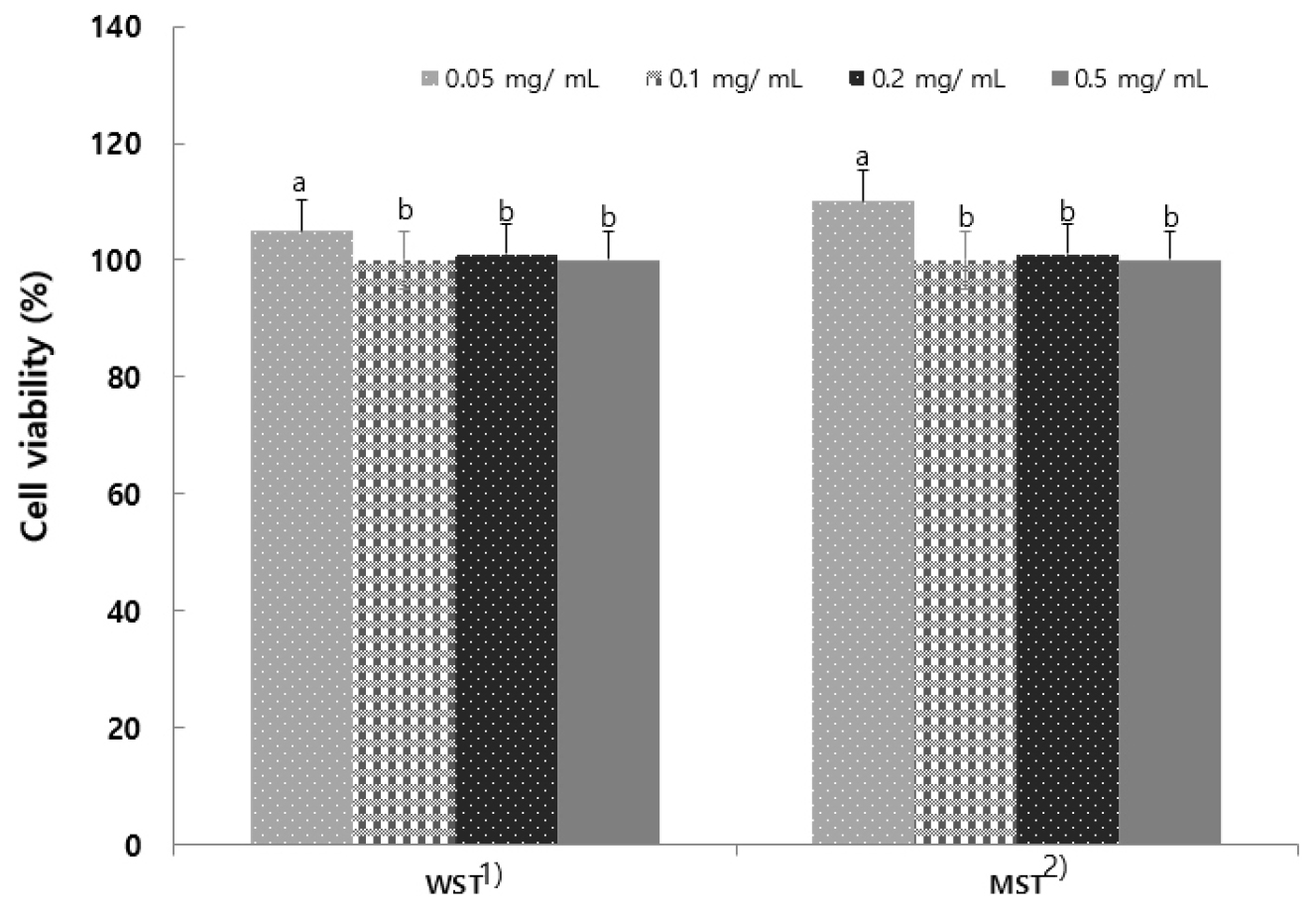
Measurement of cell viability
The MTT assay, widely used in cellular studies, is highly useful for in vitro analysis of cell proliferation and viability. The viability of RAW 264.7 cells treated with S. tamariscina extract was assessed (Fig. 3), and the results indicated that both extracts showed a 5.2% reduction at concentrations of 0.05, 0.1, 0.2, and 0.5 mg/mL, with no significant difference in the cytotoxicity of either extract. When the extracts obtained using six different solvents were treated with RAW264.7 cells to investigate cytotoxicity (Shim et al., 2018), the results were similar to the report indicating no cytotoxicity.
Verification of tyrosinase inhibitory activity
A key factor in the melanin production process is tyrosinase within the skin. This enzyme forms melanin from dihydroxyindole (DHI) during the melanin biosynthesis process, which begins with the conversion of the amino acid tyrosine to 3,4-dihydroxyphenylalanine (DOPA), DOPA to DOPA quinone, and DOPA quinone to melanin (Choi and Shin, 2015). Tyrosinase inhibitory activity was measured for S. tamariscina leaf extract (Fig. 4). It exhibited concentration-dependent inhibition of activity. A study investigating the tyrosinase inhibitory activity of Moringa oleifera extract reported an inhibition rate of 15.31% (Lim et al., 2024), and numerous studies on tyrosinase inhibition and whitening effects by natural products are also underway (Kim et al., 2018).
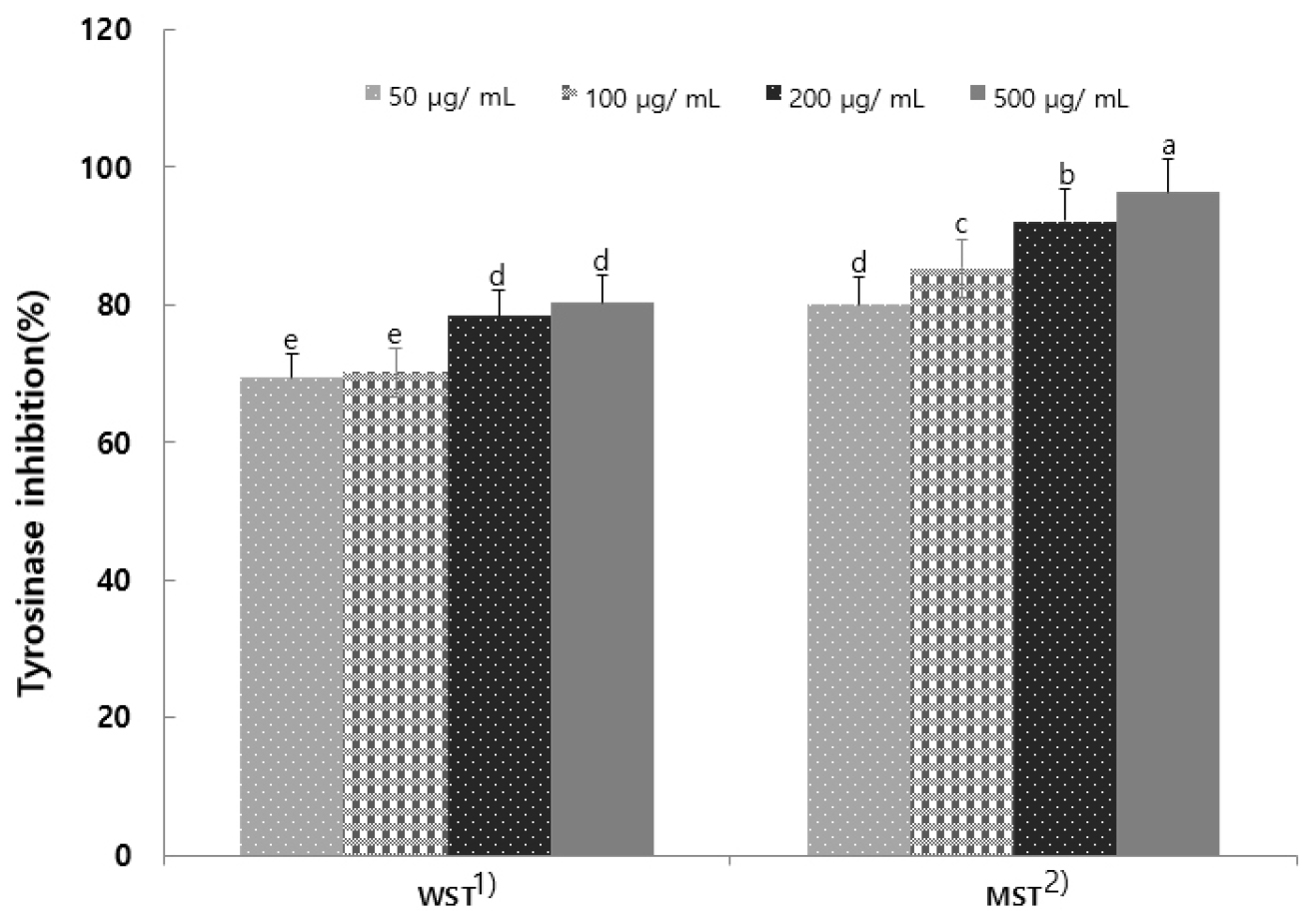
Fig. 4.
Tyrosinase activity of hot-water and 80% methanol extracts from Selaginella tamariscina. Results are presented as the mean ± SD of 3 independent in triplicate. Means with different letters on the same kind of bars are significantly different at p < 0.05 by Duncan’s multiple range test. 1) Selaginella tamariscina hot water extract; 2) Selaginella tamariscina 80% methanol extract.
Antibacterial test
The rapid proliferation of skin flora can cause or exacerbate skin inflammation. In the context of inflammatory skin diseases, the body’s defense function against harmful microorganisms weakens, leading to a loss of skin barrier function. This renders the skin vulnerable to microbial infection and can further exacerbate chronic inflammatory skin diseases (Lee et al., 2006). S. tamariscina leaf extract demonstrated antibacterial activity at all experimental concentrations. In particular, S. aureus, a yellow staphylococcus, is a bacterium that exists asymptomatically but is a pathogenic microorganism. It is a representative pyogenic bacterium found in the nose, skin, armpits, and other areas. The antibacterial effect of the S. tamariscina leaf extract against S. aureus was confirmed to be quite high, and the MST showed a larger clear zone compared to the WST (Table 6).
Summary
This study investigated the potential use of S. tamariscina leaves as a functional food and food additive ingredient by analyzing the nutritional composition from a food nutrition perspective and examining related functional properties that may exhibit physiological activity. The general composition of S. tamariscina leaves based on the dry weight was 66.03% carbohydrates, 13.20% crude protein, 1.32% crude fat, and 12.24% crude ash, with a total dietary fiber content of 31.25%. The total polyphenol content extracted with WST and 80% MST was 43.25 mg/g and 67.32 mg/g, respectively, while the total flavonoid content was 12.33 mg/g and 16.80 mg/g, respectively. The 80% MST results also demonstrated superior activity in terms of antioxidant capacity and antibacterial activity. Based on these results, the 80% MST of S. tamariscina leaves was confirmed to possess antioxidant capacity, suggesting its potential for use as a food additive or for other medicinal purposes.