Introduction
Methodology
Seed source and priming agent
Seed sterilization and priming
Seed germination
Germination and morphological parameters evaluated
qRT-PCR analysis for gene expression
Statistical analysis
Results
Analysis of the variance of the parameters
Effect of KNO3 priming on germination parameters
Effect of KNO3 priming on seedling morphological parameters
Gene expression analysis
Discussion
Introduction
Soybean [Glycine max L. (Merr.)], an important legume crop known for its high protein supplement, is grown widely around the globe as a good source of food for both human and domesticated animals (Foyer et al., 2016; Hartman et al., 2011). However, the production of soybeans has been limited due to factors like poor germination, poor seedling establishment, and poor seedling growth (Hamman et al., 2002; Sedibe et al., 2023). To solve the above-mentioned limiting factors, seed priming, a pre-sowing technique, has gained attention as an effective method for improving crop establishment, seedling vigor, and stress tolerance to address the mentioned limitations (Chakraborty and Dwivedi, 2021). Seed priming involves soaking seeds in water or a chemical solution for a predetermined amount of time and then drying them back to storage moisture contents before planting (Bujalski et al., 1991). Priming prevents radicle protrusion by carefully hydrating and dehydrating seeds and starts the germination process, and stimulates the seed in several physiological and biochemical ways, increasing its receptivity to outside stimuli like nutrients and water (Ali et al., 2020). Compared to emergence from unprimed seeds, seedling emergence from primed seeds occurs more quickly, and this process of priming has been a successful method for enhancing crop establishment, seedling vigor, and stress tolerance (Lutts et al., 2016). The external application of priming agents to seeds plays a significant role in preparing for germination phases before sowing (Moaaz et al., 2020). This technique of seed priming was first described by (Heydecker et al., 1973). Seed germination is enhanced by priming under three different stages: imbibition, germination, and growth (Waqas et al., 2019). Different techniques of priming like hydropriming (use of water), halopriming (use of salt like sodium chloride), hormonal priming (use of phytohormones), solid-matrix priming (use of solid planting materials), osmotic priming (use of osmoticums like potassium chloride, potassium nitrate (KNO3)) has crucial benefits for the crop establishment (Devika et al., 2021; Ruttanaruangboworn et al., 2017).
KNO3, a commonly used macronutrient fertilizer, is known to boost plant growth and resilience to stress (Johnson et al., 2022). Various studies have demonstrated the beneficial effects of KNO3 when used as a priming agent on seedling establishment and growth. It has been proven to be beneficial in improving seed germination and seedling establishment in tomato (Solanum lycopersicum L.) (Moaaz et al., 2020), rice (Oryza sativa L.) (Javed et al., 2020), chickpea (Cicer arietinum L.) (Choudhury and Bordolui, 2022), mung bean (Vigna radiata L.) (Entesari et al., 2012), onion (Allium cepa L.) (Jima et al., 2015), and common bean (Phaseolus vulgaris L.) (Sarmadi et al., 2014) under stressed or normal conditions. While the potential benefits of seed priming with KNO3 are well recognized, there is a notable lack of research on optimizing the concentration of the priming agent and the duration of priming specifically for soybean cultivars. Therefore, a comprehensive investigation into the optimal priming durations and concentrations of KNO3 specifically tailored to soybean cultivars is essential. This research aims to bridge this knowledge gap of finding the appropriate concentration and priming duration of soybean seeds by systematically evaluating the effects of KNO3 priming on germination and early seedling growth of soybeans.
Methodology
Seed source and priming agent
The entire experiment was conducted in the laboratory of Kyungpook National University, South Korea. Daechan and Pungsannamul, two different soybean varieties, were used for the study because these two popular varieties are widely cultivated in Korea across almost all the provinces (Baek et al., 2020; Yoon et al., 2021). The potassium nitrate (KNO3, Sigma Aldrich, USA) was used as a priming agent.
Seed sterilization and priming
To mitigate the risk of microbial contamination, soybean seeds were surface sterilized for ten minutes in a 0.5% v/v (volume/volume) sodium hypochlorite solution (NaOCl, Sigma Aldrich, USA). Subsequently, the seeds were rinsed three times with distilled water to ensure the removal of any residual sterilant. Seeds were subjected to immersion in KNO3 solution at concentrations of 25 mM, 50 mM, and 100 mM for priming durations of 1, 2, 3, 4, and 5 hours. The seeds were also subjected to hydropriming, immersed solely in distilled water, and constituted the control group for this study (0 mM of the treatment solution). The seed-to-solution volume ratio was consistently held at 1:5 (weight/volume) (Khan et al., 2019). To facilitate optimal germinating conditions, the seeds were then dried in an oven at 25 °C for 48 hours to eliminate excessive moisture after being kept in the priming solutions. The experiment was performed in a completely randomized design (CRD) with four replications.
Seed germination
Post-priming, the seeds were placed in a growth chamber to ensure optimal germination conditions, maintained at 25 ± 2°C, with relative humidity ranging from 50 to 70%, under a 12-hour light/12-hour dark photoperiod regime (Raina et al., 2013). A number of 25 seeds from each treatment group were arranged in 9-cm-diameter Petri dishes, each lined with two layers of moistened tissue paper. The seeds were oriented with their radical ends pointing downward to ensure uniform germination. To maintain the optimal moisture level of the germination medium, 10 ml of distilled water was added daily (Abbasi et al., 2019). Petri dish lids were secured to prevent water evaporation. Germinated seeds were counted starting at 12 hours and subsequently every 12 hours up to 120 hours, or 5 days. After the 120-hour germination period, specific characteristics were quantified using predefined formulas, and different germination traits were measured by selecting 10 random seedlings.
Germination and morphological parameters evaluated
Seven different germination and seedling morphological parameters were evaluated in the study as listed in Table 1. The germination percentage (GP) was evaluated on the final day (120 hrs). Here, the term germination was defined as the emergence of a radicle approximately 2 mm in length (Lamichhane et al. 2020). Similarly, mean germination percentage (MGT) means the time taken (days or hours) for average germination of the seeds; higher value of MGT means more time is taken for the seeds to germinate (Dezfuli et al., 2008; Ellis and Roberts, 1981). The seed vigor index (VI) is one of the crucial traits that affects the seedling establishment and growth and has a close relation with standard germination (Kim et al., 1994). The radicle length (RL) was measured from the junction of the hypocotyl and radicle to the root cap using a ruler. The plumule is the embryonic shoot of the plant and the plumule length (PL) was measured from the collar region of the seedling to the apex using a ruler. For the seedling's dry biomass (SB), they were first dried in an oven at 70 °Celsius for 72 hours and weighed using a balance (Ajouri et al., 2004).
Table 1.
Measurement of different seed germination parameters
| Parameters | Formula | Units | Reference |
| Germination % | GP = × 100 % | % | (Zahedifar and Zohrabi, 2016) |
| Mean germination time | MGT = | days | (Ellis and Roberts, 1981) |
| Vigor index | VI = GP × k | ----- | (Kulkarni et al., 2007) |
| Germination rate index | GRI = | ----- | (Nciizah et al., 2020) |
| Radicle length | Measured with ruler | cm | (Ajouri et al., 2004; Lee et al., 1998) |
| Plumule length | Measured with ruler | cm | |
| Seedling dry biomass | Measured with weighing balance | gm |
A workflow of the study has been presented in Fig. 1. The sterilized seeds were primed at different concentrations and different time intervals, and then they were subjected to dry for initial moisture content. Thus, obtained seeds were then experimented for germination in the Petri dish by placing them in the plant growth chamber. The final germination and morphological data were then recorded.
qRT-PCR analysis for gene expression
We chose Daechan seeds primed with 50 mM solution for 3 hours and Pungsannamul cultivar seeds primed with 100 mM solution for 4 hours based on the results. For the analysis of gibberellic acid (GA) and abscisic acid (ABA) related gene expression levels, a qRT-PCR analysis was done. The detailed methodology of qRT-PCR has been described by Methela et al. (2023). The seeds were first ground into fine powder in liquid nitrogen, and ribonucleic acid (RNA) was extracted through TRIzol® reagent (Invitrogen, USA). Using 1 µg of RNA as starting material, complementary deoxyribonucleic acid (cDNA) was synthesized based on the manufacturer’s protocol using BioFact™ RT-Kit (BioFact, Korea). Following that, a two-step PCR was carried out utilizing an Eco™ real-time PCR machine (BIO-RAD, CFX Duet, Real-Time PCR system, Singapore). The PCR reaction employed a 2× Real-Time PCR Master Mix containing SYBR® Green I (SolgTM, Korea). The procedure involved an initial denaturation step at 95 °C for 15 minutes for 40 cycles, each consisting of denaturation at 95 °C for 20 seconds, and simultaneous primer annealing and extension at 58 °C for 40 seconds. The list of the primers used along with their GeneID has been tabulated in (Table 2) which has been mentioned in our recent study (Steven et al., 2024). Tubulin gene (GmTubulin) was used for the comparison as a reference gene.
Table 2.
Primers used for gene expression analysis
Statistical analysis
All the graphs for the comparison were made in Microsoft Excel (2019). Similarly, an analysis of variance (ANOVA) was done to determine the statistical significance using the SAS program (SAS release 9.4; SAS, Gary, NC, USA). Duncan's multiple range test was utilized to compare the markedly distinct means across various priming concentrations and priming durations, with a significance level set at p < 0.05.
Results
Analysis of the variance of the parameters
ANOVA analysis revealed that all the observed parameters, final germination percentage (FGP), RL, PL, VI, SB, GRI, and MGT showed significant differences (p < 0.001) between concentration and duration of priming (Table 3). There was a significant difference among these parameters within the two selected varieties of soybean except for PL (p < 0.726). Similarly, the interaction of concentration of the priming agent and duration of priming also showed significant differences for all the parameters except RL (p < 0.0854), PL (p < 0.8961), MGT (p < 0.8236) and GRI (p < 0.2762). In summary, the ANOVA results affirm that varying concentrations of KNO3 priming, coupled with different durations of priming, exert a significant influence on germination parameters.
Table 3.
ANOVA analysis for various germination parameters
Effect of KNO3 priming on germination parameters
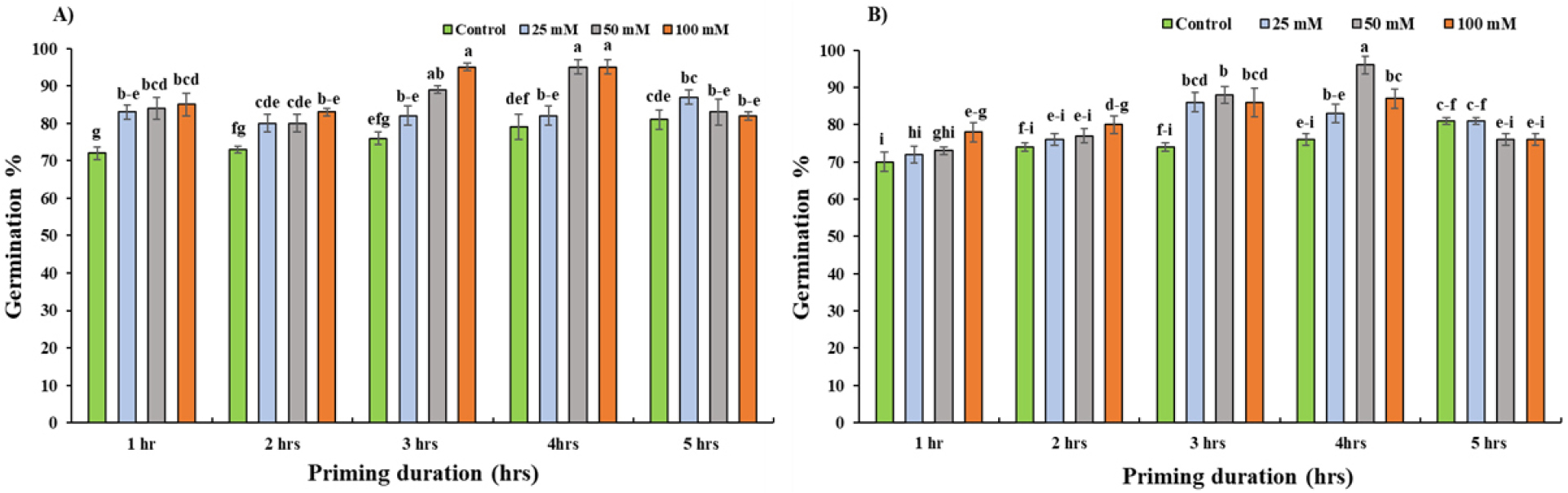
The KNO3 priming had a significant effect on GP when compared to control in almost every concentration and duration of priming except for 5 hours for Daechan and Pungsannamul and 2 hours of priming in Pungsannamul (Fig. 2). A general trend of increase in GP along with increase in priming concentration was observed with highest GP being 95% for Daechan and 96% Pungsannamul at 100 mM at 3 hours and 50 mM at 4 hours duration which was around 20% and 21% more than the controlled one in the same priming duration respectively. Compared with control priming at 50 mM and 100 mM for 3 and 4 hours had significant difference (p < 0.05) in GP for Daechan, whereas for Pungsannamul 50 mM for 4 hours stood the best among all other treatments (Fig. 2A) These two treatments combination (50 mM and 100 mM for 3 and 4 hours) had statistically significant difference compared with control for the first four hours for Daechan and third and fourth hour for Pungsannamul. However, on the highest duration of priming (5 hours), no such difference was observed between the control and treated groups i.e., there were no statistically significant differences. Interestingly, GP for Pungsannamul for control in the fifth hour of priming was higher by almost 6.5% compared to the latter two treatments (Fig. 2B).
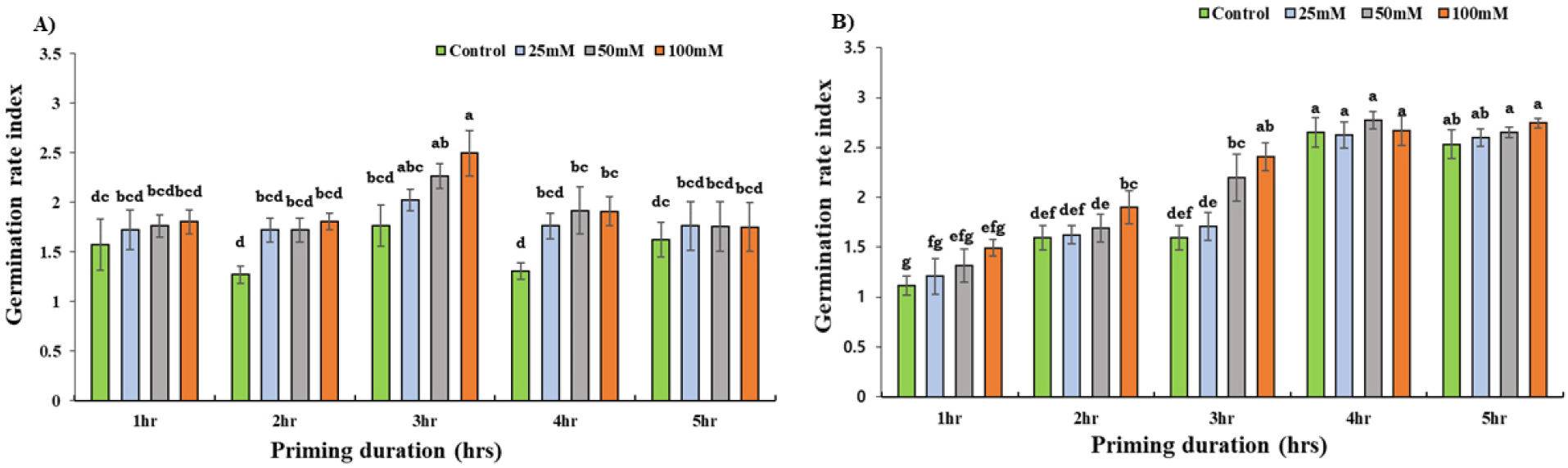
Results of the GRI were similar for both varieties in the control and treatment groups (Fig. 3). 3 hours of priming at 100 mM concentration of KNO3 had significantly (p < 0.05) higher GRI (2.49), 29% than that of the control (1.76). While 32% increased GRI was observed in 50 mM KNO3 application as compared to that of control in the 4 hours priming treatment for Daechan (Fig. 3A). For Pungsannamul, the groups subjected to priming durations of only 2 and 3 hours at 100 mM of KNO3 exhibited 25% and 34% increased GRI rates in comparison with that of control (Fig. 3B) All concentrations primed for later durations (4 and 5 hours) yielded highest GRI for Pungsannamul with an average GRI of 2.65.
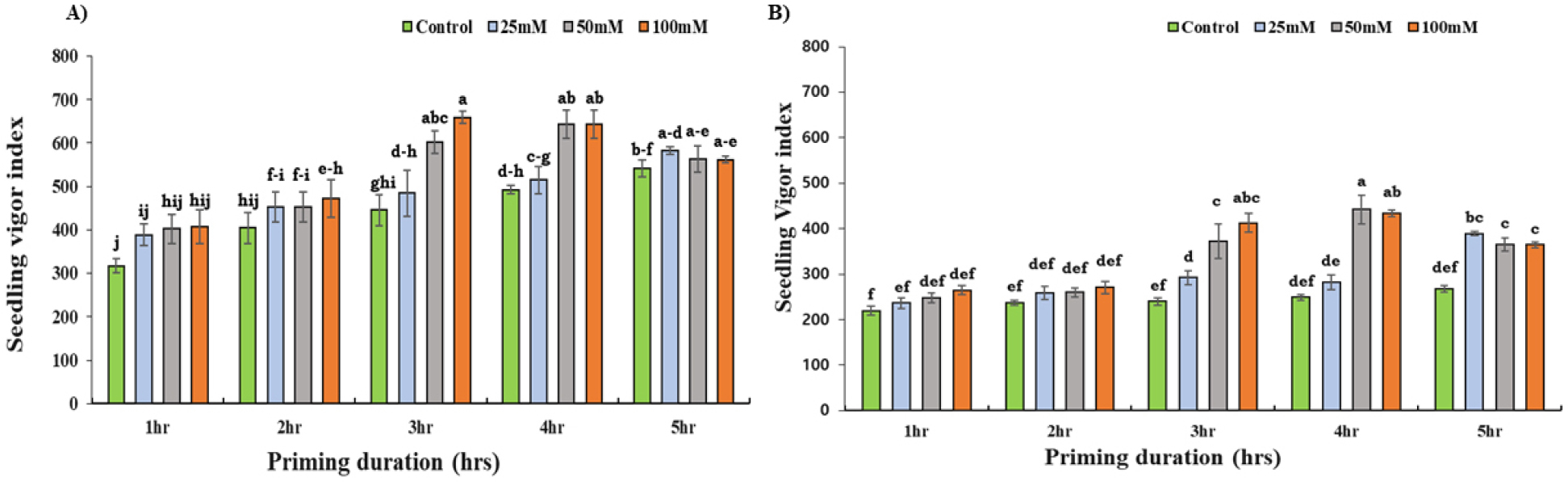
Variation in VI was observed, with generally higher VI for Daechan compared to Pungsannamul (Fig. 4). Within Daechan, seeds primed for 50 mM and 100 mM of KNO3 were significantly high (p < 0.05) in VI compared with control. These priming agents increased the VI by 40% and 23% than the control for the third and fourth hours of priming; highest among the other combinations of treatments and durations (Fig. 4A). Similar to Daechan, for Pungsannamul, the same concentrations (50 mM and 100 mM) resulted in significantly higher VI compared to the control group, with a 36% increase in VI observed under both conditions following the same duration of priming (Fig. 4B).
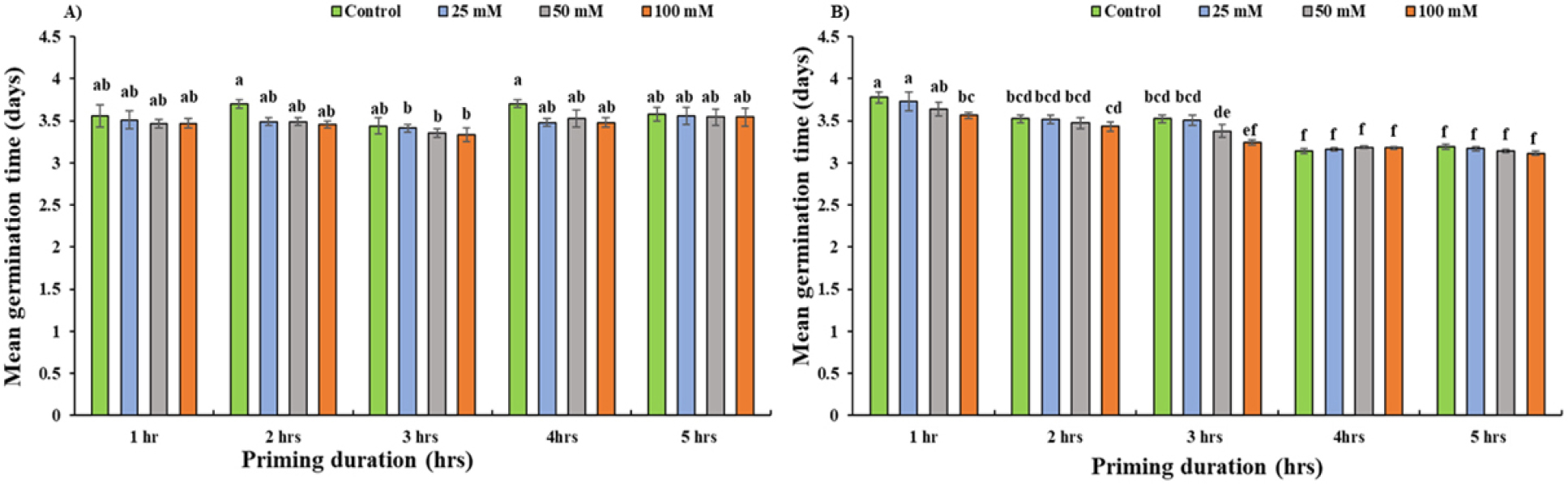
No significant differences (p < 0.05) were observed in MGT across all the treatments and priming duration in Daechan (Fig. 5A). On average controlled group had 6% higher MGT than the treatment groups in the 2 and 4 hours of priming duration although there were no significant differences (Fig. 5A). Similar result was observed in Pungsannamul, where no significant differences were observed in MGT within the different priming duration and priming concentration. However, MGT in the treatments of the first three priming durations was significantly higher than in the latter two priming durations (Fig. 5B).
Effect of KNO3 priming on seedling morphological parameters
For morphological parameters, RL, PL, and SB were measured using the method described above.
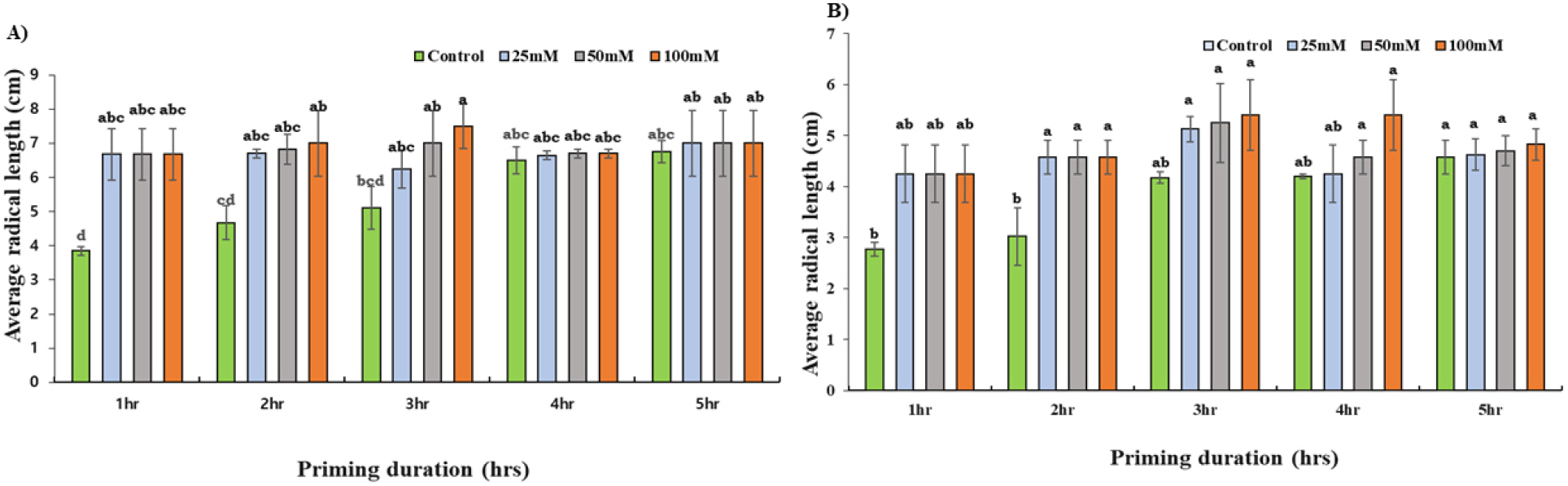
In comparison with control, the RL increased by 30% and 33% for 50 mM and 100 mM at the 2 hours duration of priming and by 27% and 32% for the 3 hours of priming for Daechan (Fig. 6A). Similarly, in Pungsannamul all the three concentrations (25 mM, 50 mM, and 100 mM) increased the RL by around 34% compared to the controlled group when primed for 1 hour and 2 hours (Fig. 6B). The next three duration of priming had no significant differences in the RL.
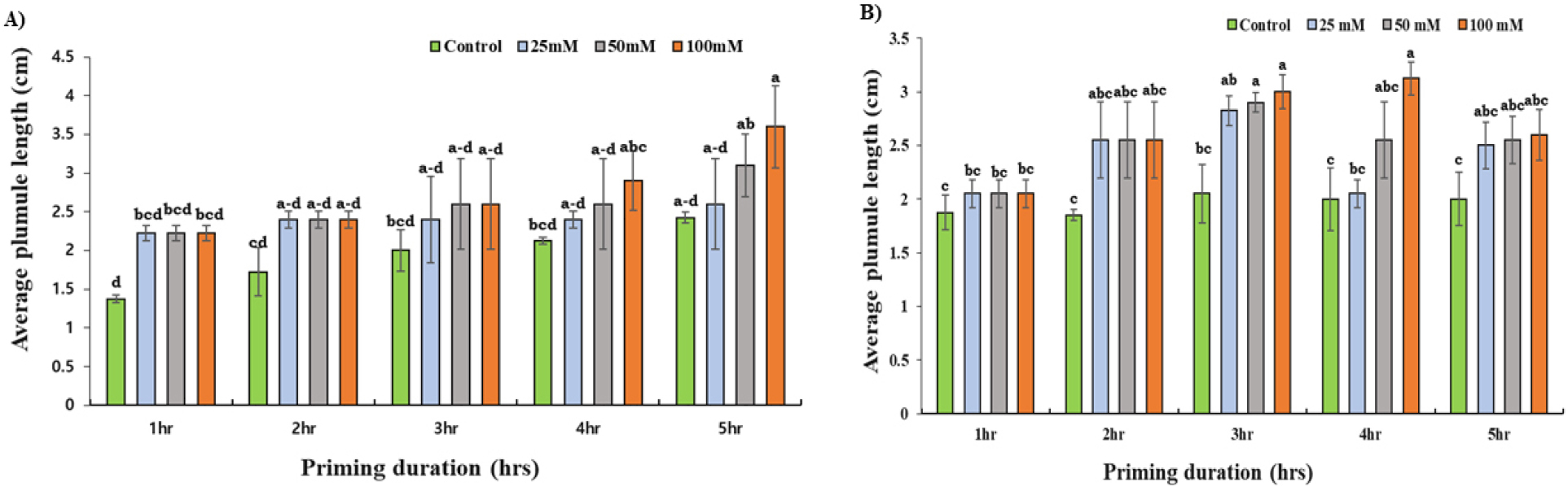
A prominent increase in PL was not observed in the Daechan variety across the different durations of priming (Fig. 7A). The three concentrations of KNO3 increased the PL by 34% and 28% for 2 and 3 hours of priming duration. However, statistical significance (p < 0.05) was merely observed. Priming Pungsannamul at concentrations of 50 mM and 100 mM for 3 hours, as well as 100 mM for 4 hours, resulted in a significant increase in PL (Fig. 7B). On average, 31% and 36% (highest) of increment was observed in 2 hours and 3 hours duration of priming respectively.
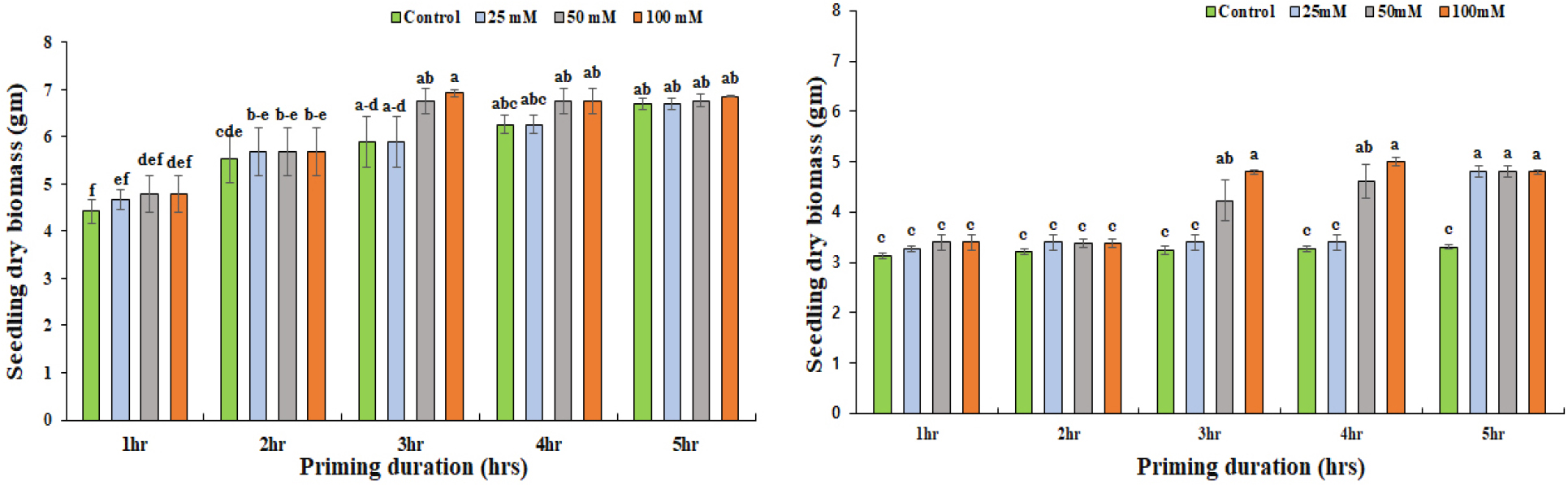
SB within the same duration of priming did not show significant differences (p < 0.05) between the control and the treatment group in Daechan (Fig. 8A). The highest increment was only by 15% for 100 mM concentration of KNO3 where seeds were primed for 3 hours. The 100 mM and 50 mM of KNO3 significantly increased the SB in Pungsannamul by 32% and 34% in both cases (Fig. 8B).
Gene expression analysis
For the gene expression analysis, Daechan seeds primed for 3 hours with 50 mM of KNO3 and Pungsannamul seeds primed for 4 hours with 100 mM of KNO3 were selected as treatment. The expression level of four GA and ABA related genes has been illustrated in Fig. 9 in comparison with the control. GA-related, gene expression of GmGA2ox1 increased by 17% in Pungsannamul (Fig. 9A) and by 21% in Daechan (Fig. 9C) in the primed seeds, although no significant difference (p < 0.05) was observed in both cultivars. However, a significant increase (p < 0.05) in gene expression of GmGA3ox3 was observed in both cultivars (Fig. 9B and 9D). In Pungsannamul, the expression level of the primed seeds increased by more than threefold compared with the control (Fig. 9B), and in Daechan, the expression level increased by 30% in the primed seeds (Fig. 9D). The ABA-related genes GmABA2 (Fig. 9E and 9G) and GmNCED5 (Fig. 9F and 9H) did not show significant differences (p < 0.05) in the primed seeds in both cultivars. However, the expression level of GmNCED5 decreased by 15% in Pungsannamul (Fig. 9F) and by 25% in Daechan (Fig. 9H) in the primed seeds compared with the control, respectively.
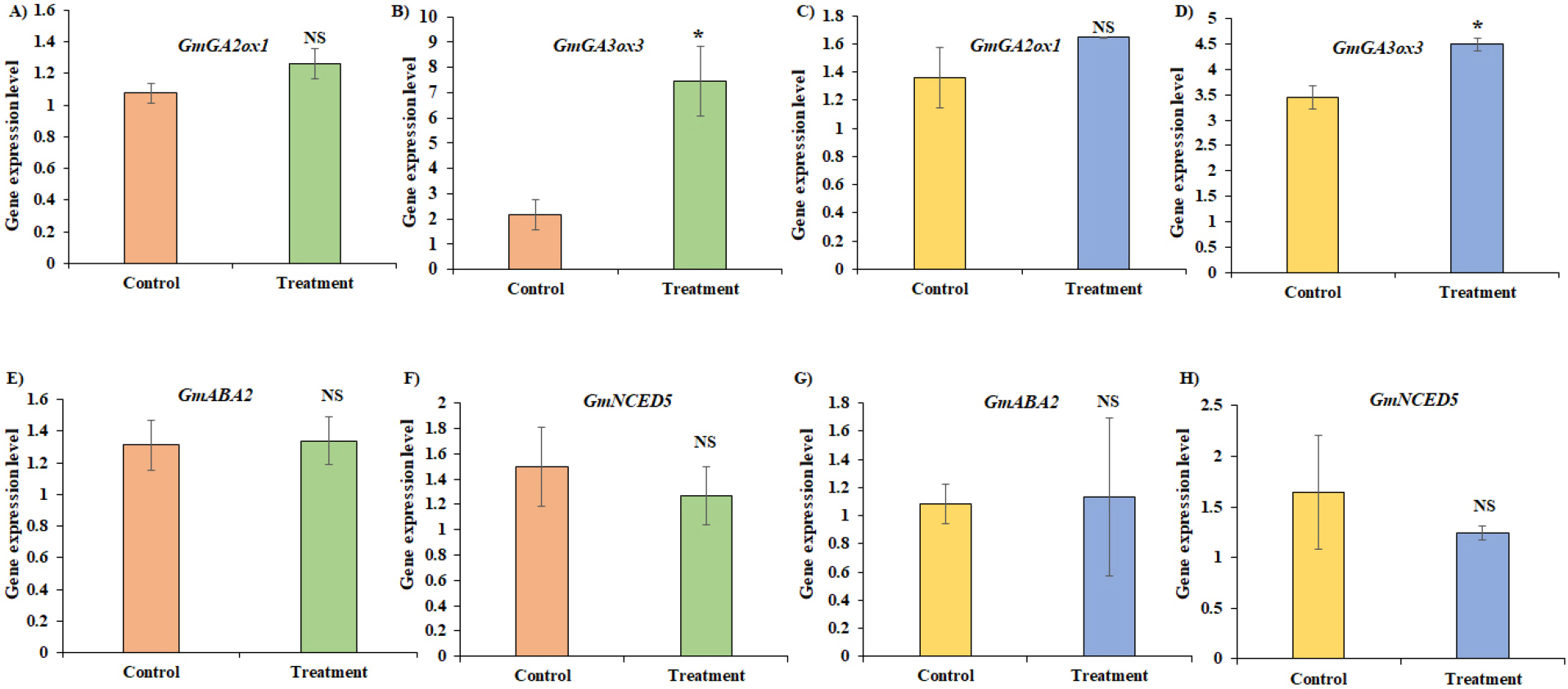
Fig. 9.
Gene expression level of GA and ABA-related genes in primed seeds and controlled seeds. Gene expression levels of (A) GmGA2ox1 in Pungsannamul, (B) GmGA3ox3 in Pungsannamul, (C) GmGA2ox1 in Daechan, (D) GmGA3ox3 in Daechan, (E) GmABA2 in Pungsannamul, (F) GmNCED5 in Pungsannamul, (G) GmABA2 in Daechan, and (H) GmNCED5 in Daechan. NS – Non-significant at p <0.05 and * - significant at p <0.05.
Discussion
Seed priming stimulates the pre-germination process through the induction of cell division and the release of hydrolytic enzymes, which enhances the seed germination and seedling establishment process (Hernández et al., 2021). The beneficial effect of priming at the physiological level starts due to the metabolic alterations within the seed due to the absorption of water (Bray, 2017). Due to this, a pre-germinative metabolism process occurs, which includes primary cellular activities like a synthesis of nucleic acids and proteins, the buildup of sterols and phospholipids, and activation of deoxyribonucleic acid (DNA) repair and antioxidant activity (Paparella et al., 2015). Seed priming focuses solely on the imbibition and activation phases of seed germination thus, when primed seeds are sown, these initial phases are shortened, resulting in accelerated germination (Khan et al., 2009; Rajjou et al., 2012). We observed that nearly every concentration of KNO3 resulted in improved germination and morphological parameters of the seedlings compared to the control group of seeds. The GP, GRI, VI, RL, PL, and SB were increased on the treated seeds. For Daechan, the combination of 100 mM concentration for 3 hours proved to be the most effective, although some parameters showed comparable results with seed priming at 50 mM and 100 mM concentrations for 4 hours. In Pungsannamul, optimal outcomes were observed with concentrations of 50 mM and 100 mM when priming durations of 3 hours and 4 hours were employed, respectively. KNO3 priming has been proven to positively impact the growth and development of crops under normal as well as stressed conditions. A significant increase in germination parameters, GP, RL, PL, and SB, was observed in two different cultivars of soybean when the seeds were primed with 6gm per liter of KNO3 under salinity stress (Ahmadvand et al., 2012). A similar result of an increase in GP (87%) was observed in soybean, along with an increase in yield attributes like biological yield, seeds per pod, and harvest index when the seeds were treated with 0.5% KNO3 for 12 hours (Agawane and Parhe, 2015). Maize seeds primed with KNO3 had better seedling establishment under lead contamination (Nawaz et al., 2017), and an increase in GP, MGT, and physiological attributes was observed in tomato seeds primed with 0.75% KNO3 (Moaaz et al., 2020). Similarly increase in yield attributes like an increase in plant height, fresh and dry weight, along with enhanced physiological parameters like chlorophyll content, photosynthesis, and stomatal conductance was observed in cucumber seeds primed with 5% KNO3 (Anwar et al., 2020) and increased GP, seedling length, SB, speed of emergence and VI was observed in the seeds of radish when primed with 2% KNO3 (Rohiwala et al., 2020). Similar results of increase in germination parameters were noted in pigeon pea at 0.3% KNO3 priming (Tiwari et al., 2014), in sesamum at 15 mM of KNO3 (Kumar et al., 2016), in wheat at 0.1% of KNO3 (Esatu et al., 2022), and in vetch plant at 0.155 M of KNO3 (Kalsa and Abebie, 2012).
GA, a phytohormone, plays a crucial role in inducing seed germination and promoting the development of the hypocotyl and stem in many plants (Peng and Harberd, 2002) whereas ABA has an antagonistic role to GA in the plant development process (Liu and Hou, 2018). The genes GA2ox and GA3ox control the synthesis and regulation of GA, which is a major plant growth regulator and plays a vital role in plant growth and development (Shah et al., 2023; Teshome and Kebede, 2021). The increase in seedling growth parameters in this study might be due to this increase in the GA contents in the primed seeds. Moreover, the expression of the ABA-related gene GmNCED5 was found to be reduced in the primed seeds. The enzyme associated with this gene; 9-cis-epoxycarotenoid dioxygenase (NCED), plays a crucial role in ABA synthesis, producing ABA in the embryo and endosperm, thereby inducing dormancy in the seeds (Frey et al., 2012; Zhang et al., 2008). In our study, a significant increase (p < 0.05) in GA-related gene GmGA3ox3 in primed seeds was observed with its expression level increased by three folds in Pungsannamul and by 30% in Daechan. The level expression level of GmNCED5 decreased by 15% in Pungsannamul and by 25% in Daechan in the primed seeds compared with the control, respectively. A similar result of an increase in expression level for GmGA3ox3 and GmGA2ox1 was found in our recent study in the seeds when they were primed with chitosan-GSNO nanoparticles and silicon (Steven et al., 2024). This study also had a decreased level of ABA-related genes; GmNCED5, in the primed seeds compared with the control. The priming agent used in this study; KNO3, serves as a nitrogen source compound, osmopriming with such nitrogenous substances initiates hormonal equilibrium adjustments, specifically involving ABA and GA, and is believed to activate aquaporins and boosts amylase activity, essential processes for seed germination and embryo development (Hernández et al., 2021; Shaheen et al., 2016). Another mechanism for enhancing the germination is due to the role of nitric oxide (NO) synthesis present in the nitrate solution, where NO breaks the dormancy by interacting with phytochrome signaling pathways and influencing ethylene biosynthesis, while also interacting with reactive oxygen species (Hernández et al., 2021; Šírová et al., 2011). The nitrate group of KNO3 also acts as a signaling molecule which enhances the seed germination (Zavariyan et al., 2015). Thus, in this study, it was found that the seeds primed with KNO3 have significant positive effects on the seedling growth parameters in soybeans. The priming of the seeds not only increased these parameters but also influenced the GA and ABA-related gene expression. Further research is recommended to examine the physiological responses of the primed seeds, such as antioxidant and hormonal activities, under varying priming concentrations and durations.