Introduction
Materials and Methods
Preparation of samples and extracts
Materials
Polyphenol and tannin content
Flavonoid content
DPPH free radical scavenging capacity
ABTS free radical scavenging capacity
Cell culture and inflammation induction
Cell viability
Nitric oxide production
mRNA expression of inflammatory cytokines
Enzyme-linked immunosorbent assay
Statistical analysis
Results
Quantification of bioactive compounds in CME
Antioxidant activity of CME
Effects of CME on cell viability and LPS-induced NO production
Effects of CME on iNOS and COX-2 expression
Effects of CME on inflammatory cytokine expression
Discussion and Conclusion
Introduction
The human body requires oxygen to generate energy essential for sustaining vital activities. Approximately 2-3% of the oxygen inhaled through the respiratory process is converted into free radicals, or reactive oxygen species (ROS), which perform various bodily functions depending on their concentration (Papa and Skulachev, 1997). ROS are crucial cell signaling molecules, facilitating redox signaling vital for cellular communication and homeostasis. However, excessive ROS production causes oxidative stress, damaging proteins, DNA, and lipids, ultimately causing cell cycle arrest, apoptosis, or necrosis (Lee et al., 2014). Oxidative stress triggers the expression of growth factors, cell cycle regulators, inflammatory cytokines, and inflammatory molecules within cells, such as nitric oxide (NO). NO is a neurotransmitter in normal physiological states, regulating vascular tone and maintaining ionic balance within inner ear lymphatics (Fessenden and Schacht, 1998). In pathological states, however, excessive NO production can result in tissue and neuronal damage, promoting cell death (Hippeli and Elstner, 1999).
The inflammatory response is triggered by invading harmful external substances, such as pathogens, and macrophages are mainly involved. Activated macrophages secrete NO and various inflammatory cytokines to eliminate foreign agents and subsequently initiate tissue repair (Medzhitov, 2008). Lipopolysaccharide (LPS), a major component of the outer membrane of gram-negative bacteria, binds to toll-like receptor 4 (TLR-4) on macrophage surfaces and functions as an endotoxin to induce inflammation (Barton and Medzhitov, 2003). LPS stimulation leads to the expression of enzymes such as inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2), which promote the production of NO and prostaglandin E2 (PGE2), respectively. Additionally, LPS stimulates the production of proinflammatory cytokines, including interleukin-6 (IL-6), interleukin-1β (IL-1β), and tumor necrosis factor-α (TNF-α) (McDaniel et al., 1996). IL-6 induces acute-phase inflammation, causing symptoms such as fever and anemia, and facilitates B cell differentiation (Berek et al., 1991). IL-1β mediates the host inflammatory response to infection and other stimuli (Freidin et al., 1992). TNF-α is a crucial mediator of acute inflammatory responses to pathogens, particularly gram-negative bacteria, and is implicated in systemic complications of severe infections (Andrews et al., 1990).
Phytochemicals are plant-synthesized chemicals that protect against sunlight, rain, wind, UV rays, pests, and microorganisms. Phytochemicals reduce oxidative stress in the body, prevent damage to intracellular molecules, and reduce the risk of chronic diseases such as diabetes, cardiovascular disease, and cancer (Kim, 2010). It is also considered a beneficial nutrient that promotes health, exhibiting anti-inflammatory, lipolytic, and immune-boosting effects. Phenolic phytochemicals, such as flavonoids, polyphenols, and phenolic acids, exhibit potent bactericidal and antioxidant effects and have been reported to help prevent cancer, cardiovascular diseases, and neurological degeneration (King and Young, 1999).
Centaurea cyanus belongs to the Asteraceae family and is proportionally distributed worldwide. The blue flowers of Centaurea cyanus have historically been used as natural dyes and decorative elements. Additionally, water-soluble extracts of the flowers have been utilized in traditional European medicine for treating ocular, neurological, dermatological, and gastrointestinal conditions, as well as tonics. Centaurea cyanus petals inhibit lipid oxidation and exhibit antihypertensive and antihemolytic activities (Escher et al., 2018). Using high-performance liquid chromatography (HPLC), Lockowandt et al. (2019) identified that Centaurea cyanus contains bioactive compounds such as α-tocopherol, citric acid, and succinic acid. α-Tocopherol, a primary form of vitamin E, has antioxidant and antithrombotic properties. Citric acid also has potent ROS-scavenging effects and is commonly found in citrus fruits. Succinic acid exerts antioxidant effects and is also involved in regulating IL-1β, an inflammatory cytokine (Tannahill et al., 2013).
Despite the potential of Centaurea cyanus as a functional material with antioxidant and anti-inflammatory properties, studies on its effects remain limited. Therefore, this study aimed to demonstrate the antioxidant activities of Centaurea cyanus methanolic extract (CME) and its anti-inflammatory effects in LPS-induced mouse macrophages (RAW 264.7 cells), evaluating its potential as a functional biomaterial.
Materials and Methods
Preparation of samples and extracts
The aerial parts of Centaurea cyanus were collected from Wolsong-ri, Jijeong-myeon, Wonju-si, Gangwon-do, Korea, and identified by Dr. Keun-Jeong Jang, a plant taxonomist at Kangwon National University. The plant material was transported at 4°C to the Natural Fragrance Laboratory at the College of Agriculture and Life Sciences, Kangwon National University, shade-dried at room temperature, and then pulverized using a plant grinder (NANIL, Korea). The CME was prepared by modifying the method described by Lim et al. (2007). A 1 kg pulverized dry sample was placed in a 5 L Erlenmeyer flask containing 2 L of methanol and allowed to soak for 24 h. The methanol extract was filtered to remove the residue and then concentrated using a rotary evaporator (EYELA NE-1101), followed by adding 50 mL of distilled water. The resulting solution was thoroughly dissolved in distilled water and freeze-dried using a lyophilizer (ILSHIN Lab Co. Ltd.). The CME yield was 7.34%.
Materials
Gallic acid, catechin, dimethyl sulfoxide (DMSO), lipopolysaccharide (LPS), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), and sodium nitrite were purchased from Sigma-Aldrich Co. Ltd. (St. Louis, MO, USA), Griess reagent was purchased from Promega (Madison, WI, USA), Dulbecco's modified Eagle's medium (DMEM), penicillin-streptomycin (P/S), and pure water were purchased from Welgene (Gyeongsan, Korea), Fetal bovine serum (FBS) was purchased from Atlas Biologicals (Fort Collins, CO, USA). RNA iso Plus was purchased from Takara Bio Inc. (Kusatsu, Japan). iNOS, COX-2, IL-6, IL-1β, TNF-α, and β-actin primers were purchased from Integrated DNA Technologies (Coralville, LA, USA). TransScript® All-in-One First-Strand cDNA Synthesis SuperMix for PCR was purchased from TransGen Biotech Co. (Beijing, China). Power SYBR® Green PCR Master Mix was purchased from Applied Biosystem Products, Thermo Fisher Scientific (Rockford, IL, USA), and ELISA kits were purchased from Invitrogen (Carlsbad, CA, USA). All other reagents were of commercially available quality.
Polyphenol and tannin content
The polyphenol and tannin content in CME was quantified using the Folin-Ciocalteu colorimetric method (Singleton et al., 1999). Polyphenols in CME react with the Folin-Ciocalteu reagent, producing a blue color. Therefore, 20 µL of 1,000 µg/mL CME was mixed with 100 µL of Folin reagent and incubated at room temperature for 3 min. Then, 80 µL of 80% sodium carbonate (Na2CO3) was added, and the mixture was protected from light and allowed to react at room temperature for 1 h. Absorbance was measured at 765 nm using a microplate reader (Molecular Devices, Sunnyvale, CA, USA). Total tannin content was determined by subtracting the non-tannin polyphenol content from the total polyphenol content, expressed as mg gallic acid equivalent (GAE)/g.
Flavonoid content
The flavonoid content in CME was measured using the aluminum chloride (AlCl3) spectrophotometric method (Uddin et al., 2015). Flavonoids react with AlCl3 to produce a yellow color. For the assay, 25 µL of 1,000 µg/mL CME was combined with 10 µL of sodium nitrite (NaNO2) and 100 µL of distilled water, then incubated for 5 min at room temperature. Subsequently, 15 µL of AlCl3 and 50 µL each of sodium hydroxide (NaOH) and distilled water were added. Absorbance was measured at 510 nm, and total flavonoid content was expressed as mg catechin equivalent (CE)/g.
DPPH free radical scavenging capacity
The DPPH radical scavenging activity of CME was assessed according to the method described by Eldeen et al. (2011). DPPH, containing stable free radicals, reacts with antioxidants and changes color from purple to yellow. Absorbance was measured to calculate the scavenging effect (%) using the following formula:
Scavenging effect (%) =
In a 96-well plate, 100 µL of cornflower extract was mixed with 100 µL of 0.2 mM DPPH solution and incubated for 30 min. The absorbance was measured at 517 nm using a microplate reader, and ascorbic acid was used as a control.
ABTS free radical scavenging capacity
The free radical scavenging capacity of the ABTS assay was measured using the method described by Adewusi et al. (2011). ABTS is characterized by a blue-green to colorless color change as cationic radicals are scavenged after reaction with antioxidants. The absorbance was measured, and the scavenging effect (%) was calculated using the following equation.
Scavenging effect (%) =
The stock solution of 7 mM ABTS with 2.4 mM potassium persulfate (K2S2O8) was refrigerated for at least 12 h in the dark and diluted with 70% ethanol to an absorbance value of 0.7 ± 0.02 at 734 nm before use. The ABTS and cornflower extract were reacted in the cow for 30 min and the absorbance was measured at 734 nm using a microplate reader, and ascorbic acid was used as a control.
Cell culture and inflammation induction
RAW 264.7 mouse macrophages were obtained from the Korea Cell Line Bank (Korea). They were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 100 units/mL penicillin-streptomycin and 10% FBS. Cells were maintained at 37°C in a 5% CO2 incubator and passaged every 2 days.
Cell viability
RAW 264.7 cells were seeded at a density of 2 × 105 cells/well in a 96-well plate and incubated at 37°C in a 5% CO2 incubator for 24 h. After replacing the medium with serum-free medium and incubating for another 24 h, cells were treated with CME at 125, 250, 500, and 1,000 µg/mL concentrations in a 100 µL medium. After 24 h, 100 µL of MTT reagent (5 mg/mL) mixed with serum-free medium (1:9) was added to each well, and cells were incubated for an additional 4 h. The medium was then removed, and 100 µL of a 1:1 mixture of 99.5% DMSO and isopropanol was added to dissolve the cells. Absorbance was measured at 540 nm using a microplate reader.
Nitric oxide production
RAW 264.7 cells (2 × 106 cells/well) were seeded in a 6-well cell culture plate and incubated at 37°C with 5% CO2 for 24 h. The medium was replaced with a serum-free medium, and after an additional 24 h incubation, the cells were pretreated with CME at various concentrations. After 1 h, LPS (1 µg/mL) was added to all wells except the control group, followed by a 24 h incubation. The supernatant from each well was collected and mixed with Griess reagent at a 1:1 ratio, and absorbance was measured at 540 nm using a microplate reader. The concentration of NO in the culture medium was calculated using a standard curve prepared with NaNO2, and the IC50 (50% inhibitory concentration) was determined.
mRNA expression of inflammatory cytokines
RAW 264.7 cells were lysed by adding 1 mL of RNA iso Plus (Takara Bio Inc., Kusatsu, Japan) to each well, followed by 200 µL of chloroform. The mixture was centrifuged at 10,000 rpm for 30 min at 4°C, and 400 µL of the supernatant was transferred to a tube containing isopropanol at a 1:1 ratio. After a 10 min centrifugation at 12,000 rpm and 4°C, the supernatant was discarded, and the precipitate was washed with 75% ethanol, followed by a 5 min centrifugation at 10,000 rpm at 4°C. The pellet was dried completely and dissolved in 50 µL of pure water, then stored at 4°C until further use. The extracted mRNA was quantified using a microplate reader and synthesized into 20 µL cDNA using TransScript® All-in-One First-Strand cDNA Synthesis SuperMix for qPCR (TransGen Biotech Co., Beijing, China). The resulting cDNA was diluted in 180 µL of pure water and used.
Real-time polymerase chain reaction (RT-PCR) was performed by mixing 4 µL of cDNA with 4.4 µL of pure water, 10 µL of power SYBR® Green PCR Master mix, and 1.6 µL of primers (iNOS, COX-2, IL-6, IL-1β, TNF-α, or β-actin). RT-PCR conditions were set as follows: initial denaturation at 95°C for 10 min, followed by 40 cycles of denaturation at 95°C for 15 s, annealing at 57°C for 20 s, and extension at 72°C for 40 s. The final step included 95°C for 15 s, 60°C for 1 min, and 95°C for 15 s (Yang et al., 2012). The sequences for each primer are presented in Table 1.
Table 1.
Primer sequences for RT-PCR analysis
Enzyme-linked immunosorbent assay
IL-6, IL-1β, and TNF-α levels induced by LPS were measured using an ELISA kit in the culture medium. After 24 h of stimulation by LPS, the culture medium was collected and centrifuged at 10,000 rpm for 10 min at 4°C to remove particulates and aggregates. Assays were performed according to the manufacturer’s protocol provided with the ELISA kit.
Statistical analysis
All measurements were expressed as mean ± standard error of the mean, and statistical significance between control and experimental groups was determined by one-way analysis of variance (ANOVA) using GraphPad Prism (GraphPad Software Inc., La Jolla, CA, USA), followed by Student-Newman-Keuls test at P < 0.05.
Results
Quantification of bioactive compounds in CME
The content of bioactive compounds in CME was evaluated by measuring the total polyphenol, total tannin, and total flavonoid. The total polyphenol content in CME was approximately 33.76 mg GAE/g. The total tannin content, representing a major component of polyphenols, was approximately 26.84 mg GAE/g, whereas the total flavonoid content was approximately 45.28 mg CE/g (Table 2).
Table 2.
Physiological active substance content of CME
| Phytochemical tests | |
| Total polyphenol | 33.76 ± 1.27 mg GAE/g |
| Total tannin | 26.84 ± 0.16 mg GAE/g |
| Total flavonoid | 45.28 ± 17.03 mg CE/g |
Antioxidant activity of CME
The antioxidant activity of CME was assessed using DPPH and ABTS assays, with results expressed as the 50% radical scavenging concentration (SC50). The SC50 value of ascorbic acid, the reference substance, was approximately 15 µg/mL or lower, consistent with previous studies (Ullah et al., 2017). CME showed free radical scavenging activities with SC50 values of approximately 271.77 µg/mL for DPPH and 397.51 µg/mL for ABTS (Table 3).
Table 3.
Antioxidative effects of CME extract using DPPH and ABTS assay
| Radical scavenging activity | |
| DPPH assay | 271.77 ± 3.07 µg/mL |
| ABTS assay | 397.51 ± 24.71 µg/mL |
Effects of CME on cell viability and LPS-induced NO production
The cytotoxicity of CME on RAW 264.7 cells was evaluated using the MTT assay. Cells were considered cytotoxic if their viability was ≤ 70% compared to the control. CME at concentrations up to 1,000 µg/mL showed no cytotoxicity (Fig. 1A). Notably, cell viability was increased at concentrations above 500 µg/mL of CME, likely due to the inherent color of CME becoming darker with increasing concentration. Therefore, further investigations were conducted at concentrations below 1,000 µg/mL. A NO assay was performed to evaluate whether CME pretreatment suppressed LPS-induced NO production in RAW 264.7 cells. The results indicated that NO production increased in LPS-induced RAW 264.7 cells, whereas CME pretreatment significantly reduced NO production (IC50 = 283.6 µg/mL) (Fig. 1B).
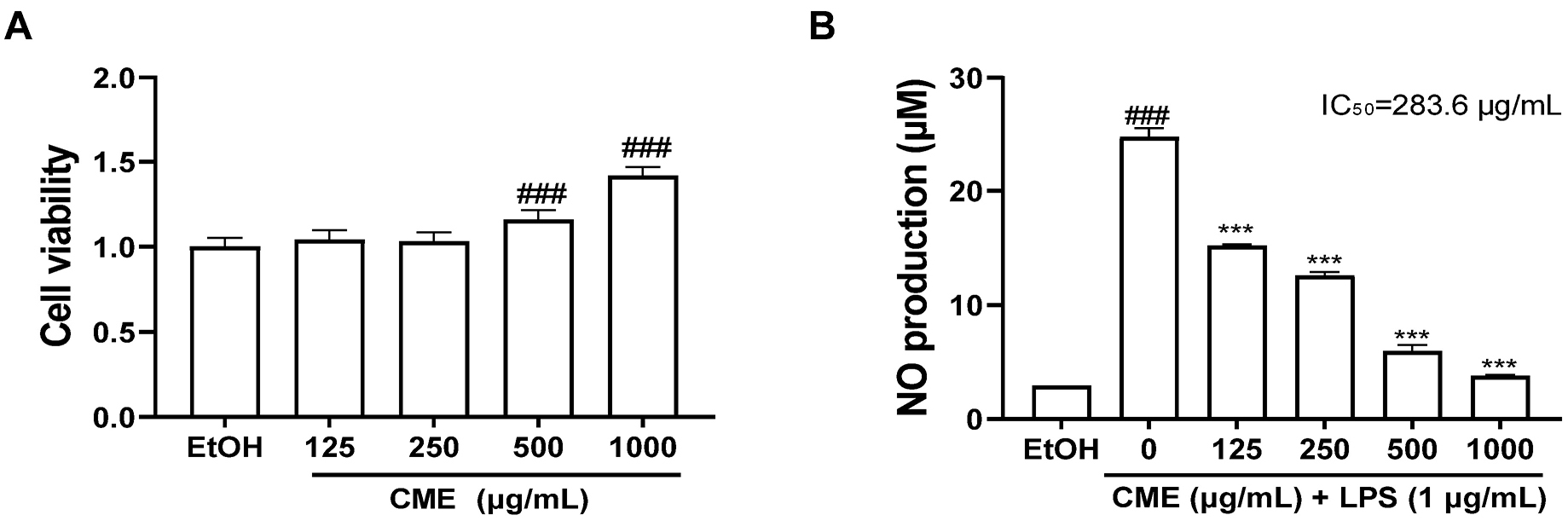
Fig. 1.
Effects of CME on cell viability and LPS-induced NO production in RAW 264.7 cells. (A) CME was treated for 24 h, and cytotoxicity was measured using MTT assay. (B) CME was treated for 1 h before LPS stimulation, and NO production was determined after LPS (1 µg/mL) was stimulated for 24 h. CME; Centaurea cyanus methanolic extract, EtOH; 70% ethanol. ###P < 0.001: significant compared to EtOH, ***P < 0.001: significant compared to LPS alone.
Effects of CME on iNOS and COX-2 expression
The effects of CME pretreatment on the iNOS and COX-2 expression, key enzymes in NO production, were assessed using RT-qPCR. The results demonstrated that LPS stimulation significantly increased the expression of iNOS and COX-2. In contrast, CME significantly decreased iNOS mRNA expression (IC50 = 189.3 µg/mL) and COX-2 mRNA expression (IC50 = 168.5 µg/mL) with increasing concentrations (Fig. 2). These results indicating that CME effectively reduces NO production by downregulating iNOS and COX-2 expression, highlighting its potential as an anti-inflammatory agent.
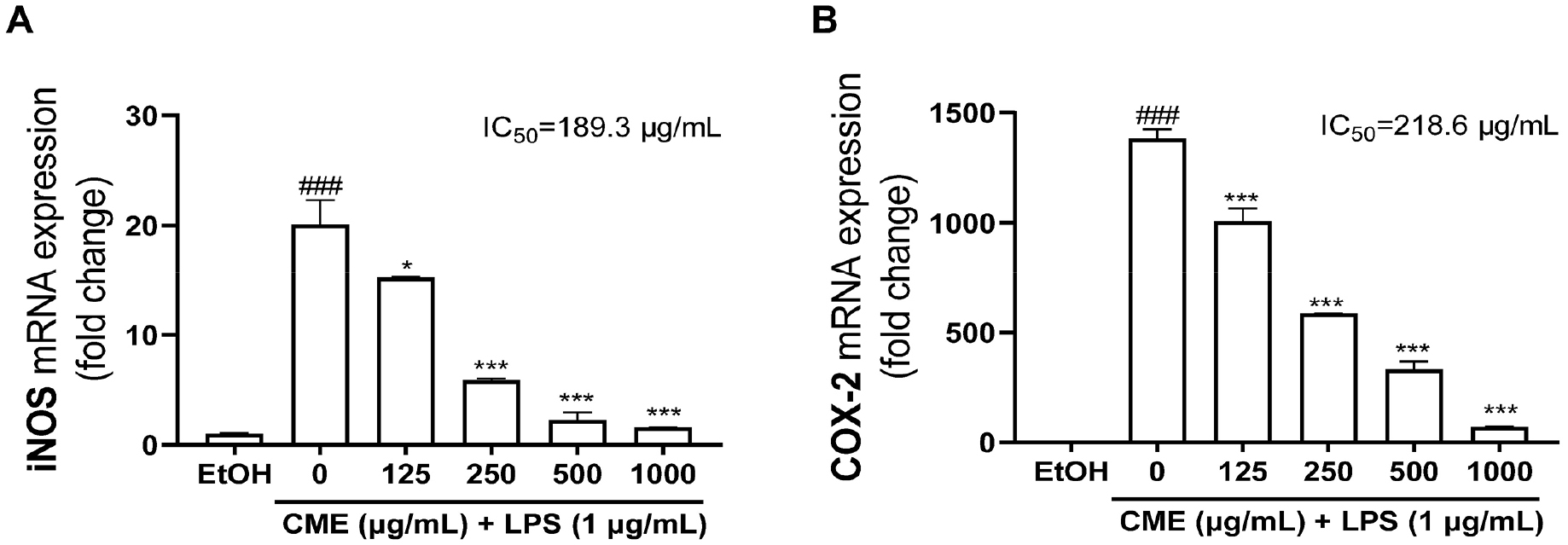
Fig. 2.
Effects of CME on iNOS and COX-2 mRNA expressions in LPS-stimulated RAW 264.7 cells. The mRNA expression levels of (A) iNOS and (B) COX-2 were determined after LPS (1 µg/mL) stimulation for 24 h, and the fold changes were presented compared with EtOH. CME; Centaurea cyanus methanolic extract, EtOH; 70% ethanol. ###P < 0.001: significant compared to EtOH, *P < 0.05; ***P < 0.001: significant compared to LPS alone.
Effects of CME on inflammatory cytokine expression
The effect of CME pretreatment in suppressing LPS-induced mRNA and protein expression of the proinflammatory cytokines, including IL-6, IL-1β, and TNF-α, was evaluated using RT-qPCR and ELISA. The mRNA expression of IL-6, IL-1β, and TNF-α were increased in LPS-stimulated RAW 264.7 cells, leading to an increase in their protein release into the culture medium (Fig. 3). CME pretreatment dose-dependently reduced IL-6 mRNA (IC50 = 158.3 µg/mL) and protein (IC50 = 306.6 µg/mL) levels (Figs. 3A, 3B). Similarly, CME suppressed IL-1β mRNA (IC50 = 468.1 µg/mL) and protein (IC50 = 152.6 µg/mL) expression (Figs. 3C, 3D). Notably, although a transient increase in IL-1β mRNA was observed at 125 µg/mL CME in LPS-induced RAW 264.7 cells (Fig. 3C), higher concentrations of CME led to a decrease in both IL-1β mRNA and protein expression. This likely reflects a localized effect within the experimental system rather than an actual increase in inflammatory response. Additionally, CME effectively reduced TNF-α mRNA (IC50 = 593.1 µg/mL) and protein (IC50 = 644.8 µg/mL) expression (Figs. 3E, 3F). These findings indicate that CME reduces the mRNA expression of IL-6, IL-1β, and TNF-α, thereby decreasing protein production and confirming its anti-inflammatory potential in LPS-induced inflammatory responses.
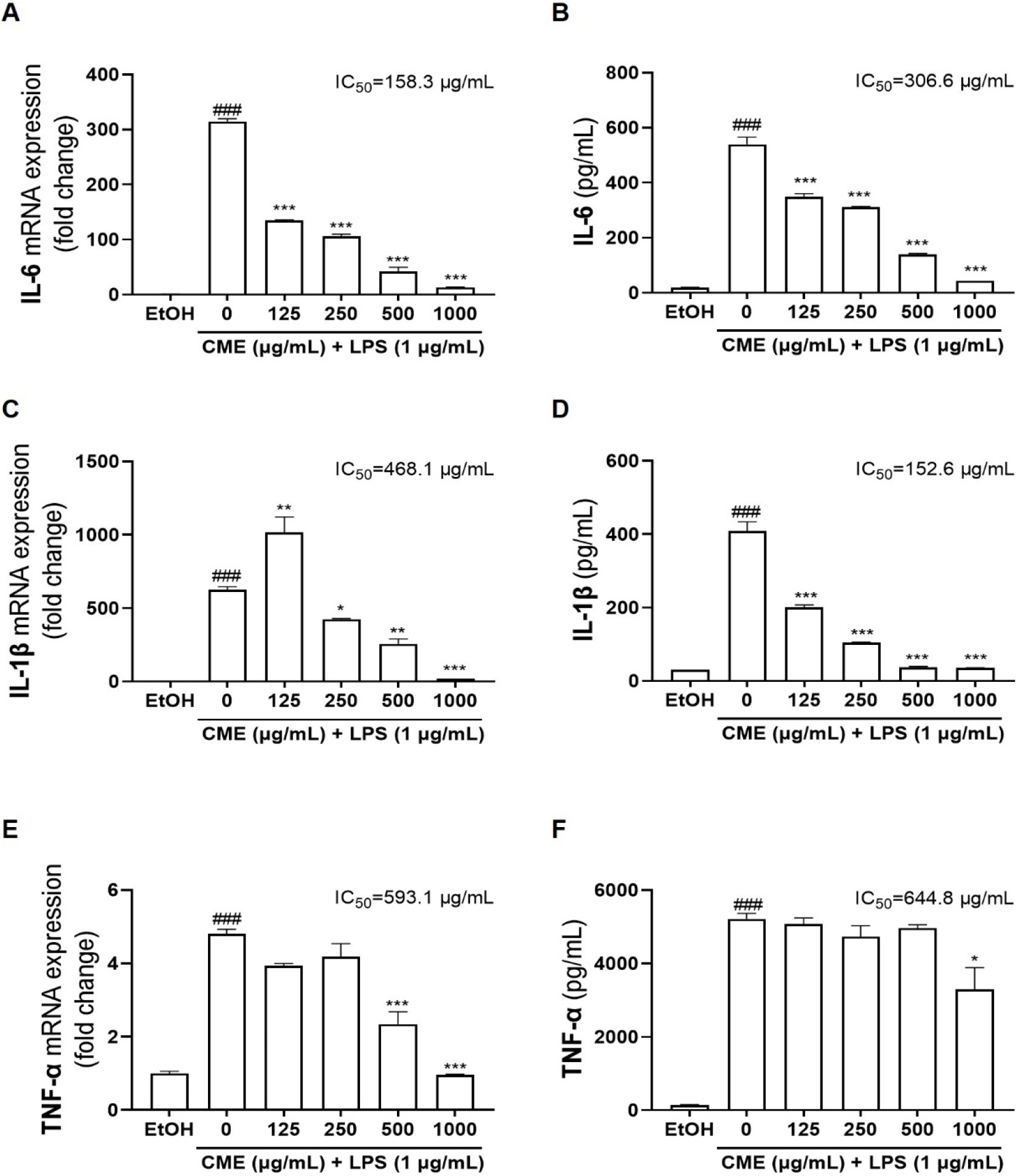
Fig. 3.
Effects of CME on inflammatory cytokines IL-6, IL-1β, and TNF-α expressions in LPS-stimulated RAW 264.7 cells. After stimulation with LPS (1 µg/mL) for 24 h, relative mRNA expression levels of (A) IL-6, (C) IL-1β, and (E) TNF-α were analyzed using RT-qPCR. Production of (B) IL-6, (D) IL-1β, and (F) TNF-α proteins in media were determined using ELISA. CME; Centaurea cyanus methanolic extract. EtOH; 70% ethanol. ###P < 0.001: significant compared to EtOH, *P < 0.05; **P < 0.01; ***P < 0.001: significant compared to LPS alone.
Discussion and Conclusion
This study aimed to investigate the potential of CME as a functional material with antioxidant and anti-inflammatory properties. Polyphenolic compounds are extensively present in plants as secondary metabolites, and they function as reducing agents, radical scavengers, and chain-breaking antioxidants owing to their unique chemical structures. Considering these properties, recent studies have focused on the antioxidant activity of plant extracts and their potential as functional materials (Scalbert et al., 2005). CME showed significant levels of bioactive compounds, with a total polyphenol content of 33.76 ± 1.27 mg GAE/g, total tannin of 26.84 ± 0.16 mg GAE/g, and total flavonoid of 45.28 ± 17.03 mg CE/g. The SC50 values of CME for antioxidant activity were 0.271 ± 0.03 mg/mL in the DPPH assay and 0.397 ± 0.24 mg/mL in the ABTS assay, respectively. According to previous studies, the antioxidant activity of dried extracts can be classified based on their SC50 values as follows: excellent if SC50 is below 0.01 mg/mL, good if it is below 1 mg/mL, moderate if it is below 10 mg/mL, and low if it exceeds 10 mg/mL (Qusti et al., 2010). Based on this classification, the Centaurea methanol extract can be considered to have excellent antioxidant activity. The SC50 of methanolic extracts of C. kroumirensis and C. sicula, plants of the genus Centaurea, evaluated using the DPPH assay for their antioxidant activity were ≤ 0.94 ± 0.01 and 2.94 ± 0.02 mg/mL, respectively (Dhouibi et al., 2020). In addition, Centaurea species such as C. nigra, C. orientalis, and C. phrygia demonstrated strong antioxidant efficacy with high polyphenol and flavonoid contents (Salachna et al., 2021). Flavonoids in Centaurea, including apigenin, cyanidin, and quercetin derivatives, have antioxidant and anti-inflammatory properties with diverse pharmacological applications (Al-snafi, 2015; Ginwala et al., 2019; Meleleo et al., 2023). Among these, cyanidin 3-O-(6”-succinyl glucoside)-5-O-glucoside, a major compound in C. cyanus, exhibits antioxidant and antimicrobial activities (Lockowandt et al., 2019). Our findings support the antioxidant potential of CME and underscore its potential as a functional ingredient. Further compound isolation using methods such as HPLC could further validate our findings.
In addition to their antioxidant activity, phenolic compounds in natural compounds play an effective role as anti-inflammatory factors (Arulselvan et al., 2016). Therefore, to determine the anti-inflammatory activity of CME, RAW 264.7 cells were pretreated with the extract for 1 h, and inflammation was induced with LPS. CME effectively reduced NO production and downregulated the expression of iNOS, which induces NO production, following LPS stimulation. Additionally, it also effectively reduced COX-2 expression. COX-2 is induced by external stimuli and inflammatory mediators, playing a key role in producing PGE2, which causes pain and inflammation, and its regulation can alleviate both (Kulesza et al., 2023). Therefore, based on the results of this study, it is anticipated that similar to the regulation of iNOS expression and subsequent inhibition of NO production by CME, modulation of COX-2 expression may also lead to a reduction in PGE2 levels.
In addition, CME effectively reduced IL-6, IL-1β, and TNF-α mRNA expression in RAW 264.7 cells and the secretion of these proteins into the culture medium. Inflammatory cytokines are important in signaling between immune cells during the immune system to foreign antigens. Overexpression of these proinflammatory cytokines causes tissue damage, organ failure, fever, blood clotting, myocardial depression, hypotension, metabolic derangements, and hypoglycemia (Voss and Cotton, 2008).
Overall, CME exhibited potent antioxidant and anti-inflammatory properties by effectively scavenging ROS owing to its high polyphenol content and inhibiting the production of NO and proinflammatory cytokines. These results suggest that CME can be a natural functional antioxidant and anti-inflammatory agent, potentially protecting tissues from oxidative and inflammatory damage. However, further studies are needed to clarify its active constituents and mechanisms of action. Moreover, animal model studies assessing in vivo efficacy and potential toxicity can further substantiate the value of CME as a functional material.




