Introduction
Methodology
Plant materials
Cell culture
MTT assay
Morphological assay
qPCR analysis
Statistical analysis
Results and Discussion
Cytotoxicity in sorghum extract-treated cancer cells
Apoptosis of sorghum extract-treated cancer cells
Expression of apoptosis-related genes in cancer cells
Summary
Introduction
The risk of cancer and chronic diseases is increasing as the metabolic balance is disrupted due to environmental pollution caused by urbanization and industrialization, and factors such as excessive stress, drinking, and smoking (Jin et al., 2021). According to WHO statistics, in 2022, cancer was the cause of death in one in five people worldwide, with 20 million new cases and an estimated total of 9.7 million deaths attributed to cancer (Bray et al., 2024; Senapati et al., 2015). Currently, radiation and chemicals are used for cancer treatment, but they are known to cause various side effects such as acne rashes, hair changes, and various cardiovascular diseases (Ryu, 2023). Therefore, research on chemotherapy using natural plant materials with low risk of toxicity or side effects in the human body is necessary (Garcia-Oliveira et al., 2021).
Apoptosis is the most critical way to treat cancer, maintain tissue homeostasis, and regulate tissues (Chae et al., 2020). If an abnormality occurs in this process, cancer cells continue to grow, promoting tumor formation. Various morphological changes occur during the apoptosis of cancer cells. This process is induced in cells by DNA damage caused by internal or external stimuli. Subsequently, through various complex steps, the cell ultimately undergoes death due to the activation of some proteolytic enzymes called caspases (Lee et al., 2015). Apoptosis is induced through both the extrinsic and intrinsic pathways. The extrinsic pathway involves binding to death receptors such as Fas and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) receptors, leading to the formation of a death-inducing signaling complex (DISC). Subsequently, Caspase-8 is activated through various signaling cascades (Lee and Kwon, 2020). The intrinsic pathway is controlled by the expression of Bcl-2 family proteins and anti-apoptotic proteins, which releases Cytochrome C into the cytoplasm and activates Caspase-9 (Shin et al., 2016). Through these pathways, caspases are activated into Caspase-3, Caspase-7, and Caspase-9. Activated caspases degrade substrate proteins such as poly ADP-ribose polymerase, an enzyme involved in DNA repair, thereby inhibiting normal cellular functions and inducing apoptosis (Kim and Kim, 2015; Pfeffer and Singh, 2018). Therefore, it is crucial to examine the functional regulation of Bcl-2 family expression, which is connected to mitochondrial pathways within the apoptosis-related extrinsic and intrinsic pathways (Kim et al., 2016).
Sorghum bicolor L. is the fifth-most widely produced crop globally, and is primarily consumed in Asia and Africa (Hamadou et al., 2023). Sorghum is rich in key compounds such as polyphenols and flavonoids, and has traditionally been used in folk medicine to aid digestion, maintain body temperature, and fortify the stomach (Ham et al., 2024; Seo et al., 2023). Sorghum seed extract has exhibited effective anticancer activity in the liver, colon, and lung cancer cells, with mechanisms that inhibit the cancer cell cycle and suppress cancer cell growth (Chen et al., 2021; Owumi et al., 2022). Although research on the anticancer effects of sorghum has been steadily conducted recently, no study has compared the cancer cell-death activity of different sorghum varieties.
Therefore, the aim of this study was to evaluate the anticancer potential of genetically diverse Sorghum bicolor seed extracts by assessing their effects on cell morphology, viability, and apoptosis-related gene expression (Bax, Bcl-2, Caspase-3, and Caspase-9) in AGS and HCT116 human cancer cells. This approach aimed to identify promising sorghum lines for future development of plant-derived anticancer agents.
Methodology
Plant materials
The 13 sorghum accessions were selected to represent genetic and geographic diversity, including lines from Australia, the Former Soviet Union, the United States, Sudan, and Guadeloupe. These accessions were previously reported to exhibit variation in phenolic content and antioxidant potential (Seo et al., 2023), which justified their selection for comparative analysis. The sorghum seeds used in the analysis were K159041 (New South Wales, Australia), K159042 (New South Wales, Australia), K159078 (Former Soviet Union), K159081 (Former Soviet Union), K159088 (Nebraska, USA), K159089 (Nebraska, USA), K159093 (Texas, USA), K159097 (Texas, USA), K159100 (Texas, USA), K159096 (Virginia, USA), K159048 (Kordofan, Sudan), and K159077 (Basse-Terre, Guadeloupe), which were distributed and used (Fig. 1). After crushing 5 g of sorghum seeds (HG-7113; Haeger, Barcelona, Spain), the seeds were mixed with 100% methanol for 48 h for extraction, and subsequently, the extract was filtered (Whatman No. 42; Whatman LTD., Maidstone, UK). The filtered solution was concentrated to the final concentration of 10,000 µg/mL using the rotary vacuum concentrate (N-1110; Eyela, Tokyo, JAPAN) and used for the experiment.
Cell culture
The gastric cancer cell line AGS and colon cancer cell line HCT116 were provided by The Korean Cell Line Bank (Seoul, Republic of Korea). RPMI-1640 Medium (Hyclone, UT, USA) was supplemented with 10% fetal bovine serum (FBS; WelGENE, Gyeongsan, Korea) and 1% penicillin/streptomycin (Thermo Fisher Scientific, Inc., Waltham, MA, USA) to support the growth of AGS and HCT116 cells. At 37°C and 5% CO2, AGS and HCT116 cells were cultivated in an incubator.
MTT assay
The apoptotic effect of sorghum extracts on AGS and HCT116 cells was assessed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) assay. Briefly, 96-well plates were seeded at a density of 1 × 105 cells/well and cultured for 24 h. Each sorghum extract was diluted to a desired amount in RPMI-1640 medium supplemented with 1% FBS and administered to cancer cells, followed by culturing for 24 h. After incubation for 4 h with MTT, absorbance was measured at 519 nm using a UV spectrophotometer (Thermo Fisher Scientific, Inc.).
Morphological assay
AGS and HCT116 cells were plated at a density of 1 × 106 cells/well in a 6-well plate and cultured until cell confluence surpassed 80%. Following the removal of culture medium, the cells were rinsed with 1× PBS and subsequently treated with sorghum extract diluted to 100 µg/mL in RPMI-1640 medium. After a 24-h incubation, morphological alterations in the cells were assessed using iSolutionLite software (IMT i-solution Inc., Quebec, Canada).
qPCR analysis
qPCR was performed to evaluate the ability of the sorghum extract to inhibit the proliferation of AGS and HCT116 cells. AGS and HCT116 cells were seeded at 1 × 106 cells/well density in a 6-well plate and cultured in an incubator for 24 h. The cells were washed with 1× PBS and treated with 100 µg/mL of diluted sorghum extract for 24 h. Total RNA was extracted using a Total RNA Extraction Kit (iNtRON Biotechnology, Seongnam, Korea). cDNA synthesis was performed using PrimeScript™RT Master Mix (Perfect Real Time; Takara Korea Biomedical Inc., Seoul, Korea). A 25-µL reaction mixture was prepared using TB Green® Premix Ex Taq™ (Tli RNaseH Plus; Takara Korea Biomedical Inc.), and the analysis was performed using CronoSTAR™96 Real time-PCR (Takara Korea Biomedical Inc.). PCR amplification conditions were initial denaturation at 95°C for 30 s, followed by 40 cycles of 95°C for 5 s and 60°C for 30 s, and after the final reaction, the analysis was performed under the conditions of 95°C for 1 min, 60°C for 15 s, and 98°C for 5 s. Nucleotide sequences of the primers used are listed in Table 1. All primers used for qPCR analysis were newly designed using Primer-BLAST (NCBI), and amplification efficiencies were confirmed to fall within the acceptable range of 90-110%.
Table 1
Nucleotide sequences of primers used in qPCR analysis
Statistical analysis
Statistical analysis was based on the results of three independent experiments, and the outcomes are expressed as the mean ± standard deviation. The data were analyzed using IBM SPSS Statistics 26 software. Duncan’s multiple range test was applied to confirm statistical significance through analysis of variance, and the statistical significance criterion was set at p < 0.05.
Results and Discussion
Cytotoxicity in sorghum extract-treated cancer cells
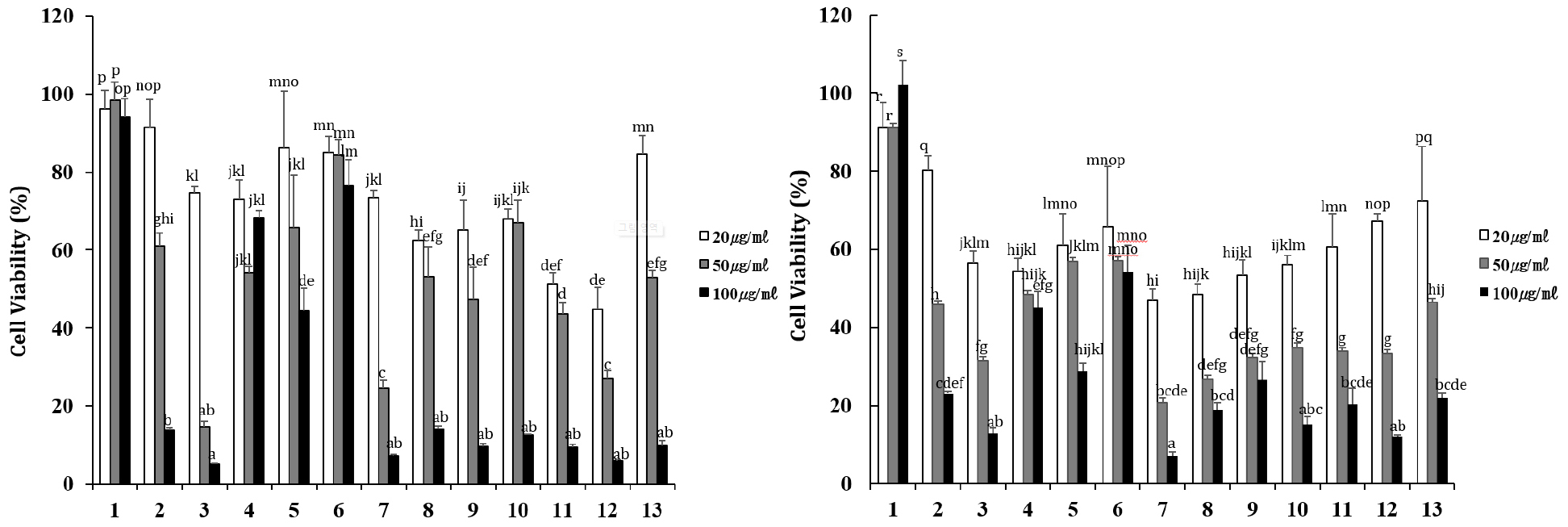
The MTT assay measures cytotoxicity by measuring the degree to which MTT is reduced by the enzymatic action of living cells to form formazan crystals (Benov, 2021). The effects of sorghum seed extract on cancer cell viability were evaluated in AGS and HCT116 cells (Fig. 2). All extract treatment groups showed concentration-dependent inhibition of cancer cell growth, and at a concentration of 100 µg/mL, AGS cells experienced cell inhibition of 5.2-76.7%, and HCT116 cells experienced cell inhibition of 7.2-54.2%. In AGS cells, the K159042 sorghum line showed the highest inhibition (5.2%), followed by the K159041, K159081, K159088, K159089, K159093, K159096, K159097, and K159100 lines (Fig. 2A). In HCT116 cells, the K159081 line showed the highest cell inhibition ability at 7.2%, followed by K159097 and K159042 (Fig. 2B). In a study comparing ovarian cancer cell inhibitory activity of ethanol and methanol extracts of sorghum, the ethanol extract had 76.3% activity and the methanol extract had 44.6% activity, indicating that the ethanol extract had greater inhibitory activity (Dia et al., 2016). Therefore, sorghum extracts have various effects on cancer cells, and additional research on cancer cell death is required.
Apoptosis of sorghum extract-treated cancer cells
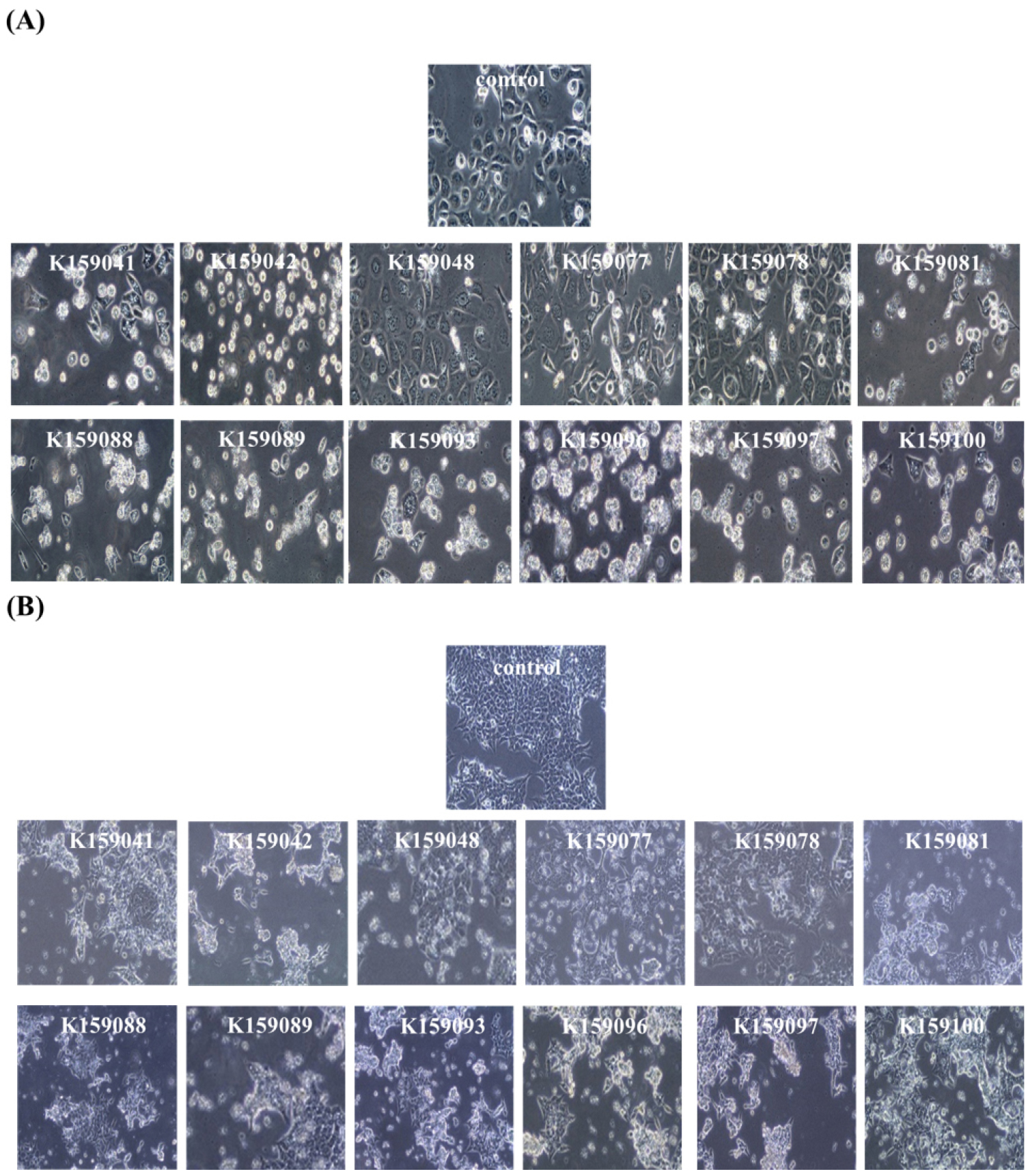
To determine the effect of sorghum seed extract on the morphology of cancer cells, each cancer cell was treated with 100 µg/mL of sorghum extract for 24 h, and then morphological analysis was performed. In the control group, AGS cells were attached in a stable form and proliferated normally. However, in most groups treated with sorghum extract, cancer cell proliferation was inhibited. However, when treated with extracts from the K159048, K159077, and K159078 sorghum lines, patterns similar to those in the control group were observed (Fig. 3A). A similar tendency was observed in HCT116 cells as in AGS cells, and it was confirmed that these three extracts had relatively low inhibitory activity against AGS and HCT116 cells compared to the extracts of other lines (Fig. 3B). Microscopic observations revealed the formation of black blisters, which is an indicative of cell membrane collapse in cancer cells (Baek, 2023). Cancer cell death, cell membrane damage, and cell disruption were observed in the remaining treatment groups, except for K159048, K159077, and K159078 sorghum lines.
Expression of apoptosis-related genes in cancer cells
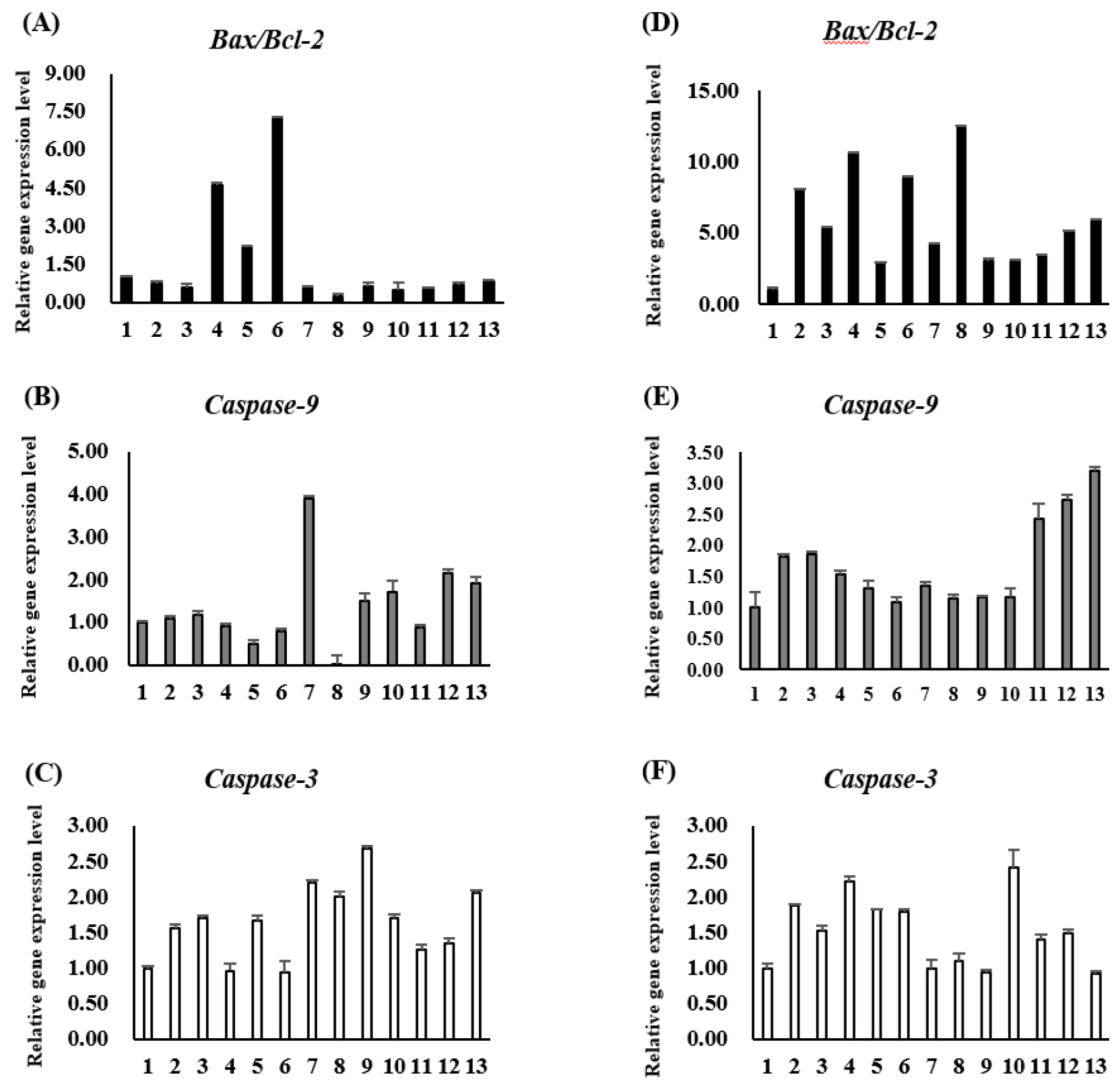
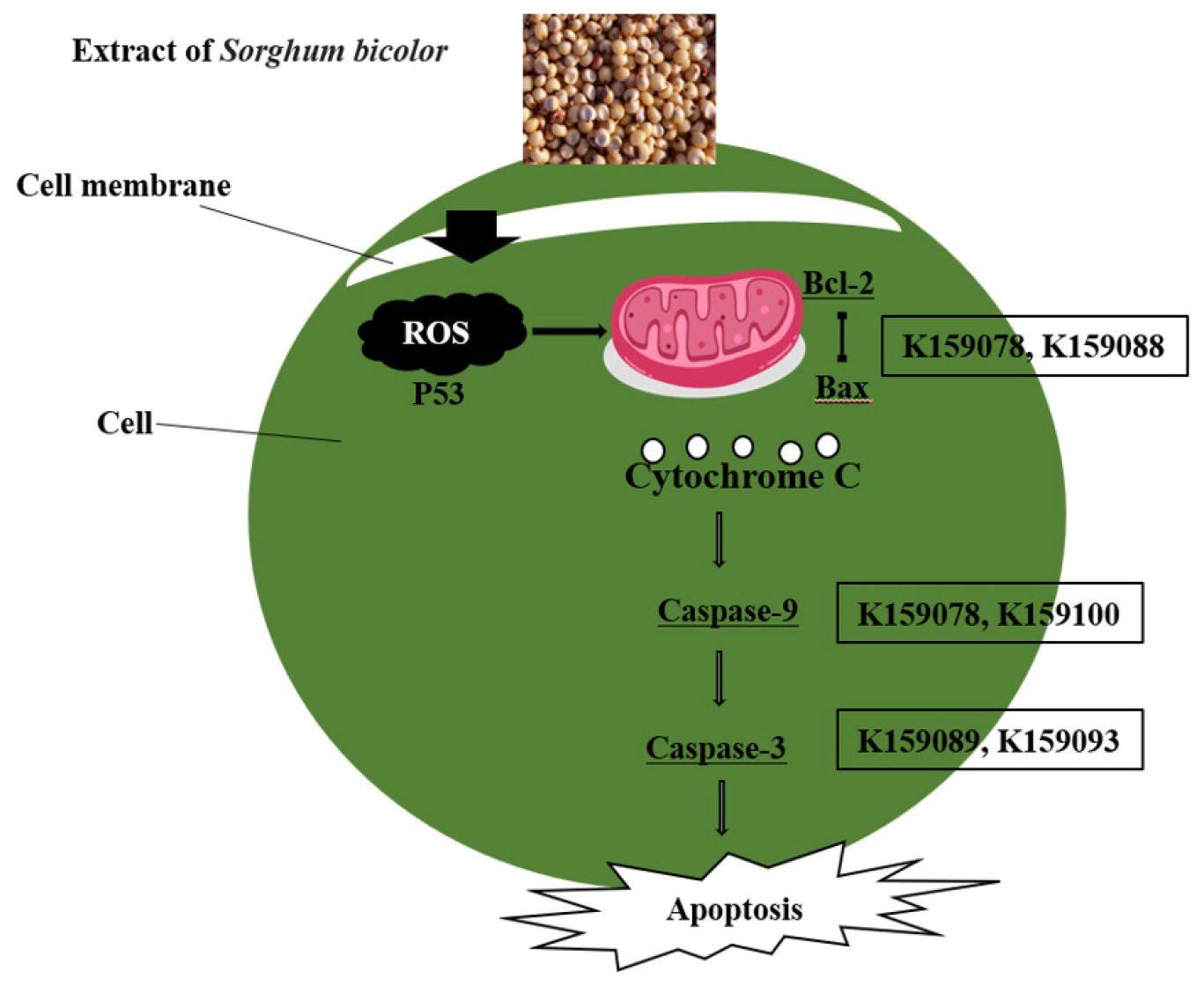
In this study, after treating AGS and HCT116 cells with sorghum extract, the mRNA expression levels of the apoptosis-related genes Bax, Bcl-2, Caspase-3, and Caspase-9 were verified. The extract with the highest Bax/Bcl-2 expression in AGS cells was K159078, followed by K159048 and K159077. In the case of Caspase-9, K159081 showed an expression level approximately 4-times higher than that in the control group. The expression of Caspase-3, a gene involved in the final stage of cell death, was higher in all treatment groups than in the control group; and in particular, K159089 showed the highest expression (Fig. 4A-C). In HCT116 cells, Bax/Bcl-2 expression was more than 12-times higher in K159088 than in the control group, and Caspase-9 was most expressed in K159100, followed by K159097 and K159096. In all treatment groups, Caspase-3 showed higher expression levels than that in the control group, similar to that observed in AGS cells; K159093 showed the highest expression level (Fig. 4D-F). The results of this experiment showed that cancer cell death and mRNA expression levels were not consistent across all treatment groups. In the apoptotic pathway, several proteins form complexes to perform their functions (D’Orsi et al., 2017; Sahoo et al., 2023) (Fig. 5). Polyphenolic and flavonoid compounds in sorghum, especially 3-deoxyanthocyanidins, have been reported to trigger mitochondrial apoptosis by modulating Bcl-2 family proteins and activating caspases. The observed upregulation of Bax and Caspase-3 may be attributed to such compounds, although further profiling is required. Therefore, even if the protein expression level is high, the final activation can be affected by the influence of signaling due to other factors in the pathway (D’arcy, 2019). Apoptosis is regulated by the Bcl-2 family and involves spontaneous death through inducer expression (Eisenberg and Levanon, 2013). The expression of Bcl-2 is reduced by p53 within the mitochondria and the expression of Bax is promoted, thereby releasing cytochrome c from the inner membrane. This causes the sequential expression of the surrounding Caspase-9 and Caspase-3, which degrade the substrate and ultimately induce apoptosis (Oh et al., 2015; Park et al., 2015). Interestingly, the K159078 extract induced high Bax/Bcl-2 gene expression despite showing low morphological evidence of apoptosis. This discrepancy may result from post-transcriptional regulation, delayed protein translation, or non-apoptotic signaling influences. It also highlights the importance of integrating multiple apoptosis indicators for accurate interpretation. Although the current study investigated apoptosis-related gene expression, no direct phytochemical profiling such as HPLC or LC-MS analysis was conducted to identify or correlate specific compounds with observed bioactivities. Future studies will aim to perform compound-level analysis to better elucidate the active constituents responsible for anticancer effects.
Summary
In this study, we examined the apoptotic ability of AGS and HCT116 cancer cells treated with sorghum seeds collected from foreign regions and morphologically analyzed the apoptotic phenomenon of cancer cells using a microscope. In addition, the anticancer activity of sorghum seed extracts was demonstrated by confirming the mRNA expression levels of apoptosis-related genes such as Bax, Bcl-2, Caspase-9, and Caspase-3. This study is expected to provide important basic data on the anticancer activity of sorghum, a crop known to possess various physiological activities.