Introduction
Materials and Methods
Plant Materials, Different Light Spectra, and Cultivation Environments
Plant Growth Parameters
Statistical Analysis
Results and Discussion
Growth and Morphological Responses
Biomass Outcomes and Trends
Evaluation of Leaf Color Parameters
Conclusion
Introduction
The genus Tillandsia within the Bromeliaceae family, is categorized as an ornamental succulent in the horticultural industry. Tillandsia species typically grow epiphytically on tree branches or in rock crevices (Estrella-Parra et al., 2019) and are commonly called air plants because of their ability to thrive in soilless environments (Techato et al., 2014). Owing to this unique characteristic, Tillandsia can often be found attached to various artificial structures in urban environments, such as electricity poles, telephone lines, TV antennas, and building exteriors (Piazzetta et al., 2019). The surface of their leaves features trichomes, which are specialized structures that facilitate the absorption of moisture and various molecules from the air (Benz and Martin, 2006; Papini et al., 2010). Owing to these micro-morphological characteristics, Tillandsia is widely utilized as an indicator species for monitoring urban air pollution in Central and South American cities (Figueiredo et al., 2007; Sánchez-Chardi, 2016; Schrimpff, 1984; Wannaz and Pignata, 2006).
From a morpho-physiological perspective, the shoots of Tillandsia are covered with a thick, white epicuticular wax layer (Gerardo et al., 2005), enabling them to partially mitigate the abiotic stress caused by excessive direct sunlight (Pierce et al., 2001; Zheng and Li, 2023; Zheng et al., 2016). Additionally, they exhibit strong resistance to drought stress because of their utilization in Crassulacean acid metabolism (CAM) for photosynthesis (Hu et al., 2023; Nowak and Martin, 1997). Unlike typical plants, their roots are not primarily involved in water and nutrient absorption but are instead considered to function mainly as anchoring structures to support the weight of the shoot parts on attachment surfaces (Abril and Bucher, 2009).
Epiphytic plant species, including Tillandsia, possess unique and attractive characteristics and are native to specific habitats characterized by high-altitude environments, arid conditions, and limited organic matter, which restrict their natural distribution. Some epiphytic species exhibit low population densities in their habitats, making them rare and vulnerable to poaching (Carmona-Higuita et al., 2024; Pérez-López et al., 2023; Rauh, 1992). Owing to their rarity and the challenges associated with their propagation, these species are traded at very high prices in the horticultural market. If endangered and rare plant species can be efficiently cultivated in controlled environments, it could benefit both farmers and the ornamental horticultural industry.
Currently, physiological studies aimed at improving the cultivation efficiency of Tillandsia are lacking. Therefore, experimental approaches related to the physiology of the cultivation are required. As mentioned previously, Tillandsia is classified as a succulent species that undergoes CAM photosynthesis. Consequently, a large supply of water may not be necessary. Additionally, owing to the limited role of the roots, which primarily serve an epiphytic anchoring function rather than nutrient absorption, the use of substrates for nutrient supply is likely to be constrained. Thus, optimizing cultivation efficiency by controlling the light environment, such as light quality, photoperiod, and light intensity, should be prioritized.
Artificial light sources, such as light-emitting diodes (LEDs) or fluorescent lamps, are widely used in the horticultural industry and controlled environmental agriculture for photoperiod control and supplemental lighting (Hemming, 2009; Kim et al., 2023, 2024b; Locatelli et al., 2024; Park et al., 2012). The adjustment of light quality or spectra, along with the photoperiod and light intensity, is known to directly influence plant growth and development (Park et al., 2024; Yan et al., 2019; Ye et al., 2024). Such light environment controls are primarily applied in facility-based cultivation systems, particularly in controlled environments, such as closed-type plant factories and nursery systems that are unaffected by sunlight (Choi et al., 2024; Kim and Park, 2023; Kozai, 2013). Changes in light spectra have been reported to significantly influence various physiological responses in plants, including alterations in plant size and biomass, number of leaves and roots (Kim et al., 2024a; Shin et al., 2024), shoot and root development during cutting propagation (Cho et al., 2019; Lee and Nam, 2023), external quality (Lee and Nam, 2022), flower induction (Cerdán and Chory, 2003; Jang et al., 2023), and germination enhancement (Kwon et al., 2023). Different plant species respond uniquely to various light spectra based on their specific traits. This highlights the importance of applying LEDs with varying spectral distributions, including red, green, blue, and white light, to optimize growth and physiological responses according to the needs of each species.
In recent years, there have been active discussions on soilless cultivation technologies, including hydroponics and aeroponics, in controlled environments such as plant growth facility systems (Hussain et al., 2014; Lakhiar et al., 2018; Putra and Yuliando, 2015). However, as previously mentioned, the morpho-physiological characteristics of Tillandsia make it difficult to apply these cultivation methods. Therefore, to explore efficient cultivation methods for Tillandsia species, it is essential to consider their unique characteristics that differentiate them from common horticultural crops, emphasizing the use of non-substrate (i.e., soilless) and limited irrigation systems. Given these challenges, Tillandsia stands out as an excellent model for studying the growth and physiological responses of epiphytic plants under controlled environmental conditions due to its unique morpho-physiological traits. These include high water-use efficiency through CAM photosynthesis, a specialized root structure adapted for epiphytic lifestyles, and strong resistance to environmental stress. Research on Tillandsia deepens our understanding of epiphytic plants and contributes to expanding their commercial and ecological applications.
Accordingly, T. ionantha, a species predominantly available in the horticultural market and known for its unique physiological adaptability, was selected as the experimental plant. This selection aims to provide foundational insights into how different light spectra affect the growth and physiological responses of this epiphytic species under controlled environmental conditions.
Materials and Methods
Plant Materials, Different Light Spectra, and Cultivation Environments
Tillandsia ionantha, an ornamental succulent, was used as the experimental plant. The plants used in the experiment had shoot heights of approximately 3 cm and shoot widths of 5 cm. The study was conducted in a controlled environment facility located in the annex of the experimental greenhouse at the Department of Environmental Horticulture, Sahmyook University, Nowon-gu, Seoul. The experimental space measured 6.4 m × 5.2 m × 2.4 m, and the bed inside the space measured 1.2 m × 0.7 m × 0.6 m (width × length × height). The study, conducted in a controlled environment facility, lasted for a period of six months.
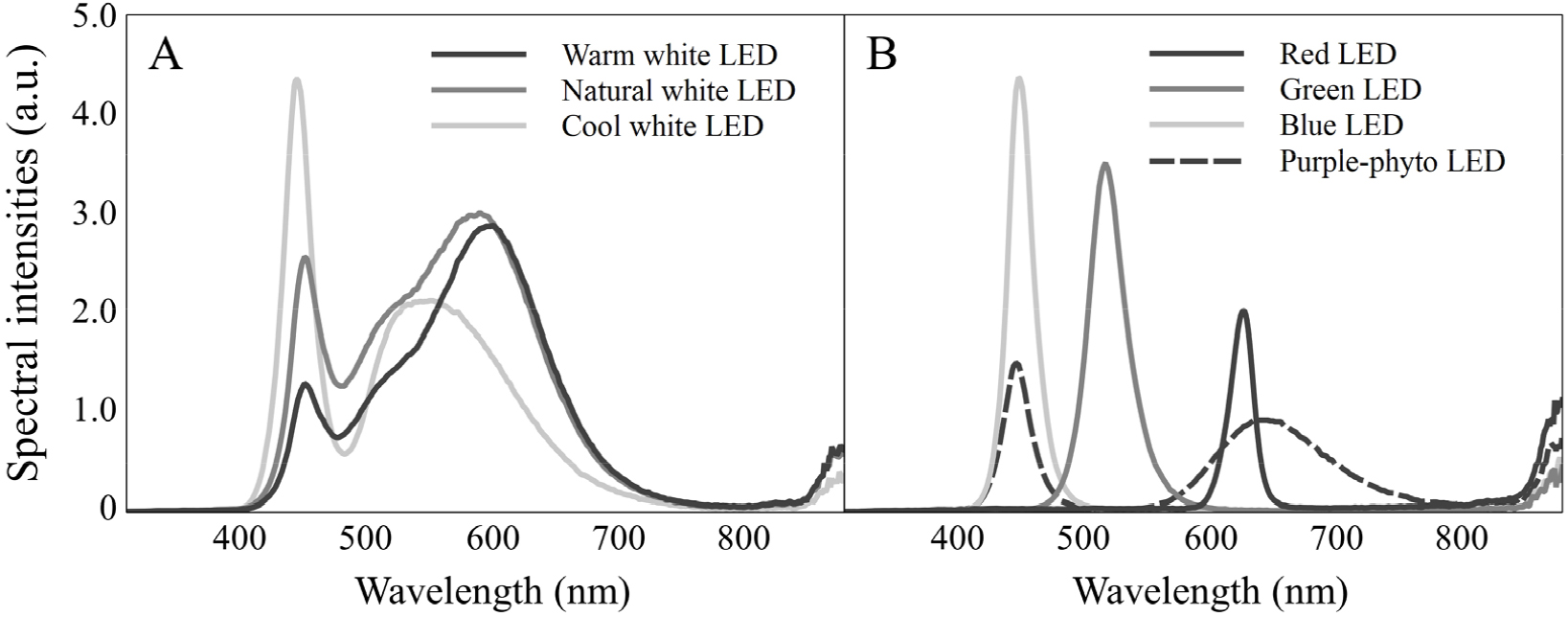
To investigate the effects of different light spectra on the growth of T. ionantha, seven types of light-emitting diodes (LEDs) were used, following the methodology of previous studies by Kim et al. (2024a) and Shin et al. (2024). These included red (630 nm), green (520 nm), blue (450 nm), purple phyto-LED (450 nm, 650 nm, and far-red wavelengths), warm white (3000 K), natural white (4100 K), and cool white (6500 K) lights (T5 LED, Zhong Shan Jinsung Electronic, China), as shown in Fig. 1. The purple phyto-LED for plant cultivation contains a relatively high proportion of far-red wavelengths (approximately 17%) in its spectral distribution. The LEDs used in this study had a length of 1.2 m, power consumption of 20 W, rated AC voltage of 220 V, and frequency of 60 Hz. The photon flux density (PFD) in the 350-800 nm range was standardized to approximately 100 µmol m-2 s-1 using a portable spectroradiometer (SpectraPen mini, Photon Systems Instruments, Czech Republic) to determine the appropriate distance between the plants and the LEDs.
The study was conducted under non-substrate conditions with plants suspended in the air using 1 mm thick wires. Irrigation was performed once every two weeks by spraying water until the entire surface of the plants was thoroughly wet. Additionally, every two months, the plants were immersed for one hour in a 1,000 ppm liquid fertilizer solution prepared a product with an N-P-K ratio of 7-10-6 (High-Grade S, Hyponex, Osaka, Japan). During the experimental period, the internal temperature was maintained at 20 ± 1°C using an automated heating and cooling system, and the relative humidity was maintained at 54.3 ± 14.3%.
Plant Growth Parameters
In this study, the growth parameters were measured to analyze the growth performance of T. ionantha grown under various light spectra. These parameters included shoot length, shoot diameter, root length, number of roots, leaf length, leaf width, number of leaves, shoot cover, shoot fresh and dry weights, root fresh and dry weights, total fresh and dry weights, shoot and root relative water content, total dry matter, and compactness index. Additionally, the external quality of the plants was evaluated using the Commission Internationale de l’Éclairage Lab (CIELAB) color space values, with specific parameters including L* (lightness), a* (red and green opponent colors), and b* (yellow and blue opponent colors).
Multiple indices were calculated to evaluate the growth responses of T. ionantha to various light spectra. Dry weight was assessed after drying samples in a heat dry oven (HK-DO135F, HANKUK S&I, South Korea) set to 85°C for 24 hours. Relative water content (RWC) and total dry matter (TDM) were calculated using Equations (1) (Lee and Nam, 2024) and (2), respectively, and the compactness index was determined using Equation (3) (Kim et al., 2016). CIELAB values were measured using a portable spectrophotometer (CM-2600d, Konica Minolta, Tokyo, Japan), set to CIELAB D65/10°, with the specular component included (SCI) to ensure accurate leaf surface reflectance measurements, as described by Lee (2023).
(FW: fresh weight; DW: dry weight; SDW: shoot dry weight; SL: shoot length)
Statistical Analysis
The experimental results were analyzed using a one-way analysis of variance (one-way ANOVA) with SAS 9.4 (SAS Institute, USA). Mean comparisons were performed using Duncan’s multiple range test (DMRT) at a significance level of 5% (p < 0.05). The experiment followed a completely randomized design (CRD), with three plants per replication and four replications, with a total of twelve plants per treatment.
Results and Discussion
Growth and Morphological Responses
The influence of the light spectra (or light quality) on plant growth and development has been extensively studied over the past several decades (Goto, 2003). Different light spectra induce phenotypic plasticity, leading to morpho-physiological changes such as shade avoidance response, development of specific vegetative and reproductive organs, and embryonic development. These changes affect plants throughout their growth cycles and at affecting various developmental stages (Lee et al., 2024a; Schmitt and Wulff, 1993; Stepanova et al., 2024; Wang et al., 2024; Zhang et al., 2024). Depending on the spectral distribution, plant species, or cultivar, these effects may promote or inhibit growth and development (Weinig and Delph, 2001). This process involves various photoreceptors such as phytochromes, cryptochromes, and phototropins (Christie, 2007; Fantini and Facella, 2020; Smith, 1982). Thus, understanding and optimizing spectral distribution settings are crucial for directing plant growth and development. Therefore, diverse experimental approaches tailored to specific plant species or cultivars are required.
In this study, we investigated the potential for cultivating Tillandsia ionantha under non-substrate conditions in a controlled environmental facility by examining its growth responses to various light spectra, considering its unique morpho-physiological characteristics. Rosette-forming plants, which either lack functional stems or have very short stems, can show significant changes in the length of vegetative organs under specific light conditions such as low-light intensity, those with a high proportion of far-red wavelengths, or even under certain monochromatic light that promotes elongation. These responses show some resemblance to the shade avoidance response observed in other plants with well-developed stems. Hirai et al. (2005) reported that visible light wavelength influences internode growth, which is a key parameter determining shoot length, and this effect varies among plant species. Similarly, light spectra are known to exert complex and diverse effects on root development (Kim et al., 2024a).
A representative image of the growth outcomes of T. ionantha plants grown under the influence of various light spectra is shown in Fig. 2. According to the growth and morphological changes observed in this study, the shoot length of T. ionantha was the longest under the blue LED light treatment (4.39 cm), as determined by Duncan’s multiple range test (DMRT). In comparison, it was shorter under warm white and cool white LED treatments, measuring 3.37 cm and 3.66 cm, respectively, suggesting that blue light promotes shoot elongation (Fig. 3). However, unlike shoot length, shoot width did not differ significantly among treatments.

Fig. 2.
Representative image of Tillandsia ionantha grown for six months under non-substrate and restricted irrigation conditions, influenced by various light spectra in a controlled environment facility. A) red; B) green; C) blue; D) purple phyto-LED; E) warm white (3000 K); F) natural white (4100 K); and G) cool white (6500 K) LED lights. The legend represents 3 cm.
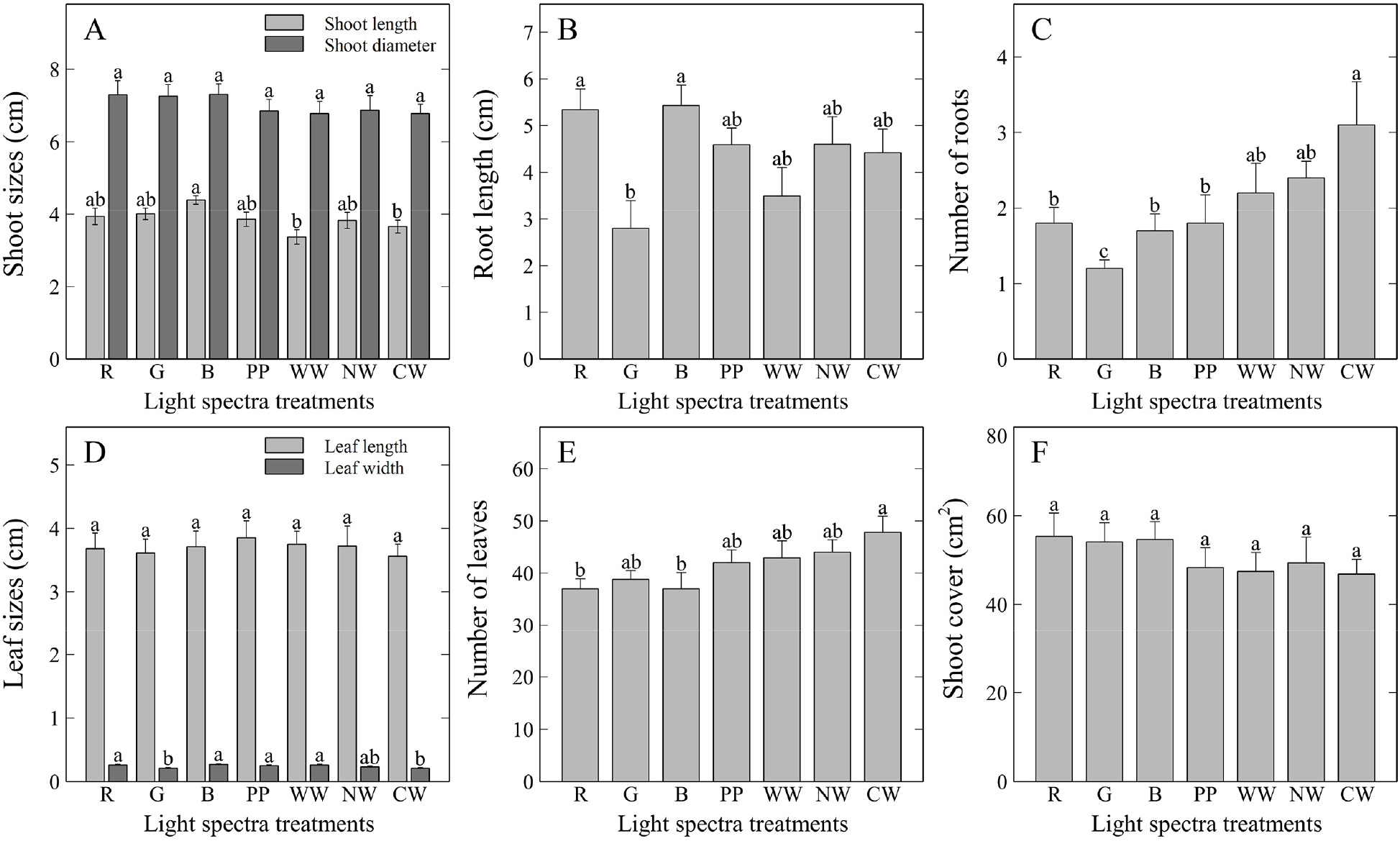
Fig. 3.
Growth and morphological responses of T. ionantha grown under various light spectra for six months. A) shoot sizes; B) root length; C) number of roots; D) leaf sizes; E) number of leaves; and F) shoot cover. R: red; G: green; B; blue; PP: purple phyto-LED; WW: warm white; NW: natural white; and CW: cool white LED lights. Vertical bars represent mean ± standard error (SE) (n = 4). Different lowercase letters indicate significant differences at p < 0.05 based on Duncan’s multiple range test (DMRT). The same lowercase letters indicate no significant differences.
For root length, red and blue LED treatments resulted in relatively longer roots, measuring 5.34 cm and 5.43 cm, respectively, compared to other treatments. Previous studies have reported that blue light increases stomatal size in chrysanthemums (Chrysanthemum) (Seif et al., 2021) and plays a more critical role than red light in improving the functionality of the photosynthetic apparatus in tomatoes (Solanum lycopersicum) (Izzo et al., 2020). Moreover, Ranade and Gil (2016) found that blue light treatments significantly improved the quality of Scots pine (Pinus sylvestris) seedlings during germination and hypocotyl development, indicating the diverse benefits of blue light. In contrast, red or green LED lights have been reported to promote plant size increases in Coleus cultivars (Park et al., 2024), highlighting species or cultivar-specific responses to different light qualities. Consistent with the results of this study, blue light was shown to be advantageous for significantly enhancing plant size-related parameters, such as shoot and root lengths in T. ionantha, supporting previous findings that plants respond selectively to different light qualities.
Regarding the number of roots, the cool white LED light treatment produced the highest count at 3.1, whereas treatments with two monochromatic LED lights (red and blue lights) and purple phyto-LED for plant cultivation yielded lower counts, ranging from 1.7-1.8. Notably, the green LED treatment resulted in the lowest root count (1.2), highlighting its relatively inhibitory effect on root development. In terms of leaf length, no significant differences were observed among the treatments. However, leaf width was relatively higher under red, blue, purple phyto-LED, and warm white LED light treatments, with values of 0.26 cm, 0.27 cm, 0.25 cm, and 0.26 cm, respectively. The cool white LED light treatment produced the highest count of 47.8 leaves, while the red and blue LED treatments resulted in the lowest counts of 37.0 leaves, suggesting that monochromatic light treatments can significantly suppress leaf development. In contrast, shoot cover showed no significant differences among the treatments.
These observations align with findings from previous studies, which have also reported specific responses of CAM plants to distinct light spectra. For instance, Wang et al. (2022) demonstrated that the CAM plant Ottelia alismoides exhibited increased leaf size under red LED light but a decrease under blue LED light. On the other hand, Macedo et al. (2011) found that the CAM plant Alternanthera brasiliana showed larger leaf thickness and leaf blade area under blue light. These findings suggest that light spectra should be carefully selected to optimize specific growth traits in CAM plants.
According to Cope et al. (2014), blue light inhibits stem elongation in lettuce (Lactuca sativa) and radishes (Raphanus sativus), while also promoting leaf expansion. This trend has also been observed in various cultivated species as well (Cosgrove, 1981). In the present study, blue light was found to be particularly effective in significantly enhancing shoot length, root length, and leaf width in T. ionantha. However, it also led to a significant decrease in the number of roots and leaves, contrasting with the observed increase in plant size. This suppression of root and leaf development may be explained by blue light’s pronounced effects on photosynthesis and photomorphogenesis, potentially shifting metabolic priorities toward shoot elongation.
To address this imbalance, a combination of blue and white lights, such as cool white LED lights, may be necessary. White light provides a broader spectrum of light that supports balanced development across multiple plant organs. Previous studies on other ornamental species have demonstrated that tailored light combinations can mitigate the trade-offs between shoot and root growth, further supporting this strategy in T. ionantha. For example, while blue light promotes shoot elongation, the diverse wavelengths of white light foster the development of roots and leaves, ensuring structural stability. These results suggest that specific light spectra can enhance certain growth parameters, while potentially disrupting the balance of overall growth, emphasizing the importance of constructing an appropriate spectral distribution for the efficient cultivation of T. ionantha.
As shown above, each light spectrum had varying effects on the growth parameters of different organs (i.e., shoots, roots, and leaves) in T. ionantha. The regulation of light spectra is not only used to promote plant growth in facility-based cultivation and supplemental lighting in plant nurseries but has also been applied to induce the development of in vitro plantlets in tissue culture (Batista et al., 2018; Seo et al., 2022), promote the expression of unique leaf patterns (Park et al., 2024), and encourage the development of specific organs such as roots and flowers (Jang et al., 2023; Kim et al., 2024a). Generally, plants grown under monochromatic light conditions exhibit restricted growth compared with those grown under purple phyto-LED and white LEDs, which provide a broader spectral distribution (Budavári et al., 2024; Kim et al., 2024a; Shin et al., 2024). However, some studies have reported that certain horticultural crops such as artichokes (Cynara scolymus) (Rabara et al., 2017) and Coleus cultivars (Park et al., 2024), show superior growth under monochromatic LED lights. Additionally, in crops such as cherry tomatoes (Solanum lycopersicum) and garden cress (Lepidium sativum), monochromatic lights have been reported to significantly improve functional traits such as antioxidant capacity (Kim et al., 2014; Ullah et al., 2019), indicating the benefits of using monochromatic lights and supplemental lighting.
Morphological parameters related to plant size in T. ionantha, such as shoot length, root length, and leaf width were found to be significantly enhanced by monochromatic LED light, including blue light. However, from a comprehensive perspective, unlike other horticultural crops grown using conventional methods, T. ionantha appears to be influenced by relatively limited parameters owing to non-substrate and restricted irrigation conditions. Considering the six-month cultivation period, T. ionantha was found to have extremely slow growth.
Biomass Outcomes and Trends
Biomass serves as a fundamental indicator of plant growth and productivity, reflecting the combined effects of physiological processes such as photosynthesis, water uptake, and nutrient assimilation. It is widely used in plant science to evaluate the efficacy of environmental treatments and their impacts on plant development, making it a critical parameter in understanding species-specific responses to varying light spectra.
For biomass (i.e., fresh and dry weights) and relative water content, shoot fresh and dry weights showed no significant differences among treatments according to DMRT (Fig. 4). However, the root fresh and dry weights were heavier under the warm white and cool white LED treatments. Combining the results for biomass and size-related parameters, it was determined that T. ionantha was relatively more affected by light spectra in the roots than in the shoots. In contrast, total fresh and dry weights showed no significant differences among the treatments. Overall, white LED lights (i.e., warm white and cool white) provided favorable conditions for root development, which was positively evaluated considering that root development plays an important role in the structural stability and epiphytic function of T. ionantha. These results also suggested the presence of growth imbalances between shoots and roots induced by different light spectra, which may be attributed to the characteristics of the species.
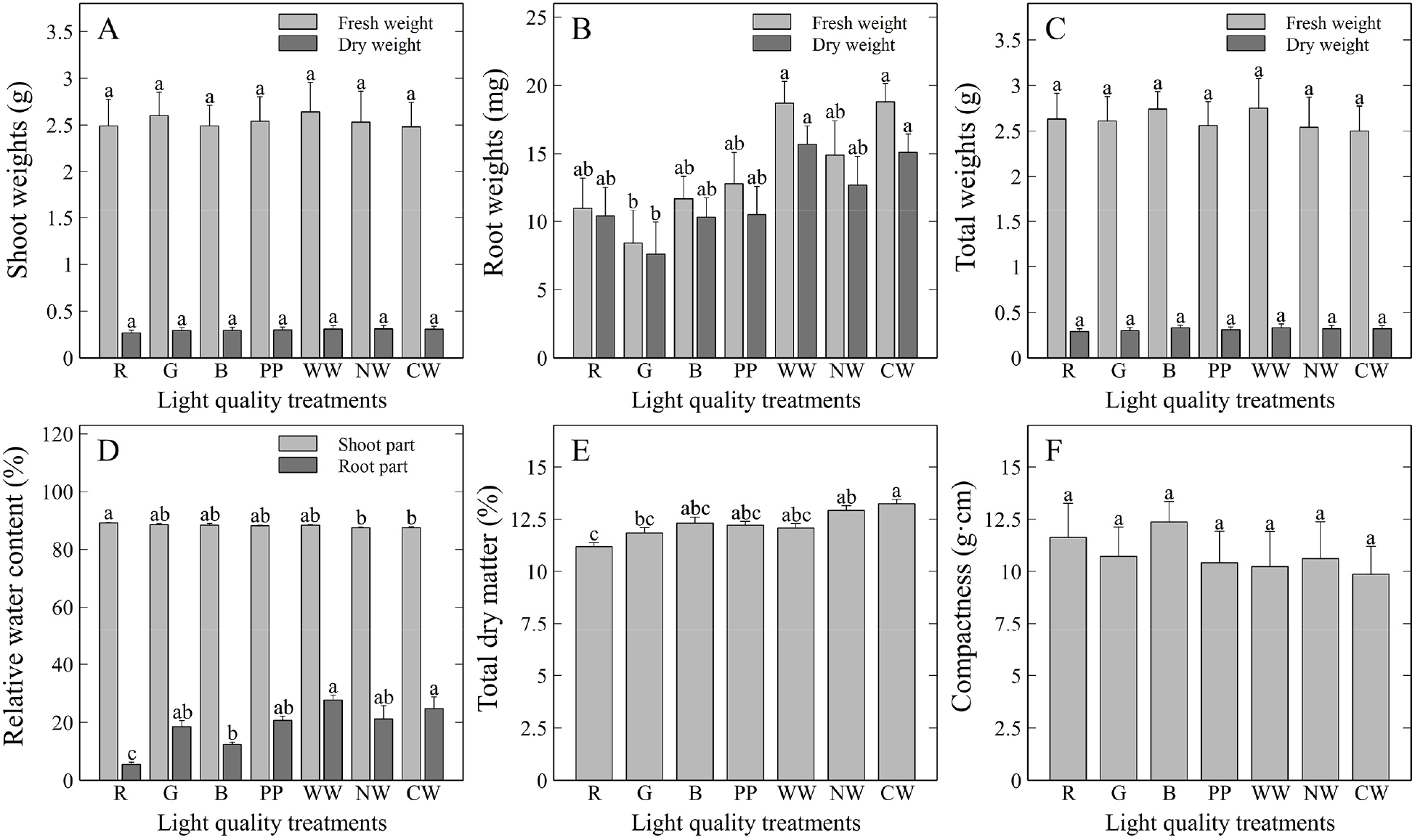
Fig. 4.
Biomass and relative water content differences of T. ionantha grown under various light spectra for six months. A) shoot weights; B) root weights; C) total weights; D) relative water content; E) total dry matter; and F) compactness index. R: red; G: green; B; blue; PP: purple phyto-LED; WW: warm white; NW: natural white; and CW: cool white LED lights. Vertical bars represent mean ± SE (n = 4). Different lowercase letters indicate significant differences at p < 0.05 based on DMRT. The same lowercase letters indicate no significant differences.
The differences in biomass responses between shoots and roots under various light spectra seem to stem from the different physiological requirements of each organ. While red and blue lights are generally effective in promoting photosynthesis and inducing shoot growth, white light, which contains a broader range of wavelengths, appears to be more favorable for the development of specific organs, such as roots. Red LED light treatment resulted in the highest shoot relative water content (88.3%), indicating that red light may improve T. ionantha’s water retention capacity. Conversely, the natural white and cool white LED treatments resulted in lower shoot relative water content. Root relative water content was higher in the warm white (27.7%) and cool white (24.7%) treatments than in the other treatments. However, the overall average was lower than that of the shoots. The highest total dry matter value (13.2%) was recorded under the cool white LED light treatment, whereas the compactness index showed no statistically significant differences among treatments.
These results highlight the importance of strategically adjusting spectral distributions to utilize light spectra complementarily and achieve a balance between plant growth and biomass enhancement.
Evaluation of Leaf Color Parameters
The CIELAB color space values are suitable for detecting subtle changes in plant coloration (Ahn et al., 2024). It has been used effectively to evaluate the external quality of various horticultural crops. These include ornamental flowers (Jang et al., 2023; Xie et al., 2023), foliage plants (Lee et al., 2024b; Shin et al., 2023), succulents (Cabahug-Braza et al., 2023; Lee and Nam, 2022), and medicinal crops (Kim et al., 2024a, 2024b). In CIELAB, the detailed parameters include L*, which represents lightness; a*, which indicates red (+) and green (-) tones; and b*, which indicates yellow (+) and blue (-) tones (Kim et al., 2024a).
CIELAB color space analysis revealed that the highest L* value was observed in the natural white LED light treatment at 64.39 (Fig. 5). The increase in L* may be attributed to the development of an epicuticular wax layer (Bibi, 2017; Bibi et al., 2021) or trichomes. Further studies are required to microscopically examine the effects of light spectra on these characteristics or associated structures. Meanwhile, a* had the lowest average value (-5.73) under the green LED light treatment, indicating that the leaf color shifted closer to green. In contrast, significant increases in this parameter were observed in all treatments, except for warm white LED light. For b*, higher values of 19.31 and 19.22 were observed under green and purple phyto-LED light treatments, respectively, indicating that the leaf color shifted closer to the yellow tone. Conversely, all treatments except for red LED light exhibited significantly lower b* values. Consequently, differences in light spectra appear to induce changes in leaf pigments, as has been reported in previous studies (Jang et al., 2023; Park et al., 2024).
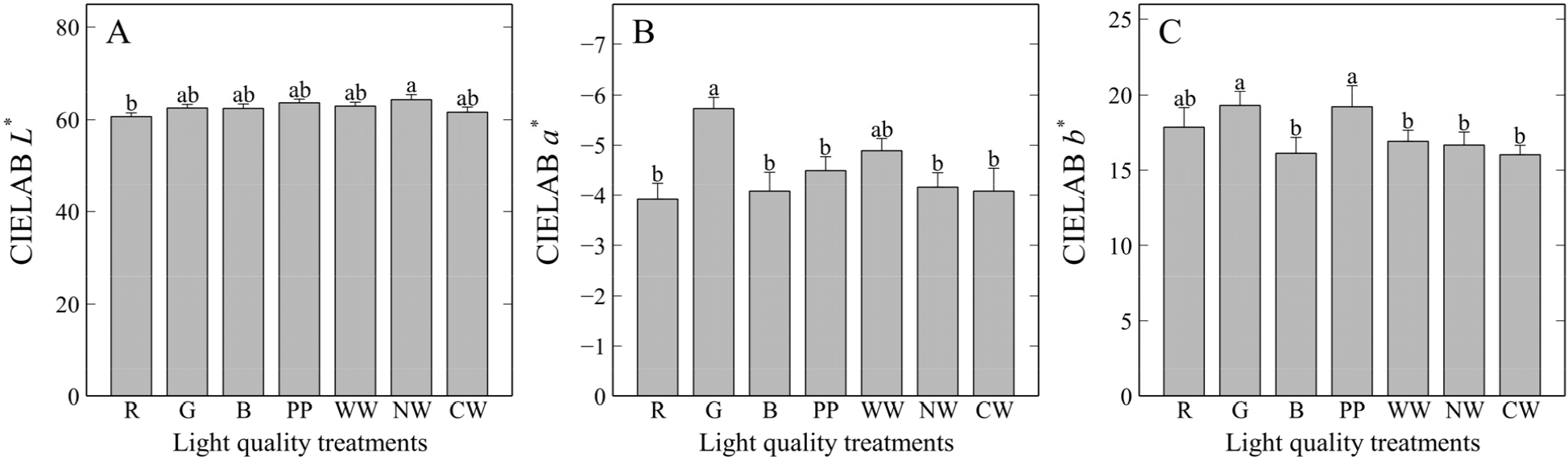
Fig. 5.
Leaf color reading values of Commission Internationale de l’Éclairage Lab (CIELAB) of T. ionantha grown under various light spectra for six months. A) CIELAB L*; B) CIELAB a*; and C) CIELAB b* values. R: red; G: green; B; blue; PP: purple phyto-LED; WW: warm white; NW: natural white; and CW: cool white LED lights. Vertical bars represent mean ± SE (n = 4). Different lowercase letters indicate significant differences at p < 0.05 based on DMRT. The same lowercase letters indicate no significant differences.
According to Cárdenas-Pérez et al. (2024), CIELAB color space values are also a promising parameter for predicting plant biomass. Subsequent studies should aim to integrate CIELAB analysis with growth and biomass accumulation metrics to explore their potential correlations. These findings suggest that CIELAB analysis can serve as a valuable tool not only for assessing the effects of light spectra on pigments and structural features but also for broader applications in plant physiology.
Overall, this study highlights the advantages of controlled environments that enable precise regulation of external environmental factors. However, this study focused solely on light spectra as a single variable and did not experimentally apply other critical factors, such as light intensity, photoperiod, temperature, relative humidity, and CO2 level. These variables are likely to significantly influence the growth and morpho-physiological responses of T. ionantha, necessitating an understanding of both individual factors and their interactions. Future research should expand to include these additional variables, examining their combined effects on the growth and physiological characteristics of T. ionantha. Such investigations could inform the development of optimized cultivation strategies that leverage the unique attributes of this species.
Conclusion
This study investigated the influence of different light spectra on the growth and external quality of Tillandsia ionantha, commonly known as an air plant, and widely used as an ornamental succulent in horticultural markets. The findings revealed that size-related parameters, such as shoot length, root length, and leaf width, were significantly enhanced under blue LED light, indicating their effectiveness in promoting plant size. In contrast, the number of roots and leaves developed most effectively under cool white LED light, highlights the advantages of broad-spectrum light for balanced development. Biomass analysis revealed a growth imbalance between shoots and roots, with root biomass being the highest under warm white and cool white LED lights, suggesting that white LED lights are particularly suitable for promoting root development. Red LED light improved the relative water content of shoots, thereby enhancing their water retention capacity. External quality was evaluated using the CIELAB color space, revealing the notable changes in leaf coloration under various light spectra. The highest L* value, representing lightness, was observed under the natural white LED light treatment, whereas a* and b* parameters, indicating red-green and yellow-blue tones, respectively, varied significantly depending on the spectral distributions. These results underscore the importance of selecting the appropriate light spectra to optimize both the growth parameters and external quality of T. ionantha. Based on these findings, a complementary combination of monochromatic and white lights may be necessary to achieve balanced shoot and root growths. Furthermore, when considering the six-month experimental period, this study confirmed the feasibility of cultivating epiphytic plant species, such as T. ionantha, under non-substrate and restricted irrigation conditions in controlled environments. Future research incorporating diverse environmental factors could further refine these findings. Overall, these results provide useful information for the stable cultivation of Tillandsia species and other epiphytic plants in controlled environments, supporting their application in ornamental plant cultivation and the horticultural industry.