Introduction
Materials and Methods
Experimental materials
Lactobacillus screening for the SSF of Dunggulle
Temperature-dependent Dunggulle SSF and lactic acid bacterial growth assays
Investigation of the bioactive substances present in the SSF products
Statistical analysis
Results and Discussion
Selection of the optimal Lactobacillus strain for SSF
Effect of fermentation temperature on SSF using L. buchneri
Evaluation of the antioxidant activities of the fermentation products under different SSF conditions
Conclusion
Introduction
Functional plants such as Dunggulle (Polygonatum odoratum) have been used for thousands of years as traditional medicines and health supplements in Korea and other East Asian countries (Zhang et al., 2014). Dunggulle is rich in mucilage and contains many functional components such as starch, carbohydrates, amino acids, and alkaloids. It is also known to be closely related to various adult diseases including obesity, malnutrition, pulmonary tuberculosis, cardiac disease, hypotension, and hypoglycemia (Kang et al., 2014; Kim et al., 2005; Lee and Lee, 1994; Shu et al., 2009).
Although such herbal medicines provide natural ingredients with a variety of physiological functions, some have complex structures that hinder their absorption by organisms (Zhang et al., 2023). In addition, the majority of bioactive ingredients present in herbal medicines are found in extremely low quantities, and are often accompanied by components that are toxic to humans and animals (Charen and Harbord, 2020; Yang et al., 2018). To address these issues, herbal fermentation has been adopted as a traditional processing technique to increase the efficacies of natural herbal medicines, reduce their toxicity profiles, and generate new chemical components (Li et al., 2020; Wu et al., 2013). Furthermore, when the fermentation of herbal medicines is performed using the appropriate beneficial bacteria, the contents of bioactive substances can be enhanced, while also providing the host with probiotics that are beneficial to the gut microbiota and the immune system (Zhang et al., 2023).
The fermentation of natural herbal medicines can be broadly categorized into liquid fermentation (LF) and solid-state fermentation (SSF). SSF is particularly advantageous due to its simple fermentation process, low cost, high productivity levels, high bioconversion rates, and reduced amounts of wastewater generation, whereas LF is known to exhibit a low efficiency (Canoy Postigo et al., 2021; Garrido-Galand et al., 2021; Lee et al., 2014). Owing to these advantages, various SSF methods utilizing single or combined strains under low moisture conditions have been investigated to increase the functional components of herbal medicines. One of the key effects associated with an improved bioactivity is the increased antioxidant activity (Zhang et al., 2023), which plays an important role in protecting the body from oxidative stress. Notably, oxidative stress is associated with a variety of chronic diseases, including cardiovascular disease, cancer, and neurodegenerative disorders. Therefore, due to the increasing awareness of the possibilities of probiotics and their close relationship with human and animal health, efforts to establish suitable probiotic strains and scientific SSF conditions continue to attract growing attention. Such efforts ultimately aim to improve the biological functionalities and safety profiles of natural herbal medicines.
In the SSF of herbal medicines, the composition and function of the selected microorganism have a significant impact on the generated fermentation metabolites and the pharmacological activities of the final fermentation products. However, a lack of scientific standards exist regarding the optimal fermentation conditions for such processes, including the pH, temperature, and incubation time (Arora et al., 2018; Zhang et al., 2023). Therefore, this study aims to investigate the changes in fermentation properties and antioxidant activities of Dunggulle fermentation products using different SSF temperatures using probiotic lactic acid bacteria. Subsequently, the optimal probiotic lactic acid bacterium is identified for the SSF of Dunggulle, and the potential of SSF to enhance the bioactive properties of natural Dunggulle is evaluated. The results of this study will be expected to contribute to the development of the SSF technology and the efficient utilization of herbal medicines.
Materials and Methods
Experimental materials
The herbal medicine used in this study was the dried roots of Dunggulle (Polygonatum odoratum, Sechang Trading Co., Ltd., Korea), which was harvested after 3 years of cultivation in Yeongju, Gyeongsangbuk-do, South Korea. The dried roots were prepared in a fine powder form using a crusher (Nankook Crusher Co., Ltd., Korea) and stored at 4°C prior to use in the SSF process.
Lactobacillus screening for the SSF of Dunggulle
Various potential probiotic lactic acid bacteria were screened for the purpose of SSF, namely Lacticaseibacillus rhamnosus, L. acidophilus, L. reuteri, L. plantarum, and L. buchneri (Agrokorea Co., Ltd., Korea). These strains were each inoculated at 1 × 104 CFU/g on a solid phase medium containing a 1:1 (g/v) mixture of Dunggulle powder and distilled water, followed by incubation at 37°C for 36 h. Each strain was cultured in triplicate, and the extent of strain growth was analyzed in terms of the number of live bacteria in each culture (CFU/g). The optimal strain of Lactobacillus was selected and used for subsequent SSF experiments.
Temperature-dependent Dunggulle SSF and lactic acid bacterial growth assays
To evaluate the effects of different SSF culture conditions on the obtained SSF products, the optimal bacterial strain (as identified in the previous study) was incorporated into the Dunggulle solid phase medium, and the antioxidant activities of the products were evaluated following fermentation at different incubation temperatures. After modification and preliminary validation of the methods described by Slizewska and Chlebicz-Wójcik (2020) and Carneiro et al. (2013), the solid medium was prepared by mixing the herbal powder and distilled water in a 1:1 (g/v) ratio, and the bacterial strain was inoculated (1 × 105 CFU/g) on the prepared medium. Subsequently, the incubation temperature was set according to the method of Hussain et al. (2016), and incubation was performed at three different temperatures (25, 30, and 35°C) for 8 d. Each culture was performed in triplicate, and samples were taken for analysis every 2 d during the course of the culture.
To analyze the growth of lactic acid bacteria as a function of the incubation temperature during SSF, the collected SSF products were diluted stepwise to 10-8 using sterile 0.85% saline, and bacterial counts were performed using MRS agar (BD, Franklin Lakes, NJ, USA). More specifically, the prepared plates were incubated at 37°C for 2 d and the live bacterial counts (CFU/g) were determined. Each experiment was performed in triplicate. To measure the system pH, the SSF products were mixed with distilled water in a 1:9 (g/v) ratio, and incubated in a shaking incubator (BF-60SIR, BioFree Co., Ltd., Korea) for 10 min at 100 rpm. After shaking, the mixture was filtered through filter paper (F1004 Grade, CHM Lab Croup, Spain) to remove any solids, and the pH was measured using a pH meter (Starter 3100, Ohaus Co., USA).
Investigation of the bioactive substances present in the SSF products
The properties of the bioactive substances present in the SSF products were measured with regards to their DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activities, ABTS (2,2’-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid)) radical scavenging activities, total phenolic contents, and total flavonoid content. For this purpose, extracts were obtained according to the method of Lee et al. (2012), wherein the dried sample (3 g) was placed in a flask with hot (95°C) distilled water (10×), and extracted over 3 h with constant stirring. The extraction process was performed twice and the extracted solutions were combined. Subsequently, the mixed extracts were centrifuged at 3,000 rpm for 20 min, and the supernatant was filtered through filter paper (No. 4, Whatman, UK), lyophilized, and dissolved in dimethyl sulfoxide (DMSO, 10 mg/mL total extract concentration) for analysis.
The DPPH radical scavenging activity was measured according to the method of Carneiro et al. (2013), with some modifications. In a 96-well plate, the SSF product extract (20 µL) was mixed with a solution of DPPH (180 µL) and reacted for 30 min in the dark. After this time, the absorbance was measured at 517 nm using a microplate reader, and the DPPH radical scavenging activity was calculated as described by Carneiro et al. (2013).
The ABTS radical scavenging activity was determined based on the method of Re et al. (1999). More specifically, the ABTS radical cation solution was prepared by mixing a 7 mM ABTS solution with a 2.45 mM potassium persulfate solution in a 1:1 ratio (v/v), and allowing to stand in the dark for 12-16 h prior to testing. The ABTS radical cation solution was then diluted with distilled water to give an absorbance of 0.700 ± 0.05 at 734 nm. Subsequently, in a 96-well plate, the diluted ABTS solution (900 µL) was mixed with the SSF product extract (100 µL), and after allowing to stand for 1 min (up to 6 min) at room temperature, the absorbance was measured at 734 nm. The ABTS radical scavenging activity was calculated as described by Prior et al. (2005).
The total phenolic content was determined according to the method of Folin and Ciocalteu (1927) with some modifications. More specifically, the Folin-Ciocalteu reagent (0.2 mL) was added to a test tube containing the extract (0.2 mL) and distilled water (1.8 mL). The resulting mixture was allowed to react for 3 min, and after this time, a saturated sodium carbonate solution (0.4 mL) was added and mixed. Subsequently, distilled water (1.4 mL) was added, and the mixture was allowed to react for 1 h at room temperature. After this time, the absorbance was measured at 725 nm using a microplate reader, and results were expressed as millimolar gallic acid equivalents (mM GAE) using a calibration line ranging from 0.25 to 1.00 mM.
The total flavonoid content was determined based on the methods of Park et al. (2008) and Saeed et al. (2012). The extract solution (300 µL) was mixed with 30% methanol (3.4 mL), a 0.5 M NaNO2 solution (150 µL), and a 0.3 M AlCl3-6H2O solution (150 µL). After allowing to react for 5 min, a 1 M NaOH solution (1 mL) was added and mixed thoroughly. The absorbance was then measured at 510 nm against the indicator. The total flavonoid content was calculated using standard curves prepared with quercetin solutions at concentrations of 20, 40, 60, 80, and 100 mg/L and was expressed as quercetin equivalents (µg/mL QE).
Statistical analysis
All experimental results were expressed as the mean ± standard deviation based on assays performed in triplicate. The results were analyzed using SPSS statistical software (version 18; SPSS Inc., Chicago, IL, USA). One-way analysis of variance (ANOVA), two-way ANOVA, and Duncan’s multiple range test were employed to test for significant differences at p < 0.05.
Results and Discussion
Selection of the optimal Lactobacillus strain for SSF
L. rhamnosus, L. acidophilus, L. reuteri, L. plantarum, and L. buchneri were screened to identify the optimal lactic acid bacterium for SSF, and the obtained results (i.e., bacterial growth and pH values) are presented for the various SSF cultures in Table 1. All of the investigated lactic acid bacteria exhibited growth on the Dunggulle solid medium, with L. buchneri demonstrating the highest colony count (1180.00 ± 110.00 CFU/g) (p < 0.05), followed by L. plantarum, L. reuteri, L. acidophilus, and L. rhamnosus (p < 0.05). Additionally, it was observed that the pH values of the solid cultures tended to decrease with superior bacterial growth, resulting in significantly lower values for L. buchneri and L. plantarum than for the other bacteria (p < 0.05).
To date, various lactic acid bacteria, archaea, yeasts, and molds have been used for the fermentation of herbal medicines (Li et al., 2020). For the purpose of this study, the effects of Lactobacillus spp. were considered due to the fact that these bacteria are well-known probiotics in humans and animals (Anee et al., 2021; Kim et al., 2019). Based on the results presented in Table 1 for the SSF of Dunggulle, it was confirmed that L. buchneri gave the optimal results. The Lactobacillus species evaluated in the current study have been previously employed in advanced research on fermentation to enhance the bioactive compounds present in traditional herbal medicines. For example, L. buchneri was used for the fermentation of Triticum aestivum, Helianthus tuberosus, and Smallanthus sonchifolius (Kang et al., 2020), L. plantarum was used for the fermentation of Rhizoma Atractylodis macrocephalae (Wang et al., 2015), and L. reuteri was used for the fermentation of Angelica sinensis (Huang et al., 2014). It has also been reported that compared to traditional methods that rely on spontaneous fermentation (Bose et al., 2012; Ma et al., 2025; Zhang et al., 2023), herbal fermentation processes performed using probiotic lactic acid bacteria may be more efficient in enhancing the therapeutic effects of herbal ingredients and reducing toxic side effects.
Table 1
Screening of lactic acid bacteria for the SSF of Dunggulle
| L. rhamnosus | L. acidophilus | L. reuteri | L. plantarum | L. buchneri | |
| Bacterial growth1) | 4.20 ± 0.40e | 76.00 ± 4.50d | 560.00 ± 30.00c | 840.00 ± 20.00b | 1180.00 ± 110.00a |
| pH change2) | 0.30 ± 0.05c | 0.42 ± 0.06c | 0.89 ± 0.04b | 1.40 ± 0.01a | 1.10 ± 0.02ab |
Effect of fermentation temperature on SSF using L. buchneri
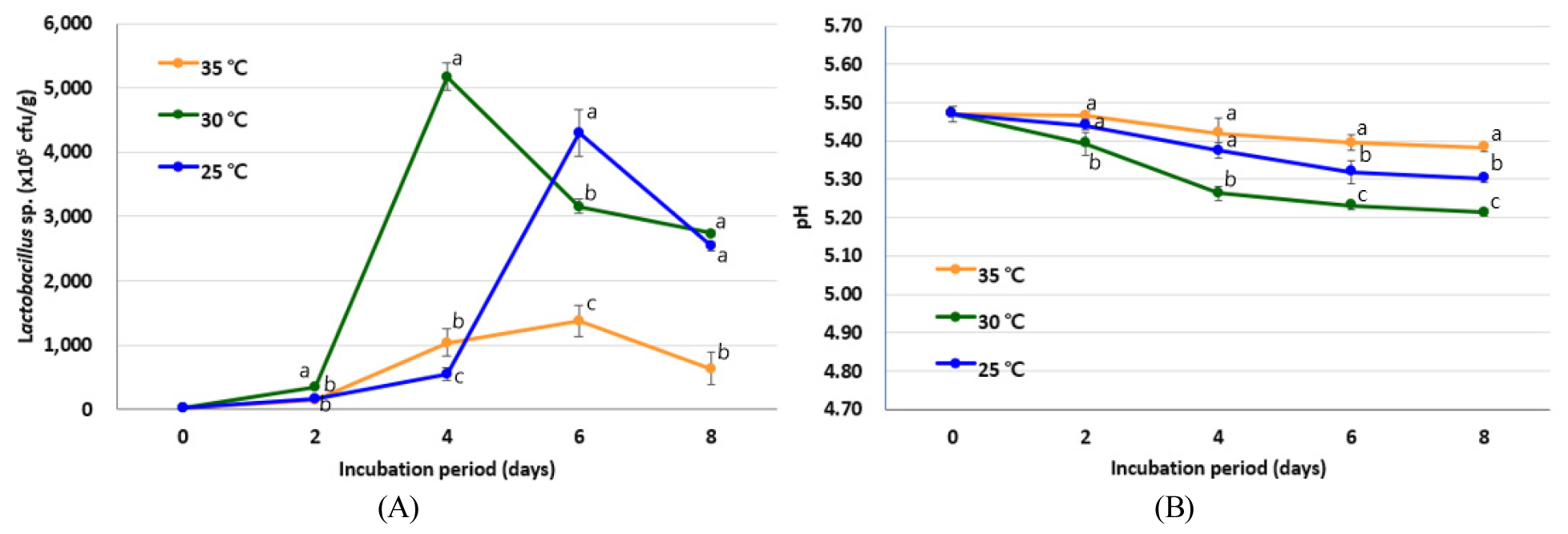
Fig. 1 shows the effects of different fermentation temperatures on the growth of L. buchneri and the resulting pH changes following incubation for 8 d at 25, 30, and 35°C. As mentioned above, this bacterium was selected for subsequent experiments due to the fact that it exhibited the greatest degree of bacterial growth during the SSF of Dunggulle.
Prior to incubation, the bacterial count was determined to be 30.30 ± 2.35 × 105 CFU/g (Fig. 1(A)), while on the second day of fermentation, it was found that an incubation temperature of 30°C led to a higher bacterial count (i.e., 337.33 ± 7.02 × 105 CFU/g) than those obtained at the other temperatures (p < 0.05). Notably, at 30°C, the extent of bacterial growth increased significantly after the second day of incubation, reaching a peak (5,173.33 ± 215.02 × 105 CFU/g) on the fourth day, after which a decreasing trend was observed. This peak bacterial count was also higher than those achieved at other incubation temperatures. More specifically, for the fermentation process performed at 25°C, the bacterial count increased significantly after day 4, peaking at 4,303.33 ± 468.01 × 105 CFU/g on day 6, and decreasing thereafter. Additionally, at 35°C, bacterial growth peaked at 1,376.67 ± 344.43 CFU/g on day 6, and declined thereafter, maintaining the lowest trend of bacterial growth over the entire incubation period compared to the other temperatures. Furthermore, as shown in Fig. 1(B), the pH was 5.47 ± 0.028 before fermentation, and decreased as the fermentation process progressed, reaching 5.21-5.38 at the end of the fermentation period (day 8). The earliest drop in pH was observed for the 30°C system due to the more rapid bacterial growth observed under these conditions, and this temperature also yielded the lowest pH throughout the entire incubation period.
It is well known that SSF is affected by a number of factors, including the fermentation temperature, the medium pH, the inoculum, and the incubation time, which also affect the quality of the fermented product. In the preliminary experiments performed herein, good bacterial growth was observed when distilled water (pH 7.0) was mixed with the SSF substrate (i.e., the dried herb) in a 1:1 (v/g) ratio. Thus, using these conditions, SSF was performed using L. buchneri at different culture temperatures and incubation times, wherein L. buchneri exhibited the greatest degree of bacterial growth over a 4 d incubation period at 30°C (Fig. 1). The ideal temperature for Lactobacillus growth is generally considered to be between 30 and 40°C, while the optimal pH is between 5.5 and 6.2°C. However, a wide variety of lactic acid bacteria are able to grow at temperatures ranging from 2 to 53°C and at pH values between 4.5 and 6.5, with some species thriving under even lower pH conditions (König and Berkelmann-Löhnertz, 2017). For example, Slizewska and Chlebicz-Wójcik (2020) reported that the maximum growth of Lactobacillus spp. was obtained during SSF using a substrate powder to water ratio of 1:1.5, a pH of 6.0, and a temperature of 37°C. In addition, Cho et al. (2007) reported that the incubation of L. buchneri at 30°C and pH 5.0 led to the highest GABA content after 48 h. Furthermore, the fermentation of L. brevis with Artemisia princeps Pamp for 3 d at 30°C gave good results (Joh et al., 2010). Moreover, favorable results were obtained upon the incubation of B. longum, L. acidophilus, and Leuconostoc mesenteroides with Codonopsis lanceolata for 48 h at 30°C (Weon et al., 2013), and the incubation of B. breve and B. bifidum with Radix Astragali at 30°C for 3 d (Sheih et al., 2011).
Evaluation of the antioxidant activities of the fermentation products under different SSF conditions
The antioxidant activities of the Dunggulle SSF products obtained using L. buchneri under different conditions were subsequently evaluated, as detailed in Table 2. It was found that the antioxidant activity (i.e., DPPH radical scavenging activity, ABTS radical scavenging activity, total phenolic content, and total flavonoid content) increased at all incubation temperatures (25, 30, and 35°C) compared to those obtained pre-fermentation.
Table 2
Antioxidant activities of the Dunggulle SSF products obtained using L. buchneri under different culture conditions (temperatures and times)
| Temperature | Incubation period (d) | |||||
| 0 | 2 | 4 | 6 | 8 | Mean ± SE | |
| DPPH radical scavenging activity (%) | ||||||
| 25°C | 22.96 ± 1.79d | 28.01 ± 1.50Cc | 33.66 ± 1.13Bb | 35.44 ± 1.24Ba | 33.24 ± 1.80Bb | 30.66 ± 0.253B |
| 30°C | 22.96 ± 1.79b | 37.23 ± 1.15Aa | 37.53 ± 1.93Aa | 37.91 ± 1.95Aa | 36.58 ± 1.47Aa | 34.44 ± 0.305A |
| 35°C | 22.96 ± 1.79d | 34.97 ± 1.77Bbc | 36.29 ± 1.93Aab | 37.24 ± 1.80ABa | 34.59 ± 1.74Bc | 33.21 ± 0.226A |
| Mean ± SE | 22.96 ± 0.326e | 33.40 ± 0.355d | 35.83 ± 0.326b | 36.86 ± 0.365a | 34.80 ± 0.326c | |
| ABTS radical scavenging activity (%) | ||||||
| 25°C | 49.31 ± 2.12c | 53.54 ± 1.75Bb | 56.22 ± 1.30Ba | 55.74 ± 0.52ab | 54.32 ± 1.59ab | 53.82 ± 0.358A |
| 30°C | 49.31 ± 2.12d | 58.47 ± 0.14Aa | 59.30 ± 0.24Aa | 55.18 ± 0.66b | 52.96 ± 0.21c | 55.05 ± 0.406A |
| 35°C | 49.31 ± 2.12c | 57.11 ± 1.55Aa | 57.66 ± 1.89Ba | 55.81 ± 2.08a | 52.49 ± 1.49b | 54.47 ± 0.333A |
| Mean ± SE | 49.31 ± 0.465d | 56.37 ± 0.476ab | 57.73 ± 0.450a | 55.58 ± 0.500b | 53.26 ± 0.476c | |
| Total phenolic content (mM GAE) | ||||||
| 25°C | 0.30 ± 0.002c | 0.30 ± 0.002Bc | 0.31 ± 0.010Bb | 0.32 ± 0.009Bab | 0.33 ± 0.004a | 0.312 ± 0.002C |
| 30°C | 0.30 ± 0.002c | 0.33 ± 0.012Aab | 0.35 ± 0.005Aa | 0.35 ± 0.005Aa | 0.32 ± 0.016b | 0.329 ± 0.002A |
| 35°C | 0.30 ± 0.002c | 0.33 ± 0.006Aab | 0.33 ± 0.017Bab | 0.34 ± 0.013Ba | 0.32 ± 0.006b | 0.322 ± 0.002B |
| Mean ± SE | 0.30 ± 0.003d | 0.32 ± 0.003c | 0.33 ± 0.003ab | 0.34 ± 0.003a | 0.32 ± 0.003bc | |
| Total flavonoid content (µg/mL QE) | ||||||
| 25°C | 67.75 ± 1.26b | 69.30 ± 1.30Bb | 69.83 ± 1.63Bb | 76.50 ± 1.41Aa | 68.83 ± 1.53b | 70.44 ± 0.325B |
| 30°C | 67.75 ± 1.26d | 79.70 ± 1.30Aa | 76.83 ± 1.53Aab | 71.50 ± 1.83Bc | 69.50 ± 1.83cd | 73.06 ± 0.348A |
| 35°C | 67.75 ± 1.26c | 71.17 ± 2.08Bab | 71.00 ± 1.73Bab | 73.50 ± 2.00Ba | 69.00 ± 1.38bc | 70.48 ± 0.355B |
| Mean ± SE | 67.75 ± 0.444d | 73.39 ± 0.439a | 72.56 ± 0.444b | 73.83 ± 0.444a | 69.11 ± 0.444c | |
More specifically, after day 2 of SSF, the DPPH radical scavenging activity was found to be significantly higher (p < 0.05) for the fermentation products obtained at 30°C compared to those produced at 35 and 25°C, and no significant changes were observed beyond this point. However, it should be noted that slightly higher activities were observed on day 6 for the 30 and 35°C conditions. Additionally, on day 2, the fermentation products generated at 25°C exhibited the lowest DPPH scavenging activity compared to those produced at the other temperatures (p < 0.05). In terms of the ABTS radical scavenging activity, the highest value was achieved (p < 0.05) on day 4 of the SSF process performed at 30°C; reduced activities were observed for the products generated at 35°C (moderate activity) and 25°C (lowest activity). Beyond this peak on day 4 at 30°C, a downward trend was observed thereafter. With regards to the total phenolic content, the highest values were observed for the fermentation products produced on days 4 and 6 at a temperature of 30°C (p < 0.05). For both the 30 and 35°C conditions, the phenolic contents peaked on days 4 and 6, respectively, prior to subsequently decreasing, while at 25°C, the phenolic content continued to increase over time. Moreover, on days 2 and 4, the highest total flavonoid contents (p < 0.05) were observed for the SSF products generated at 30°C. A peak was observed for the 30°C fermentation product on day 2 followed by a declining trend thereafter, while the 35 and 25°C fermentation products reached their peak phenolic contents on day 6.
In a similar report, it was found that the use of probiotics to perform SSF had a positive effect on the antioxidant activities of traditional herbal medicines (Lee et al., 2015). In addition, L. plantarum and E. faecium have been demonstrated to significantly enhance the production of bioactive substances, such as polysaccharides, total saponins, and flavonoids from Astragalus membranaceus (Qiao et al., 2018). Furthermore, fermentation with Lactobacillus pentoses increased the quercetin and kaempferol contents of Lespedeza cuneata G. Don extracts by 242.9 and 266.7%, respectively (Seong et al., 2017), and the fermentation of Puerariae Radix with Bifidobactericum breve increased the daidzein and genistein contents by 785 and 1,010%, respectively (Wen et al., 2010). In the current study, the SSF of Dunggulle using L. buchneri at 30°C led to a 165% enhancement in the DPPH radical scavenging activity on day 6, in addition to a 120% increase in the ABTS radical scavenging activity on day 4, a 117% increase in the total phenolic content on days 4 and 6, and a 118% increase in the total flavonoid content on day 2. Although the intracellular fibrous structures of natural herbs and the presence of colloids hinder the release of active ingredients from the herbs themselves, the above results clearly demonstrate that fermentation using probiotics can secrete various hydrolytic enzymes to degrade the cell wall components and increase the available contents of bioactive substances in the fermented herbs. This facilitates the release of the natural active substances from the herbs and thereby increases the extraction rate of the active ingredients (Li et al., 2020; Zhang et al., 2022). With these considerations in mind, it is clear that the appropriate combination of probiotics with the SSF protocol can enhance the availability of bioactive substances through the enzymatic purification of herbal medicines. However, more specific studies related to the fermentation mechanisms of such microorganisms and their effects on the quality characteristics of the herbal medicines are required in the future.
Conclusion
The application of probiotics in microbial fermentation via SSF is receiving growing research attention due to their potential benefits in improving the functionalities of natural herbal medicines. The present study investigated the effects of different fermentation times and temperatures on the antioxidant activities of Dunggulle (Polygonatum odoratum) root fermentations performed using various probiotic lactic acid bacteria (i.e., L. rhamnosus, L. acidophilus, L. reuteri, L. plantarum, and L. buchneri). As a result, it was found that L. buchneri exhibited the highest degree of bacterial growth (p < 0.05), and so was employed for the SSF of Dunggulle at temperatures of 25, 30, and 35°C over an 8 d culture period to observe the extent of bacterial growth and bioactive substance production. Consequently, L. buchneri demonstrated a growth peak on day 4 of incubation at 30°C, at 25 and 35°C, growth peaks were observed on day 6 (p < 0.05). It was therefore evident that L. buchneri exhibited a greater degree of proliferation at 30°C than at the other temperatures examined herein (p < 0.05). Furthermore, compared to the dried herb before fermentation, the antioxidant activity of the Dunggulle SSF product was enhanced following SSF at all investigated temperatures. More specifically, at a temperature of 30°C, the DPPH radical scavenging activity increased by 165% on day 6, the ABTS radical scavenging activity increased by 120% on day 4, the total phenolic content increased by 117% on days 4 and 6, and the total flavonoid content increased by 118% on day 2. These results demonstrate that SSF performed using L. buchneri clearly enhances the antioxidant function of natural Dunggulle, thereby providing a basis for the utilization of probiotic-based SSF technologies to improve the quality characteristics and physiological functions of herbal medicines.