Introduction
Materials and Methods
Materials
Sample Preparation
Sample Extraction
Color Measurements
Moisture Content and pH Measurements
Measurement of the Total Anthocyanin Content
Measurement of the Electron Donating Ability Using the DPPH Radical Assay
Total Polyphenol Content
Total Flavonoid Content
Evaluation of the ABTS Radical Scavenging Activity
Statistical Analysis
Results and Discussion
Color Evaluation of the Roasted Seoritae
Changes in the Moisture Content and pH
Total Anthocyanin Content
Total Polyphenol and Total Flavonoid Contents
Measurement of the DPPH Radical Scavenging Activity
ABTS Radical Scavenging Activity
Conclusion
Introduction
With the recent Westernization of diets and the rising consumption of animal-based foods, demand has increased for natural, health-promoting foods that can mitigate various chronic degenerative diseases. Foods exhibiting desirable functional properties are also in high demand. Specifically, bioactive functional substances derived from natural foods have received particular research interest. For example, the antioxidants present in natural foods act by minimizing the loss of specific vitamins and essential amino acids by reacting with free radicals, while also delaying and preventing the rancidity of dairy products (Lee et al., 2005). Additionally, beans contain nutrients such as proteins and vitamins, which are lacking in white rice. Research into the ability of beans to help prevent adult diseases, such as hypertension or arteriosclerosis, has boosted interest in their value as nutritional and healthy foods (Kim and Cho, 2005). Beans are typically consumed after boiling, steaming, and roasting, which can increase the abundance of bioactive compounds and enhance the aroma, taste, and texture (Chau and Cheung, 1997). However, since excessive heat can cause the breakdown of amino acids and other compounds, it is essential to determine the appropriate heat treatment range to optimize each specific component. For example, roasting is used to extract the inherent flavors and colors of coffee, cocoa, and barley tea. During the roasting process, the soluble solids content increases, and brown pigments and aromatic compounds are also produced (Kim et al., 2014; Suh and Chun, 1981). Browning compounds generated through amino-carbonyl reactions during roasting have been found to exhibit high antioxidant activity, thereby inhibiting lipid rancidity (McClure et al., 2022). Beans are broadly classified by color into white, black, and green varieties. Notably, Seoritae, which is harvested after being exposed to frost (seori), has a black outer shell but a green interior. Black foods have been shown to exhibit high antioxidant activity, aid blood circulation, and be highly effective for promoting kidney function (Cha, 2012). Seoritae is a representative ingredient of black foods, along with black rice and black sesame seeds. Due to the fact that Seoritae possesses a richer and more savory flavor than soybeans, it is commonly added to rice, porridge, and rice cakes (Cho et al., 2015). According to the Rural Development Administration’s National Standard Food Composition Table (RDA, 2021), Seoritae has a protein content of 38.68%, which is higher than those of soybeans (36.21%), peas (25.37%), black soybeans (36.11%), and small black beans (37.32%). Seoritae also contains high levels of bioactive substances, including amino acids such as lysine, glutamic acid, and aspartic acid (Im et al., 2016). Furthermore, it has been reported that Seoritae contains approximately twice as many phenolic compounds and flavonoids as soybeans, as well as high contents of functional components, such anthocyanins and isoflavones (Zhang et al., 2024). Due to these excellent characteristics, Seoritae is added to various products, including macarons (Kim et al., 2020), scones (Moon et al., 2022), and bean paste (Song et al., 2020) in the form of a powder or extract. With these considerations in mind, the aim of the current study is to identify the functional components and antioxidant activity of Seoritae cultivated in Hongcheon as foundational data for its utilization as a functional food ingredient, and to establish its optimal roasting conditions based on the roasting temperature and time.
Materials and Methods
Materials
Seoritae produced in 2024 was purchased (Hongcheon, Gwangwon-do, Korea). Foreign substances were removed, and the beans were sorted before use. Only premium grade reagents were used.
Sample Preparation
Seoritae was soaked in water for 1 h, drained, and then roasted on trays (200 g batches) in a preheated convection oven (RJCV-6, Rajin, Gyeonggi-do, Korea) at the desired temperature (120, 130, 140, 150, or 160°C) and time (5, 10, 15, or 20 min). After subsequent cooling, the samples were pulverized using a grinder (IKA M20, IKA, Staufen, Germany) to produce powdered samples for use as experimental materials. These powders were stored in sealed glass bottles in a cool, dark place until required for use.
Sample Extraction
Ethanol (70%) was added to the prepared Seoritae powder (50 g) in a flask, mixed thoroughly, then extracted under reflux for 4 h. The extract was filtered through Whatman No. 2 filter paper (Whatman International, Kent, UK) and the filtrate was concentrated at reduced pressure (EN-1000, Tokyo Rikakikai Co., Ltd., Tokyo, Japan). After subsequent freezing at -70°C, the sample was freeze-dried (Labconco Co., Kansas City, MO, USA) to obtain a powder, which was employed for bioactivity evaluations.
Color Measurements
Chroma measurements were performed using a chroma meter (CR-200b, Minolta Co., Tokyo, Japan). The average values of L (brightness), a (redness), and b (yellowness) were obtained by measuring each sample in triplicate. A standard white plate was used after correction to L, a, and b values of 98.34, 0.28, and 2.67, respectively.
Moisture Content and pH Measurements
The moisture content of the roasted Seoritae powder was determine using a 1 g sample and applying the 105°C oven-drying method at atmospheric pressure following AOAC guidelines (AOAC, 2019). For pH measurements, the sample (5 g) was mixed with distilled water (45 mL). The resulting mixture was centrifuged at 10,000 g and 4°C for 10 min (Supra 22K, Hanil Scientific Inc., Kimpo, Korea), and the pH of the supernatant was measured (PHM 210, Radiometer Analytical, Lyon, France).
Measurement of the Total Anthocyanin Content
The total anthocyanin content of the roasted Seoritae powder extract was calculated by measuring the absorbance of the reaction solution at maximum absorption wavelengths of 510 and 700 nm (DU 730, Beckman Coulter, Brea, CA, USA). For this purpose, 0.025 M potassium chloride buffer (pH 1.0, 830 µL) and 0.4 M sodium acetate buffer (pH 4.5, 830 µL) were added to an ethanol extract of the sample (170 µL) (Giusti and Wrolstad, 2001).
Measurement of the Electron Donating Ability Using the DPPH Radical Assay
The electron donating abilities (EDA) of the extracts were evaluated by measuring their reducing power using the DPPH (α,α-diphenyl-picrylhydrazyl) radical scavenging assay. To a test tube containing ethanol (1 mL), were added the sample (10 µL), 100 mM sodium acetate buffer (pH 5.5, 990 µL), and 0.5 mM DPPH solution (absolute EtOH solution, 0.5 mL). The resulting mixture was stirred and allowed to react in the dark for 5 min, after which time the DPPH radical concentration was measured using an ultraviolet (UV) spectrophotometer at 517 nm (Lee and Lee, 2008). The EDA (%) was expressed as [(1 - As / Ac) × 100], calculated by substituting the absorbance values of the experimental group and the control group into As and Ac, respectively.
Total Polyphenol Content
The total polyphenol content was measured based on the reduction of the Folin-Ciocalteu reagent to a molybdenum blue complex by the phenolic compounds present in the sample (Duval and Shetty, 2001). To each sample (1 mL) was added 10% Folin-Ciocalteu reagent (1 mL) and a 2% aqueous Na2CO3 solution (1 mL). The resulting solution was mixed and allowed to stand at room temperature for 1 h prior to measuring the absorbance of the supernatant at 750 nm using a microplate reader (Molecular Devices, Sunnyvale, CA, USA). Gallic acid was used as a standard, and the total polyphenol content was calculated from the standard calibration curve (y = 16.785x - 0.0343, R2 = 0.9992).
Total Flavonoid Content
The total flavonoid content was determined using a modified version of the method described by Moreno et al. (2000). To each sample (0.5 mL) was added 10% aluminum nitrate (0.1 mL), 1 M potassium acetate (0.1 mL), and ethanol (4.3 mL) in sequence. After mixing, the solution was allowed to rest at room temperature for 40 min prior to measuring the absorbance at 415 nm. The total flavonoid content of the extract was calculated from the standard calibration curve (y = 3.0124x - 0.052, R2 = 0.9983) obtained using a rutin standard (Sigma Co., USA) within a concentration range of 0-100 µg/mL.
Evaluation of the ABTS Radical Scavenging Activity
The ABTS radical scavenging activity was measured by reacting an ethanol extract of the sample (20 µL) with a 7 mM ABTS solution and a 2.45 mM potassium persulfate solution for 16 h in the dark to promote radical generation. After this time, an aliquot (10 µL) of the sample was added to a 96-well microplate with the ABTS solution (290 µL), mixed, and reacted for 6 min in the dark. The absorbance was then measured at 734 nm to calculate the radical scavenging activity (Re et al., 1999) according to the following equation:
Statistical Analysis
The results obtained from repeated measurements (at least in triplicate) were analyzed using one-way ANOVA with SPSS (Statistical Package for the Social Sciences, Version 26.0, IBM-SPSS Inc., Chicago, IL, USA). The significance between each measured mean was verified at the p < 0.05 level using Duncan’s multiple range test.
Results and Discussion
Color Evaluation of the Roasted Seoritae
The color changes resulting from roasting of the Seoritae powder are detailed in Table 1. It can be seen that Seoritae roasted at 120°C showed no significant changes in brightness, redness, or yellowness over time. However, for the samples processed at temperatures > 130°C, increased temperatures and processing times led to reduced brightness but increased redness and yellowness. It has been reported that the dark coloration of foods subjected to high-temperature treatment originates primarily from non-enzymatic browning reactions (Rodriguez-Amaya and Amaya-Farfan, 2024). Indeed, chickpeas and red beans also show a tendency for reduced brightness and increased redness at higher roasting temperatures (Im et al., 2016; Jogihalli et al., 2017). These changes became more pronounced with longer roasting times and higher temperatures, consistent with previous research (Im et al., 2016). Specifically, these changes have been attributed to the browning compounds formed through the non-enzymatic Maillard reaction between proteins (amino groups) and carbohydrates (carbonyl groups) within the samples. These compounds were mainly responsible for the increased yellowness. Anthocyanins, which are abundantly present in the hulls of Seoritae, are known to possess high antioxidant activity. As previously, reported, the heat-induced decomposition of anthocyanins generates brown substances through polymerization reactions (Kim et al., 2020). Thus, the color changes observed in Seoritae during roasting were thought to result from the Maillard reaction and the thermal degradation of anthocyanins.
Table 1
Effects of different roasting temperatures and times on the color values of Seoritae
| Conditions1) | Color value | |||
| Roasting temperature (°C) | Roasting time (min) | L3) | a4) | b5) |
| 0 | 0 | 74.32 ± 0.21a2) | -7.26 ± 0.28m | 15.32 ± 0.87f |
| 120 | 5 | 74.02 ± 0.26ab | -6.14 ± 0.36l | 17.36 ± 0.74e |
| 130 | 73.87 ± 0.24b | -5.41 ± 0.26k | 19.98 ± 0.45e | |
| 140 | 70.21 ± 0.71c | 0.14 ± 0.77g | 21.02 ± 0.46d | |
| 150 | 66.32 ± 0.64 | 0.98 ± 0.74f | 22.31 ± 0.66c | |
| 160 | 65.74 ± 0.31 | 2.64 ± 0.44d | 22.98 ± 0.76c | |
| 120 | 10 | 73.97 ± 0.11b | -6.01 ± 0.42l | 18.36 ± 0.53 |
| 130 | 70.22 ± 0.84c | -5.06 ± 0.32k | 20.14 ± 0.26d | |
| 140 | 69.22 ± 0.41c | 0.98 ± 0.81f | 22.36 ± 0.57c | |
| 150 | 68.65 ± 0.33d | 1.87 ± 0.53e | 23.24 ± 0.41b | |
| 160 | 67.21 ± 0.41e | 3.84 ± 0.34c | 23.89 ± 0.61b | |
| 120 | 15 | 73.64 ± 0.25b | -5.47 ± 0.12 | 18.00 ± 0.27 |
| 130 | 69.26 ± 0.43cd | -3.25 ± 0.26j | 20.89 ± 0.51d | |
| 140 | 65.32 ± 0.29f | 2.59 ± 0.72d | 22.96 ± 0.22c | |
| 150 | 64.95 ± 0.38g | 3.65 ± 0.74c | 23.75 ± 0.22b | |
| 160 | 61.02 ± 0.45i | 5.94 ± 0.26b | 24.02 ± 0.55a | |
| 120 | 20 | 73.55 ± 0.77b | -2.01 ± 0.34i | 19.22 ± 0.41 |
| 130 | 67.21 ± 0.45e | -0.66 ± 0.28h | 21.65 ± 0.33d | |
| 140 | 64.25 ± 0.29h | 3.62 ± 0.29c | 23.76 ± 0.12b | |
| 150 | 64.02 ± 0.47h | 4.25 ± 0.64c | 23.99 ± 0.72ab | |
| 160 | 59.21 ± 0.32j | 7.21 ± 0.21a | 24.41 ± 0.23a | |
Changes in the Moisture Content and pH
The moisture content and pH changes observed for the roasted Seoritae are detailed in Table 2. The highest pH (6.88) was observed in the group treated at 120°C for 5 min, and this value decreased significantly as the roasting temperature and time were increased (p < 0.05). This decrease in pH aligns with previous research (Moon et al., 2011), and was attributed to binding of the soybean protein amino groups with sugars, which leads to the breakdown of soluble amino acids and the production of organic acids, such as citric acid. The lowest moisture content was observed following processing at 160°C for 20 min, indicating that this parameter decreased with increasing roasting temperature and time (p < 0.05). As reported previously (Poojary and Lund, 2022), sugars and proteins present in the beans react with non-reducing sugars through a dehydration-condensation reaction upon heating, thereby reducing the moisture content. Similar results were reported by Hwang and Moon (2022), showing that tissue porosity increases, density reductions, and physicochemical changes (through lipid oxidation and the decomposition of phenolic compounds) contribute to reduced moisture content.
Table 2
Effects of different roasting temperatures and times on the moisture content and pH of Seoritae
| Conditions1) | pH | Moisture content (%) | |
| Roasting temperature (°C) | Roasting time (min) | ||
| 0 | 0 | 6.89 ± 0.02a2) | 8.97 ± 0.14a |
| 120 | 5 | 6.88 ± 0.02a | 7.88 ± 0.02b |
| 130 | 6.87 ± 0.01a | 7.86 ± 0.02c | |
| 140 | 6.62 ± 0.02a | 7.72 ± 0.01d | |
| 150 | 6.67 ± 0.02a | 7.50 ± 0.01e | |
| 160 | 6.58 ± 0.01a | 6.88 ± 0.01f | |
| 120 | 10 | 6.85 ± 0.01a | 7.70 ± 0.02d |
| 130 | 6.76 ± 0.02b | 7.40 ± 0.02e | |
| 140 | 6.61 ± 0.01d | 7.01 ± 0.01f | |
| 150 | 6.52 ± 0.02a | 6.84 ± 0.01g | |
| 160 | 6.50 ± 0.01a | 6.64 ± 0.01h | |
| 120 | 15 | 6.78 ± 0.02b | 6.58 ± 0.01i |
| 130 | 6.74 ± 0.01bc | 5.64 ± 0.01k | |
| 140 | 6.63 ± 0.01d | 4.29 ± 0.01m | |
| 150 | 6.53 ± 0.01e | 3.79 ± 0.01o | |
| 160 | 6.48 ± 0.01a | 3.21 ± 0.01p | |
| 120 | 20 | 6.63 ± 0.02d | 6.02 ± 0.02j |
| 130 | 6.55 ± 0.01e | 5.99 ± 0.01j | |
| 140 | 6.49 ± 0.01f | 4.88 ± 0.02l | |
| 150 | 6.40 ± 0.02g | 3.98 ± 0.01n | |
| 160 | 6.35 ± 0.01h | 0.94 ± 0.02q | |
Total Anthocyanin Content
Anthocyanins, which are widely distributed in the plant kingdom, are water-soluble flavonoid pigments exhibiting purple, red, and blue hues. They are known to possess antioxidant activity and exert anticancer, anti-inflammatory, and vision-protective effects (Yi and Kim, 2010). Katsuzaki et al. (2003) reported that the anthocyanins present in black beans are primarily represented by delphinidin-3-glucoside, cyanidin-3-glucoside, and petunidin-3-glucoside, which are abundantly contained in the seed coat. The total anthocyanin contents of the roasted Seoritae powder extracts are detailed in Table 3, showing that the untreated group exhibited the highest anthocyanin content (0.75 mg/g). However, upon increasing the roasting time and temperature, the anthocyanin content decreased significantly, ranging from 0.24 to 0.70 mg/g (p < 0.05).
Table 3
Effects of different roasting temperatures and times on the total anthocyanin content in Seoritae
| Conditions1) | Total anthocyanin content(mg/g) | |
| Roasting temperature (°C) | Roasting time (min) | |
| 0 | 0 | 0.75 ± 0.01a2) |
| 120 | 5 | 0.70 ± 0.00b |
| 130 | 0.68 ± 0.01c | |
| 140 | 0.68 ± 0.01c | |
| 150 | 0.65 ± 0.00d | |
| 160 | 0.62 ± 0.01e | |
| 120 | 10 | 0.68 ± 0.01c |
| 130 | 0.67 ± 0.02cd | |
| 140 | 0.66 ± 0.01d | |
| 150 | 0.65 ± 0.01d | |
| 160 | 0.50 ± 0.01f | |
| 120 | 15 | 0.66 ± 0.01d |
| 130 | 0.65 ± 0.01d | |
| 140 | 0.54 ± 0.01g | |
| 150 | 0.51 ± 0.01h | |
| 160 | 0.40 ± 0.01j | |
| 120 | 20 | 0.55 ± 0.02g |
| 130 | 0.51 ± 0.01h | |
| 140 | 0.49 ± 0.01i | |
| 150 | 0.40 ± 0.02j | |
| 160 | 0.24 ± 0.01k | |
Total Polyphenol and Total Flavonoid Contents
Polyphenolic compounds, which are secondary metabolites found in natural plants, possess antioxidant properties and exert protective effects against cancer, bacterial infections, diabetes, and obesity (Sinha, 2019). The total polyphenol and total flavonoid contents of the roasted Seoritae powder samples were therefore analyzed, as detailed in Table 4. The untreated Seoritae exhibited the highest total polyphenol content (39.66 mg gallic acid equivalents per gram, GAE/g), and the roasting conditions were found to have no effect on the change in total polyphenol content. The total flavonoid content of the untreated Seoritae was determined to be 32.14 mg rutin equivalents per gram (RE/g). Although the total flavonoid content decreased slightly as the roasting temperature and time were increased, no significant differences were observed. These results differ from those reported by Jeong et al. (2016), who found that roasted mung beans exhibited higher total flavonoid content than raw mung beans. Moreover, the changes in the total polyphenol and total flavonoid contents resulting from Seoritae roasting showed significant increases compared with the results obtained for raw mung beans (Kim et al., 2014). This was attributed to the decomposition of phenolic compounds under the roasting conditions employed herein.
Table 4
Effects of different roasting temperatures and times on the total polyphenol and total flavonoid contents in Seoritae
| Conditions1) |
Total polyphenol (mg GAE/g) |
Total flavonoids (mg RE/g) | |
| Roasting temperature (°C) | Roasting time (min) | ||
| 0 | 0 | 39.66 ± 2.31NS2) | 32.14 ± 1.18 |
| 120 | 5 | 40.21 ± 1.32 | 33.08 ± 2.74 |
| 130 | 40.56 ± 1.26 | 33.21 ± 2.08 | |
| 140 | 40.32 ± 0.98 | 32.97 ± 1.54 | |
| 150 | 41.01 ± 1.23 | 32.92 ± 1.23 | |
| 160 | -0.0028 ± 0.0001 | 32.87 ± 2.36 | |
| 120 | 10 | 40.91 ± 0.88 | 33.00 ± 1.74 |
| 130 | 40.88 ± 1.84 | 32.88 ± 2.36 | |
| 140 | 41.72 ± 1.69 | 32.75 ± 1.98 | |
| 150 | 41.01 ± 1.23 | 32.01 ± 1.87 | |
| 160 | 40.98 ± 0.69 | 31.99 ± 1.69 | |
| 120 | 15 | 40.68 ± 0.97 | 32.99 ± 1.62 |
| 130 | 40.99 ± 1.64 | 32.81 ± 1.37 | |
| 140 | 40.83 ± 0.97 | 32.28 ± 1.36 | |
| 150 | 40.90 ± 1.06 | 31.96 ± 1.88 | |
| 160 | 40.82 ± 1.29 | 31.61 ± 1.67 | |
| 120 | 20 | 40.99 ± 1.38 | 32.01 ± 2.36 |
| 130 | 40.00 ± 1.52 | 31.94 ± 0.99 | |
| 140 | 39.93 ± 1.22 | 31.87 ± 1.39 | |
| 150 | 40.01 ± 1.26 | 31.64 ± 1.17 | |
| 160 | 39.98 ± 1.89 | 31.55 ± 1.08 | |
Measurement of the DPPH Radical Scavenging Activity
The DPPH radical scavenging activity is a measure of antioxidant activity, and is based on the ability of antioxidant compounds to inhibit oxidation by donating hydrogen atoms to DPPH radicals (Yen and Chen, 1995). It can also be used as an indicator of the potential to delay free radical-induced aging in the human body (Torel et al., 1986). As detailed in Fig. 1, the untreated group showed the lowest DPPH radical scavenging activity of 30.32%. Upon increasing the roasting temperature and time, this activity also increased, reaching a maximum of 49.32% following processing at 160°C for 20 min. These results are consistent with a previous report (Koriyama et al., 2022) indicating that the radical scavenging activity increases with higher roasting temperatures across different types of beans.
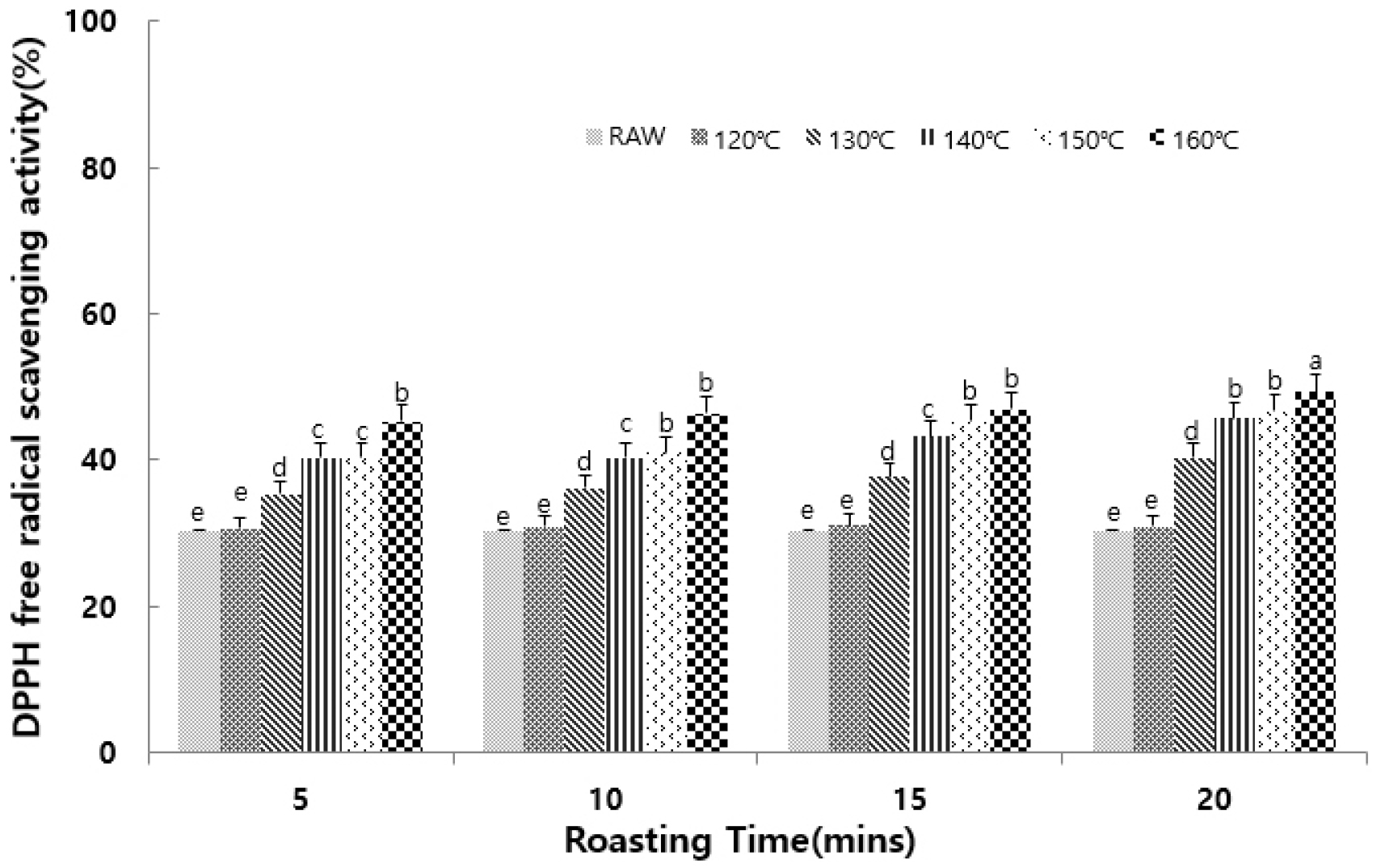
Fig. 1.
Effects of different roasting conditions on the DPPH radical scavenging activity of Seoritae. Results are presented as the mean ± SD of 3 independent experiments performed in triplicate. Means with different letters (a–e) on the same bar are significantly different at p < 0.05, according to Duncan’s multiple range test.
ABTS Radical Scavenging Activity
The ABTS cation radical assay represents an effective method for assessing the antioxidant activity of both hydrophilic and lipophilic substances, and is based on the decolorization of its characteristic blue-green color through radical cation quenching (Miller et al., 1993). As shown in Fig. 2, the lowest ABTS radical scavenging activity was observed for the control group (35.21%), showing an increasing trend at higher roasting temperatures. Consequently, the Seoritae sample roasted at 160°C for 20 min exhibited the highest activity of 73.2%, which is significantly different to that observed for small black soybeans roasted at 120°C for 20 min (Lee et al., 2014).
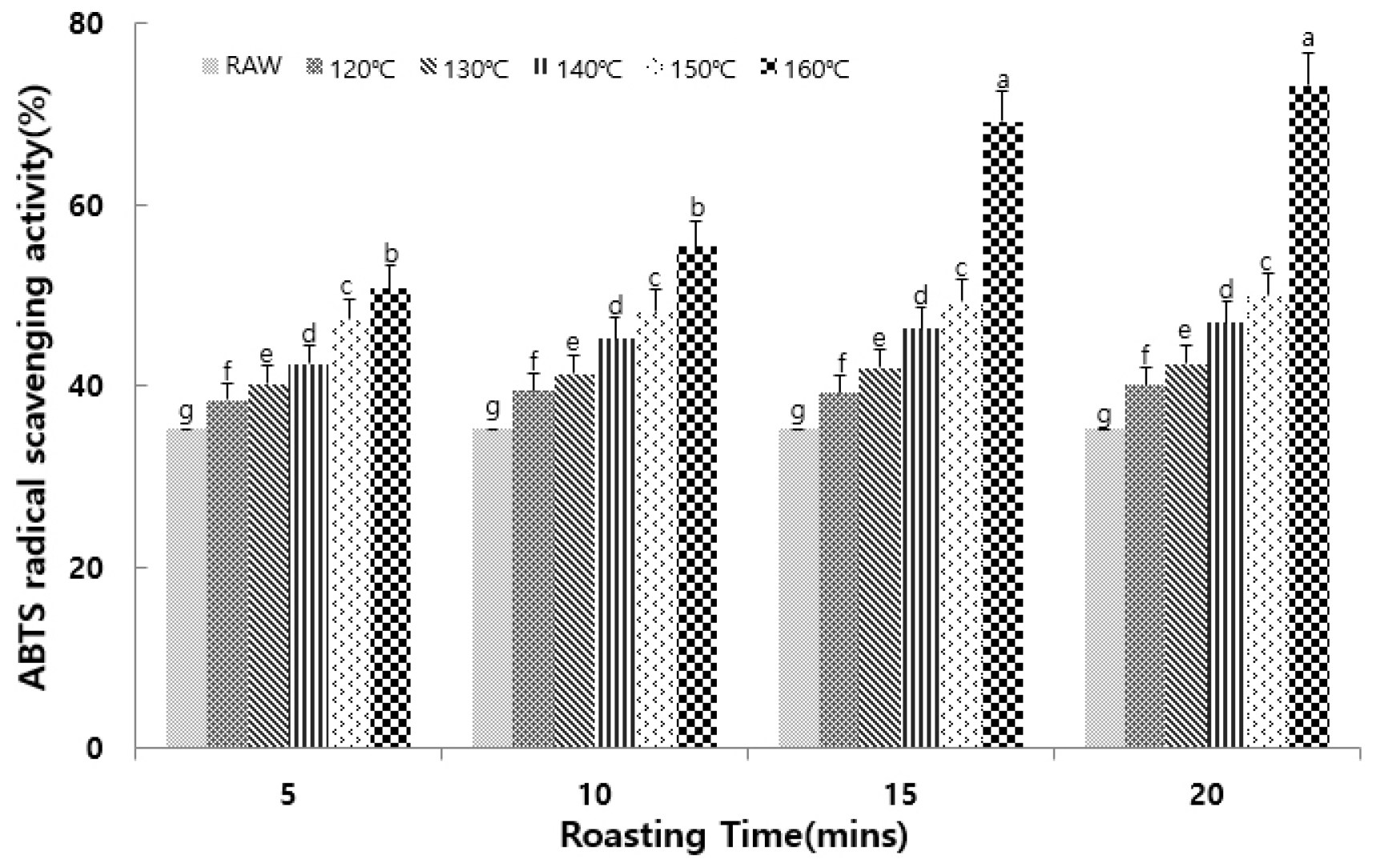
Fig. 2.
Effects of different roasting conditions on the ABTS radical scavenging activity of Seoritae. Results are presented as the mean ± SD of 3 independent experiments performed in triplicate. Means with different letters (a–e) on the same bar are significantly different at p < 0.05, according to Duncan’s multiple range test.
Conclusion
This study was conducted to establish foundational data for manufacturing processed products using Seoritae from Hongcheon County, Korea. The beans were roasted at varying temperatures (120, 130, 140, 150, and 160°C) and roasting times (5, 10, 15, and 20 min) to measure the characteristics and antioxidant activity of Seoritae, and to conduct a quality evaluation. It was found that the brightness of the Seoritae powder decreased as the roasting temperature and time were increased, while redness and yellowness increased. Additionally, the moisture content and pH decreased significantly with increasing roasting temperature and time. The highest total anthocyanin content was observed for the untreated Seoritae (0.75 mg/g), with a significant decreasing trend being observed (i.e., 0.24-0.70 mg/g) as the roasting temperature and time were increased. In contrast, the total polyphenol and total flavonoid contents showed no significant differences under the different roasting conditions evaluated herein. This was thought to be due to the different methods used to measure the total polyphenol and flavonoid contents, which resulted in varying values. Moreover, the antioxidant activity (DPPH free radical scavenging activity, ABTS scavenging activity) showed a tendency to increase with increasing roasting temperature and time, reaching its highest level after roasting at 160°C for 20 min. Based on these results, further research should be performed using sensory evaluations and response surface methodology to identify the optimal roasting conditions for maximizing the bioactive compound content without compromising the sensory preferences of consumers.




