Introduction
Materials and Methods
Study Area and Period
Fish Sampling and Identification
Fish Community and G. brevibarba Population Analysis
Results and Discussion
Fish Fauna and Community Structure
G. brevibarba Population Analysis
Age Distribution and Potential Growth Capability
Introduction
The Pyeongchang River is created by the convergence of two streams, the Soksa and Heungjeong Streams, which flow from the Gyebangsan and Heungjeongsan Mountains in Pyeongchang-gun, Gangwon-do, respectively. This river has a length of 198.25 km and joins the Namhangang River at Hasong-ri, Yeongwol-eup (ME, 2024; MOLIT, 2013). The portion of the river that meets the Jucheon River at the Hanbando Wetland and then flows into the Namhangang is also referred to as the “Seogang River” (Kim, 2015). The river is in a natural, unspoiled state, and its constant high water flow creates a perfect habitat for many aquatic species. In particular, well-developed riffles, river pools, and heterogeneous substrates of boulders, cobbles, gravel, and sand are essential habitats for periphytic algae, aquatic insects, and benthic fishes.
The slender shoveljaw gudgeon (Gobiobotia brevibarba) is a freshwater fish species belonging to the subfamily Gobioninae of the family Cyprinidae. It is a Korean endemic species, mainly found in the mid- to upper Han and Geum River systems (Chae et al., 2019; Kim et al., 2005). This species exhibits a unique body form that is convex dorsally and ventrally flattened, enabling it to attach to and live in the crevices of streambed swift-flowing riffles. They generally live and move actively in streambeds composed of gravel and fine gravel (Kim, 1997) and are omnivores that feed mostly on aquatic insect larvae, including those of midges and mayflies (Choi et al., 2001b). In addition, newly hatched juveniles of G. brevibarba have poor swimming ability and exhibit hidden behavior in the gaps between gravel and fine gravel (Choi et al., 2001a). This life history illustrates the importance of particulate substrates and flow requirements in riffle habitats for the survival and reproduction of this species.
Nonetheless, the habitats of G. brevibarba are under constant threat from human disturbance, including dam construction, river maintenance, and water pollution, resulting in a decline in its population (Choi et al., 2001b). Similar patterns of habitat disturbance and population decline have been observed in other rivers, including the Seomgang (Ko et al., 2011a). As a result, the Ministry of Environment classified it as a “Specific Wildlife and Plant” in 1996 and has been safeguarding it as an Endangered Wild Animal Class II from 2005 to date (ME, 1996, 2005, 2025). Accordingly, ecological studies that enable stable maintenance and conservation of G. brevibarba population are vital and urgent.
In this context, the present study was conducted to evaluate the status of fish communities in the Seogang area of the Pyeongchang River in Yeongwol. In addition, this study aimed to investigate the ecological aspects of the class II Endangered Wild Animal species G. brevibarba, including its habitat, population (growth and age structure), and potential growth. The findings of this study provide essential scientific baseline information for effectively conserving and restoring the G. brevibarba population.
Materials and Methods
Study Area and Period
This study used five surveys conducted from September 2022 to April 2023.
∙ 1st Survey: September 2-3, 2022
∙ 2nd Survey: November 8, 2022
∙ 3rd Survey: February 23, 2023
∙ 4th Survey: March 22, 2023
∙ 5th Survey: April 2, 2023
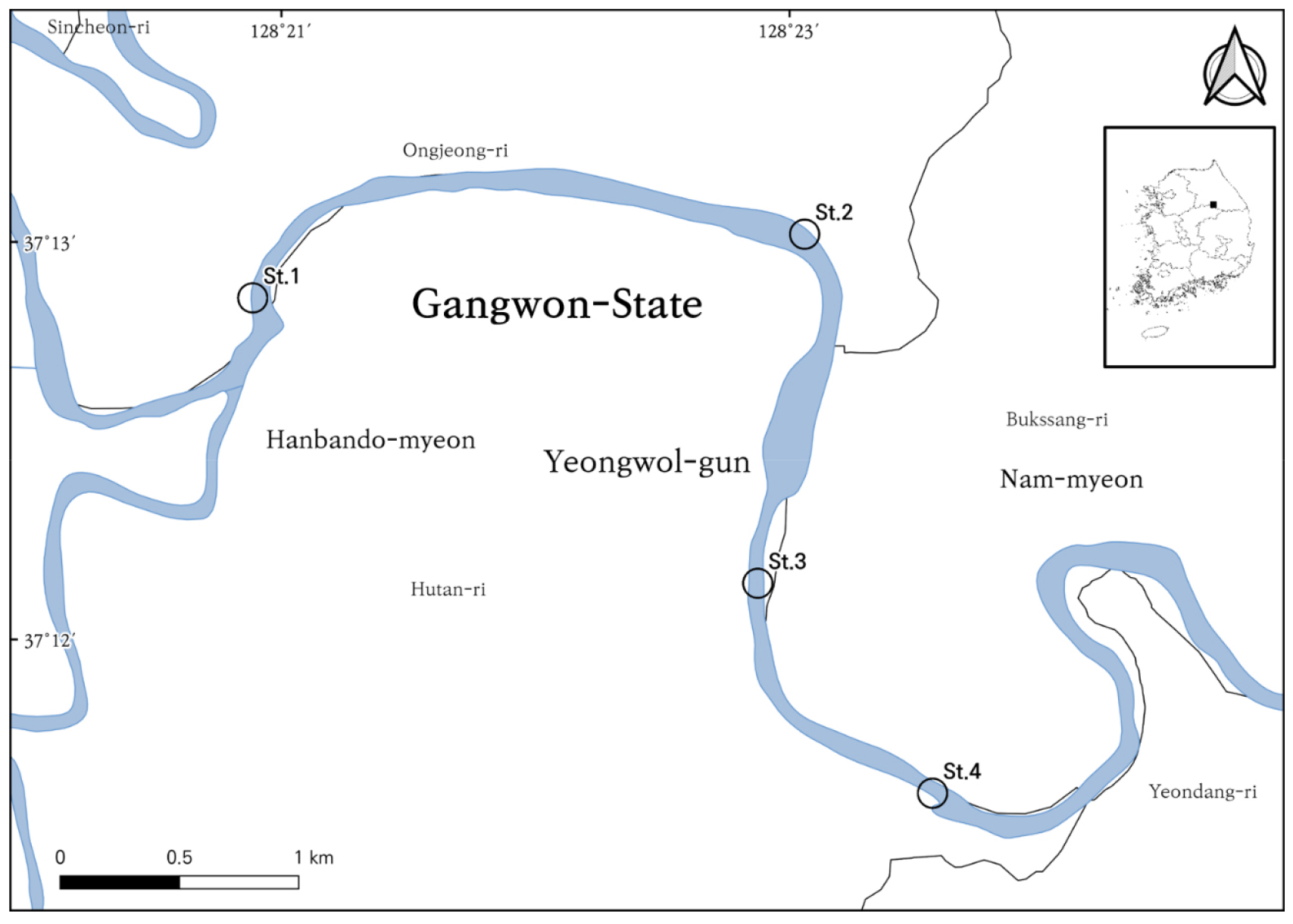
Four riffle zones have been established in the lower reaches of the Pyeongchang River in the Seogang section. The site coordinates, along with their administrative locations, are shown in Table 1 and Fig. 1.
Table 1
Address and location of each surveyed station in the Pyeongchang river, Yeongwol
Fish Sampling and Identification
Fish sampling was conducted seasonally at four sites (St. 1-4) in the Seogang section of the Pyeongchang River between May 2023 and April 2024, with a total of five surveys. At each site, a uniform sampling effort was applied using a casting net (7 × 7 mm mesh) and a scoop net (4 × 4 mm mesh). Casting nets were thrown 7 ± 3 times, and a scoop net was used for 40 ± 20 minutes at each site. This was an attempt to keep sampling relatively uniform, while allowing for variation in flow and habitat between sites.
Importantly, the endangered class II wild animal species G. brevibarba was collected with prior permission (No. 2023-04) from the Wonju Regional Environmental Office under the Wildlife Protection and Management Act (Appendix 1). In addition, the authorities granted permission to use a casting net to protect the legality and transparency of the study, which may have contravened the Inland Fisheries Act (Appendix 2).
All fish were identified to the species level in the field, photographed, released, and their abundances were recorded. Individuals that were visually indistinguishable in the field were preserved in 10% formalin and transported to the laboratory for identification under a dissecting microscope. An anesthetic (MS-222) was used to temporarily immobilize G. brevibarba immediately after capture. Their total length (TL; ± 0.1 mm) and weight (W; ± 0.01 g) were then accurately recorded. Once all measurements were taken, each fish was checked to ensure complete recovery from anesthesia before being returned to the water to reduce stress and the risk of injury.
Fish identification was performed using recent domestic fish keys and classification standards, primarily by referring to Chae et al. (2019), Choi et al. (2002), Kim (1997), Kim and Park (2002), and Kim et al. (2005). The scientific names are in accordance with the most recent National List of Species (December 31, 2022) provided by the National Institute of Biological Resources (NIBR, 2020). Species under legal protection were identified according to Appendix 1 of the Enforcement Rules of the Wildlife Protection and Management Act (partially amended on June 9, 2023) by the Ministry of Environment and the Cultural Heritage Administration’s White Paper on Natural Monuments (CHA 2003). The system of classification and organization of the fish followed the internationally recognized system of Nelson (2006).
Fish Community and G. brevibarba Population Analysis
In this study, the fish community and G. brevibarba population characteristics were analyzed using fish specimens collected from each survey site.
Fish Community Analysis
We calculated the number of species, number of individuals, and the Shannon-Weaver diversity index (Shannon and Weaver, 1963) to assess the diversity of the fish community at each site. We also analyzed the rank-abundance curve to visually represent and quantitatively evaluate the species abundance distribution patterns of each community (Whittaker, 1965; Wilson, 1991).
The RAC analysis was performed using the ‘radfit’ function within the ‘vegan’ package (Oksanen, 2011) of the R software (R Core Team, 2012). The optimal model was selected from five different models-Null, Log-normal (Preston, 1948), Mandelbrot (Mandelbrot, 1965), Preemption (Motomura, 1932), and Zipf (Zipf, 1949)-by choosing the model with the lowest Akaike’s information criterion value. The average slope (AS) of the RAC was calculated as follows (Kim et al., 2013):
where N is the total number of species, Sn is the number of individuals of the nth ranked species, and S1 is the number of individuals of the dominant species.
G. brevibarba Population Characteristics Analysis
The following analyses were performed on the G. brevibarba populations collected from the study area. First, length-frequency analysis was performed using the method of Ricker (1971) to analyze the length-frequency distribution, thereby estimating the population size structure and tentative age groups.
For the length-weight relationship, the relationship between TL and weight (W) was derived to understand the growth pattern of the population. This relationship is a key indicator of a population’s growth rate and its adaptability to the environment.
The condition factor (K), an index of fish health and nutritional status, was calculated using the Fulton-type formula (Anderson and Neumann, 1996). The condition factor is an important indicator that provides diverse ecological information, including nutritional status, reproductive capacity, habitat quality, and food resource utilization ability of a population (Anderson and Gutreuter, 1983; Busacker et al., 1990; Ney, 1993).
Growth Model Analysis
Based on the tentative age groups estimated from the length-frequency analysis, the von Bertalanffy growth model (von Bertalanffy, 1938) was applied to determine the potential growth capability of the population. Through this model, we estimated the Brody growth coefficient (k), which represents the population’s growth rate, and the asymptotic length (L∞), the maximum theoretical TL of the population. Specifically, L∞ was visually represented using a Walford plot (Walford, 1946).
To enhance the accuracy of the model, the theoretical TL at age zero (t0) was set to 5.5 mm, which was the actual mean TL of newly hatched G. brevibarba, as reported by Ko et al. (2011b).
Results and Discussion
Fish Fauna and Community Structure
During the survey period, 2,157 individuals across 23 species and 8 families were identified at the four study sites on the Pyeongchang River, Yeongwol (Table 2). Analysis of the composition by family revealed that fish from the family Cyprinidae comprised the highest proportion, with 13 species (56.6%), followed by Cobitidae with 3 species (13.1%) and Odontobutidae with 2 species (8.8%). Additionally, one species each from the families Siluridae, Bagridae, Amblycipitidae, Centropomidae, and Gobiidae was present (4.3% each). The dominance of Cyprinidae and Cobitidae is a common trend observed in most riverine ecosystems flowing into the western and southern coasts of the Korean Peninsula, and is attributed to the high adaptability of these species to a wide range of habitats and food sources (Jeon, 1980).
Table 2
A list and individual number of fishes collected at the Pyeongchang river, Yeongwol from September 2022 to April 2023
| Scientific name | Station | Total | R.A. (%) | Remarks | ||||
| 1 | 2 | 3 | 4 | |||||
| Cyprinidae | ||||||||
| Acheilognathus signifer | 75 | 69 | 39 | 67 | 250 | 11.6 | E II, EN | |
| Acheilognathus yamatsutae | 60 | 43 | 12 | 14 | 129 | 6.0 | EN | |
| Pungtungia herzi | 9 | 113 | 98 | 142 | 362 | 16.8 | ||
| Pseudopungtungia tenuicorpa | 3 | 1 | 4 | 0.2 | E II, EN | |||
| Coreoleuciscus splendidus | 95 | 52 | 130 | 53 | 330 | 15.3 | EN | |
| Sarcocheilichthys variegatus wakiyae | 8 | 8 | 0.4 | EN | ||||
| Hemibarbus mylodon | 1 | 1 | 0.1 | E II, NM, EN | ||||
| Pseudogobio esocinus | 1 | 1 | 0.1 | |||||
| Gobiobotia macrocephala | 5 | 13 | 29 | 21 | 68 | 3.2 | E II, EN | |
| Gobiobotia brevibarba | 49 | 82 | 476 | 34 | 641 | 29.7 | E II, EN | |
| Rhynchocypris oxycephalus | 1 | 1 | 0.1 | |||||
| Zacco koreanus | 13 | 14 | 27 | 22 | 76 | 3.5 | EN | |
| Zacco platypus | 31 | 12 | 13 | 27 | 83 | 3.9 | ||
| Cobitidae | ||||||||
| Misgurnus anguillicaudatus | 1 | 1 | 0.1 | |||||
| Iksookimia koreensis | 1 | 1 | 2 | 0.1 | EN | |||
| Koreocobitis rotundicaudata | 2 | 2 | 20 | 13 | 37 | 1.7 | EN | |
| Siluridae | ||||||||
| Silurus microdorsalis | 1 | 1 | 0.1 | EN | ||||
| Bagridae | ||||||||
| Pseudobagrus koreanus | 1 | 1 | 0.1 | EN | ||||
| Amblycipitidae | ||||||||
| Liobagrus andersoni | 21 | 10 | 14 | 31 | 76 | 3.5 | EN | |
| Centropomidae | ||||||||
| Coreoperca herzi | 11 | 13 | 10 | 32 | 66 | 3.1 | EN | |
| Odontobutidae | ||||||||
| Odontobutis platycephala | 1 | 1 | 0.1 | EN | ||||
| Odontobutis interrupta | 3 | 3 | 0.1 | EN | ||||
| Gobiidae | ||||||||
| Rhinogobius brunneus | 11 | 2 | 2 | 15 | 0.7 | |||
| Number of families | 5 | 7 | 6 | 5 | 8 | |||
| Number of species | 15 | 14 | 14 | 17 | 23 | |||
| Number of individuals | 392 | 427 | 874 | 464 | 2,157 | |||
Based on the proportion of individuals, G. brevibarba was the dominant species, accounting for 29.7% (641 individuals), whereas Pungtungia herzi was the subdominant species, accounting for 16.8% (362 individuals). The next most abundant species were Coreoleuciscus splendidus (15.3%, 330 individuals), Acheilognathus signifer (11.6%, 250 individuals), Acheilognathus yamatsutae (6.0%, 129 individuals), and Zacco platypus (3.9%, 83 individuals). The fact that G. brevibarba was the dominant species suggests that the Pyeongchang River provides abundant riffle zones with fast flows and well-developed gravel and fine gravel substrates, which are preferred by this species. This indicated that the river offers suitable environmental conditions for G. brevibarba spawning and habitation. In contrast, 12 species, including Rhinogobius brunneus, Sarcocheilichthys variegatus wakiyae, Pseudopungtungia tenuicorpa, Odontobutis interrupta, and Iksookimia koreensis, were rare, with individual composition rates of less than 1.0%.
The legally protected species identified in this study included five fish species designated as Endangered Wild Animals Class II by the Ministry of Environment: A. signifer, P. tenuicorpa, Hemibarbus mylodon, G. macrocephala, and G. brevibarba. H. mylodon is a Natural Monument designated by the Cultural Heritage Administration. Among these, A. signifer, G. macrocephala, and G. brevibarba were consistently found in relatively high numbers across all survey sites, suggesting that healthy populations were maintained within the Pyeongchang River. However, P. tenuicorpa and H. mylodon appeared in very low numbers (fewer than five individuals) at only one or two sites, indicating a poor distribution area and population size. This suggests that these species may be more sensitive to specific habitat conditions or have low population densities, thus making them conservationally vulnerable. Consequently, further distribution surveys and development of conservation measures for these species are warranted.
Of the 23 fish species identified in the study area, 17 were Korean endemic species: A. signifer, A. yamatsutae, P. tenuicorpa, C. splendidus, S. variegatus wakiyae, H. mylodon, G. macrocephala, G. brevibarba, Zacco koreanus, I. koreensis, Koreocobitis rotundicaudata, Silurus microdorsalis, Pseudobagrus koreanus, Liobagrus andersoni, Coreoperca herzi, Odontobutis platycephala, and O. interrupta. This represents a very high endemic species ratio of 74.0%, which is more than 2.5 times higher than the Korean average endemism rate of 28.8% (Kim et al., 2005). This high proportion of endemic species clearly indicates that the Pyeongchang River has minimal habitat disturbance, excellent aquatic environment preservation, and high ecological value in terms of fish diversity. This can be interpreted as a key indicator of the well-maintained naturalness of a river.
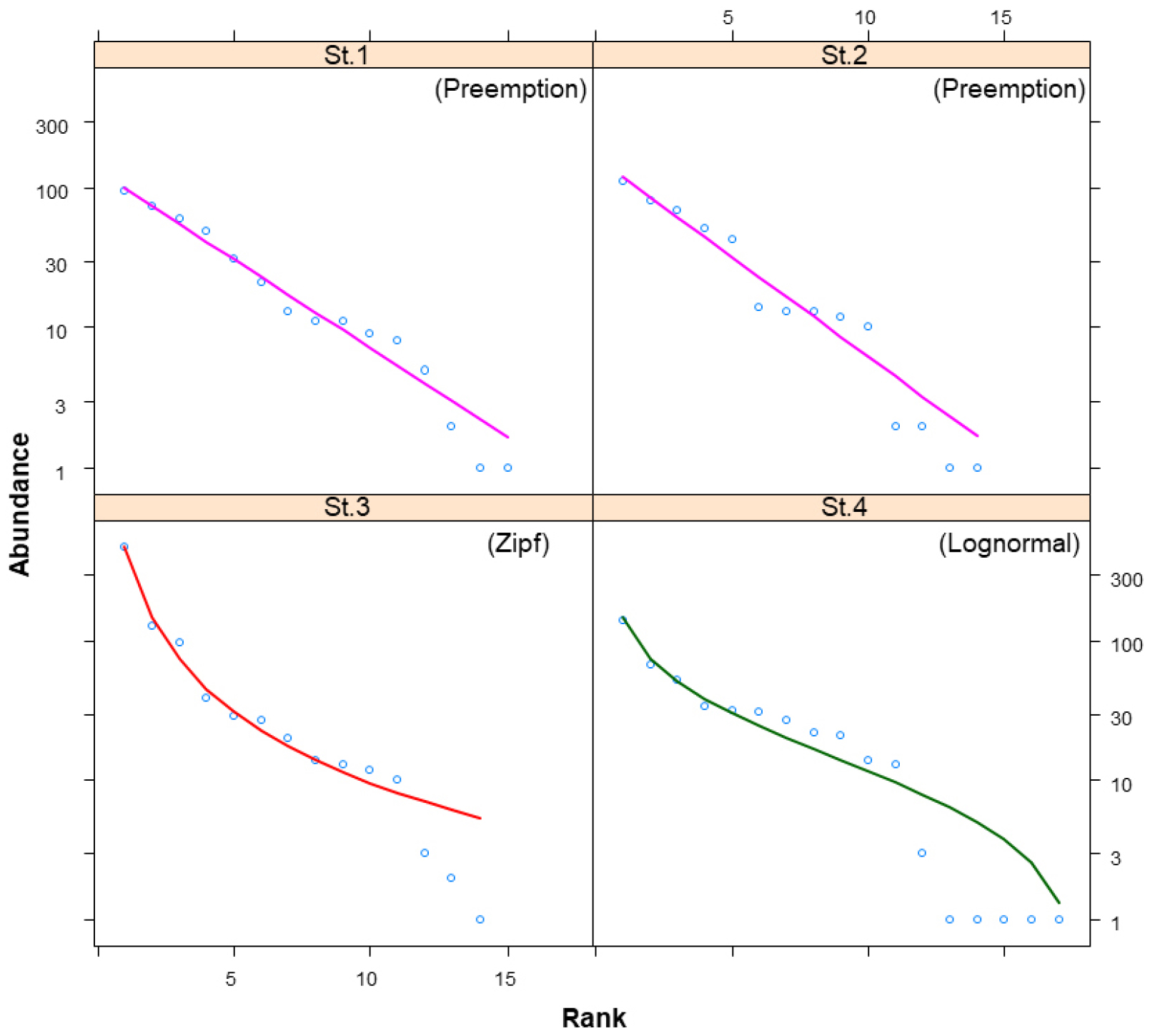
The AIC values used to determine the model fit of the RAC for each survey site, along with the derived RACs, diversity indices, and average slopes of the curves, are presented in Table 3 and Fig. 2. The RAC is an effective method for simultaneously representing richness and evenness, which are the two core components of species diversity (Magurran, 2004; Park, 2021; Whittaker, 1965). Analysis of the RAC models for fish communities at each site showed that the Preemption model was the best fit for St.1 and St.2, the Zipf model for St.3, and the Log-normal model for St.4.
Table 3
Akaike’s information criterion (AIC) values of ranked abundance curve for each model. The best model was selected by the lowest AIC value (H′: diversity index, AS: average slope)
∙ Preemption model (St.1, St.2): This model typically shows a linear distribution, reflecting a pattern in which the influence of a particular dominant species is very strong in communities exposed to extreme environmental conditions or disturbances. This often results in a relatively low species diversity or the dominance of a few species (Whittaker, 1965; Wilson, 1991). Despite the relatively high number of species found at St.1 (15 species) and St.2 (14 species), the appearance of the Preemption model suggests that either the proportion of a specific dominant species was overwhelmingly high (e.g., the high dominance of G. brevibarba) or that microhabitat heterogeneity or localized disturbance factors were at play.
∙ Zipf model (St.3): The Zipf model is suitable for describing a phenomenon in which, despite the presence of various species, a small number of species are overwhelmingly dominant within the community (Wilson et al., 1996). At St.3, a total of 14 species were found, but the fact that the dominant and subdominant species accounted for approximately 70% of the entire community suggests that evenness among the species within the community was relatively low. This raises the possibility that certain environmental conditions are particularly favorable for a small number of species.
∙ Log-normal model (St.4): This model signifies a community structure in which species are neither overwhelmingly dominant nor subordinate to competition. Instead, they maintain a balance between species richness and abundance, resulting in stable communities (Wilson et al., 1996). The fact that the fish community at St.4 fitted the Log-normal model suggests that it has a lower dominance of specific species and fewer external environmental disturbances than the other three sites, indicating a relatively stable community state.
The Shannon-Weaver diversity index at the sites ranged from 1.59 to 2.20, with the lowest value at St.3 (1.59) and the highest at St.4 (2.20). The overall average diversity index was 2.00, indicating that the Pyeongchang River fish community was in generally favorable conditions. The AS of the RAC was also relatively gentle, ranging from 0.07 to 0.12, suggesting that the richness and evenness of the fish community were well maintained.
G. brevibarba Population Analysis
Population Size Distribution and Site-Specific Characteristics
To compare the total length and weight distribution of G. brevibarba populations among sites, the TL range was calculated using percentiles and visualized as a box plot (Fig. 3). The analysis showed that the largest individuals were found at St.1, with a median TL of 85.0 ± 15.0 mm. The median TLs were 82.0 ± 19.4 mm at St.2, 74.0 ± 17.3 mm at St.3, and 51.0 ± 20.7 mm at St.4, showing a significant decreasing trend in median TL toward the downstream sites. Weight showed a similar pattern to TL, with the median weight being highest at St.1 (5.4 ± 2.8 g), followed by St.2 (4.8 ± 3.7 g), St.3 (3.4 ± 2.8 g), and St.4 (0.9 ± 3.4 g).
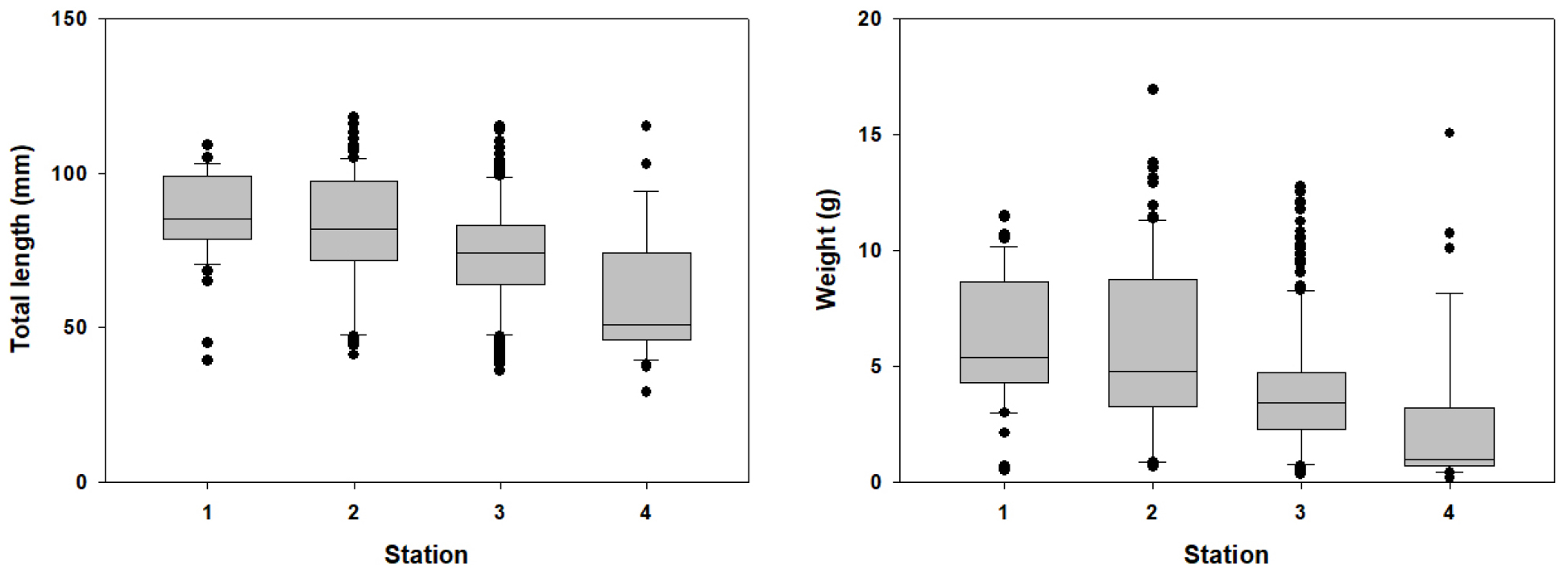
Fig. 3.
Total length and weight of the G. brevibarba population in the surveyed stations. The boundary of the box closest to zero indicates the 25th percentile, a line within the box marks the median, and the boundary of the box farthest from zero indicates the 75th percentile. Whiskers (error bars) above and below the box indicate the 90th and 10th percentiles (Chambers et al., 1983).
The differences in individual size distribution among the sites suggest a close relationship with the riverine environment in which the G. brevibarba population resides. Specifically, the environmental characteristics of decreasing flow velocity and streambed particle size towards downstream sections likely led to the formation of microhabitats with abundant food sources and suitable refugia for juveniles. This can be interpreted as the result of juveniles tending to concentrate in relatively stable downstream areas rather than in the upper reaches, which are characterized by strong currents and large streambed structures.
Growth and Condition Factor Analysis
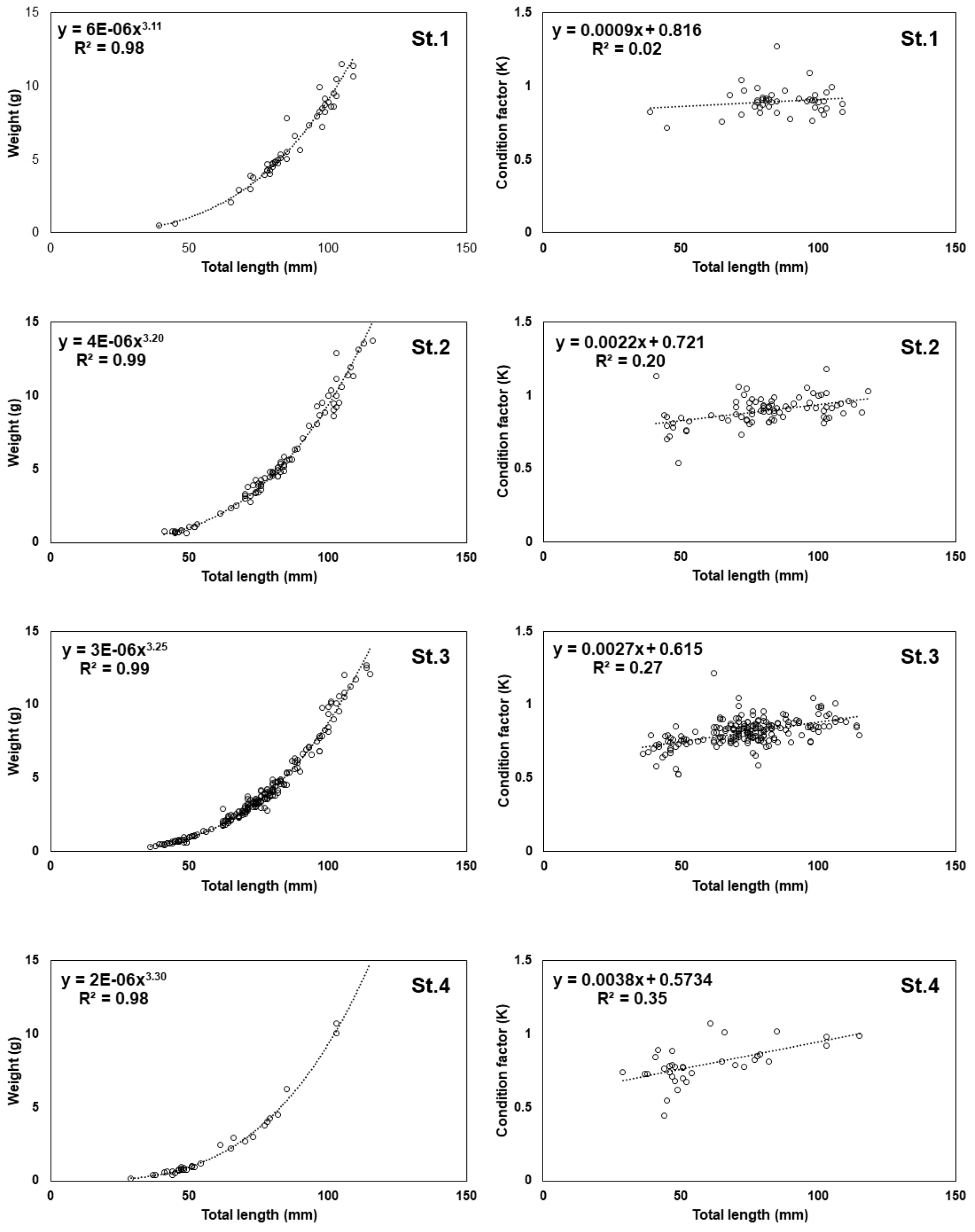
We analyzed the relationship between total length and weight to assess the growth patterns and health of the G. brevibarba population. The analysis revealed that the allometric coefficient (b-value) of the length-weight relationship was greater than 3.0 at all survey sites, indicating favorable growth. This indicated that the G. brevibarba population generally exhibited either isometric growth or, more commonly, a trend in which weight increased faster than length. This suggested that habitat has a positive effect on growth. The highest b-value was recorded at St.4 (3.30), followed by St.3 (3.25), St.2 (3.20), and St.1 (3.11).
The trend lines for the condition factor (K) also showed positive slopes at all sites. Notably, St.4 had the highest slope, whereas St.1 had the lowest. This indicated a complex interaction between the population growth stage and habitat. At St.1, where larger individuals were prevalent, feeding efficiency and growth rate may have slowed after reaching a certain size. In contrast, the population at St.4 was composed of smaller juveniles who were likely engaged in active feeding and rapid growth. These results support the conclusion that downstream sites provide a favorable environment for the growth and development of young G. brevibarba.
Overall, the Pyeongchang River area is considered to have suitable environmental conditions for the growth and habitation of G. brevibarba. Specifically, upstream sites function as primary habitats for large individuals and provide an environment conducive to spawning. Conversely, downstream sites are thought to serve as crucial nursery grounds and are better suited for the growth and density maintenance of the juvenile population.
Age Distribution and Potential Growth Capability
Age Structure Analysis
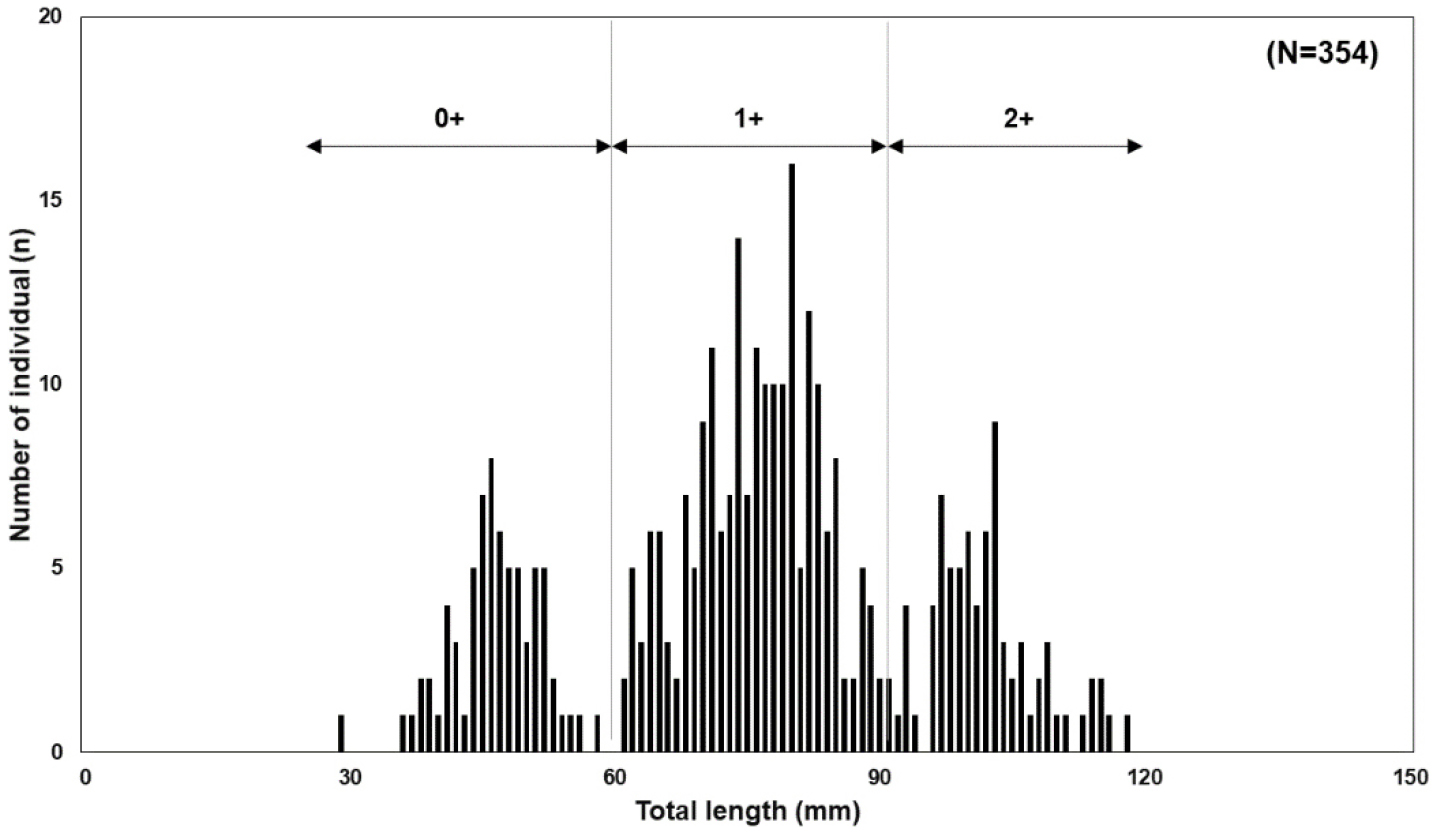
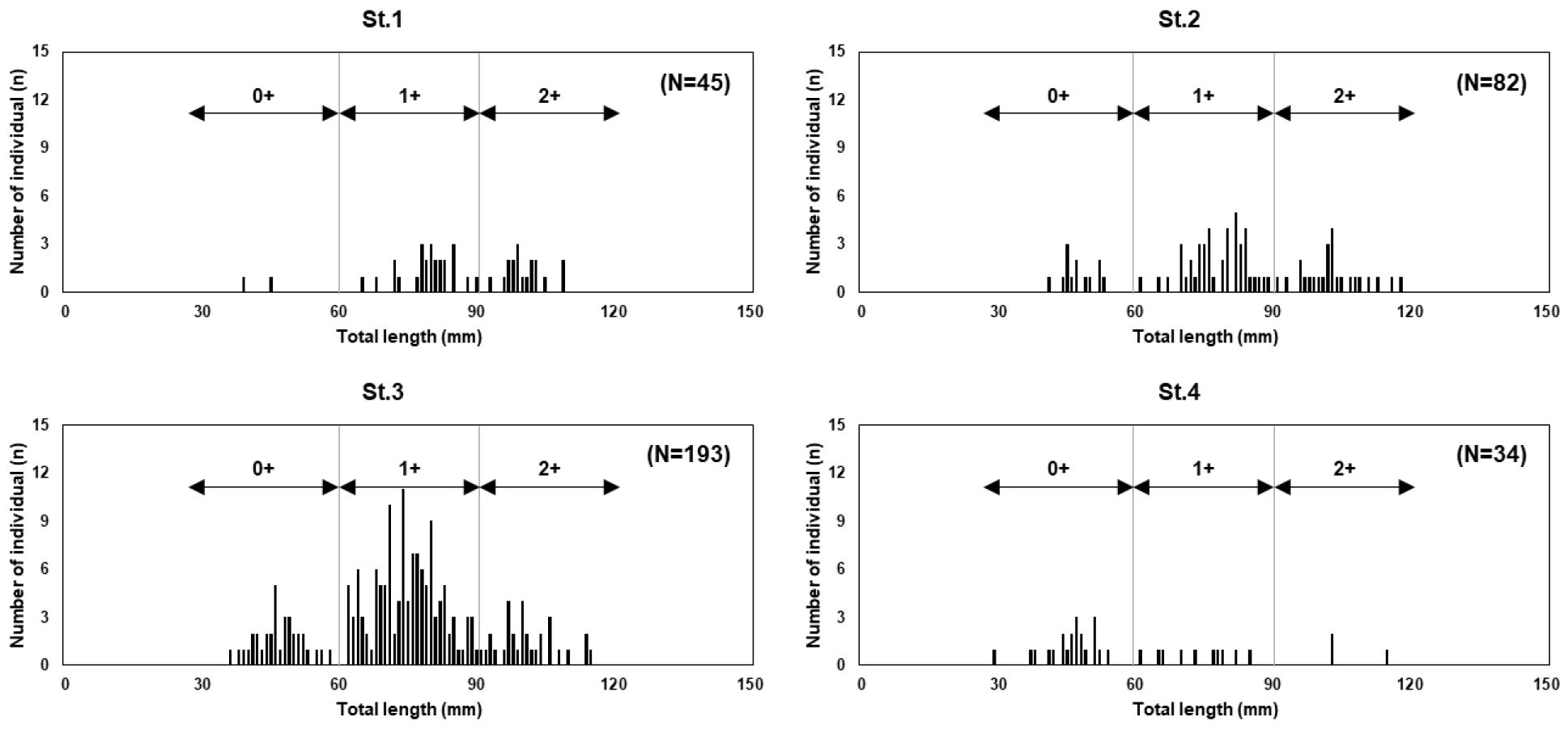
The TL of the G. brevibarba population in the Pyeongchang River ranged from 29 to 118 mm, with a mean TL of 75.7 ± 19.1 mm. Based on the length-frequency distribution analysis, we estimated the following age groups: individuals with a TL of 29-60 mm were classified as young-of-the-year (0+ year), 56-92 mm as one-year-olds (1+ year), and those over 88 mm as two-year-olds (2+ years) or older (Fig. 4, 5). This age-specific length classification is generally consistent with the growth characteristics of G. brevibarba presented by Kim (1997) (1-year: 40 mm, 2-year: 60-80 mm; 3+ years: 100-120 mm), which supports the reliability of our age estimation results.
A comparison of the length-frequency distribution and age structure at each survey site revealed the following results (Fig. 6):
St.1 had a TL range of 39-109 mm, with mature individuals (1+ year and 2+ year or older) being the dominant group, except for a few individuals.
St.2 had a TL range of 41-118 mm, with a dominant distribution of mature individuals, similar to St.1.
St.3 had a TL range of 36-115 mm, with one-year-olds (1+ year) comprising the highest proportion, followed by young-of-the-year (0+ year) and two-year-olds (2+ year) or older.
St.4 was dominated by young-of-the-year individuals, followed by one-year-olds and two-year-olds or older, showing a clear difference in age structure among the sites.
These differences in age structures among the sites were closely related to habitat characteristics. A stable and relatively fast-flowing habitat in the upper reaches (St.1 and St.2) was suitable for larger individuals (2+ years or older). The fast current in the upstream sections is a limiting factor for the habitation of smaller young-of-the-year individuals, thereby reducing their occurrence. In contrast, downstream sites such as St.3 and St.4 provide microhabitats with a diverse distribution of fine gravel and a relatively slow current, creating a favorable environment for the growth of young individuals. The presence of individuals older than one year at all survey sites suggests that the G. brevibarba population exhibits a relatively wide range of habitat adaptability within the Pyeongchang River.
Potential Growth Capability Analysis
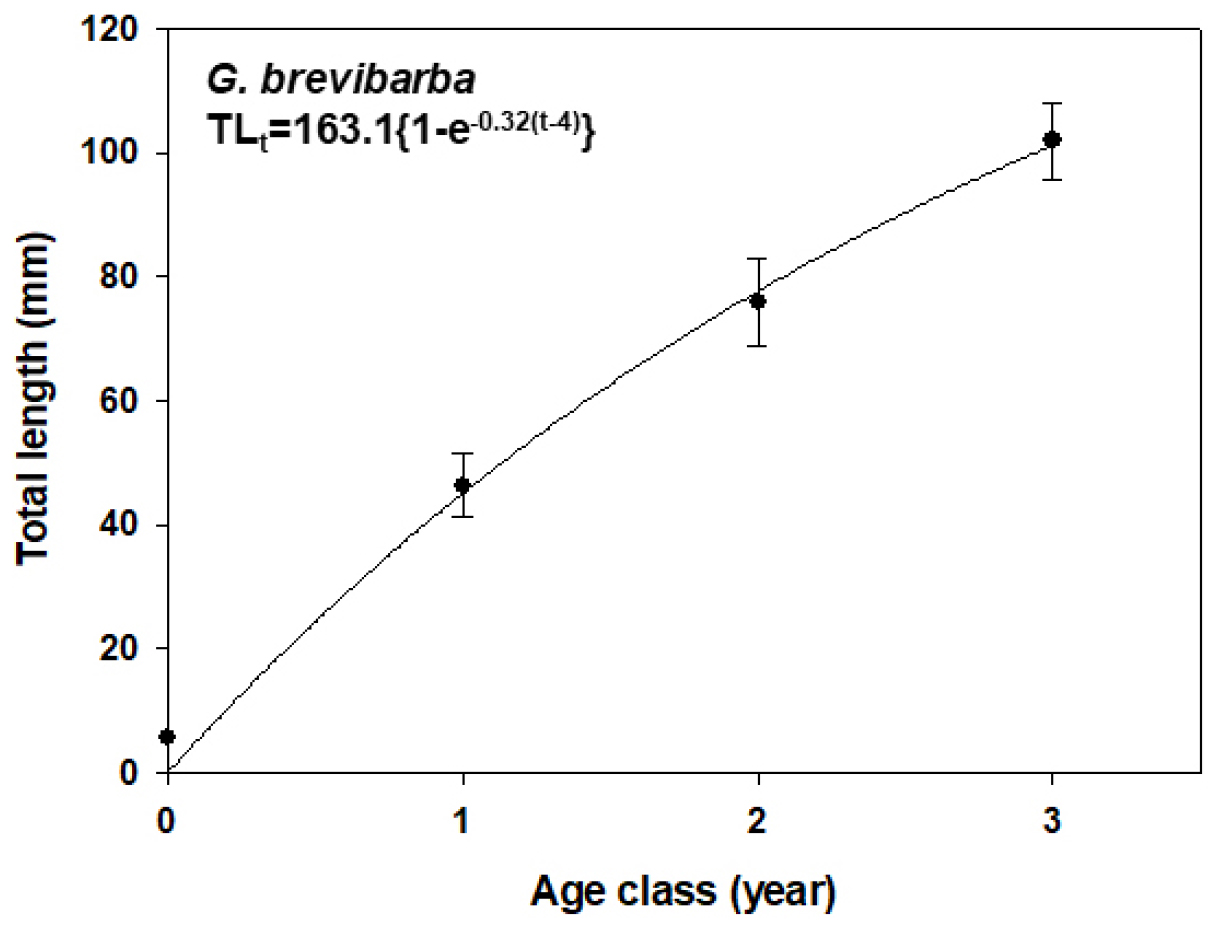
Fish growth follows a typical pattern of high growth rates in the early and middle stages, which gradually slow down in the later stages until a maximum asymptotic size is reached. The von Bertalanffy growth model (von Bertalanffy, 1938) is highly suitable for estimating this biological growth pattern (Choi et al., 2006). In this study, the von Bertalanffy growth model was applied to the G. brevibarba population based on age classes (four classes were applied to the model), which were categorized through the length-frequency distribution.
The model estimated the asymptotic length (L∞) of the G. brevibarba population to be 163.1 mm and the Brody growth coefficient (k) to be 0.32 (Fig. 7). This indicated that the Pyeongchang River G. brevibarba population has sufficient growth potential.
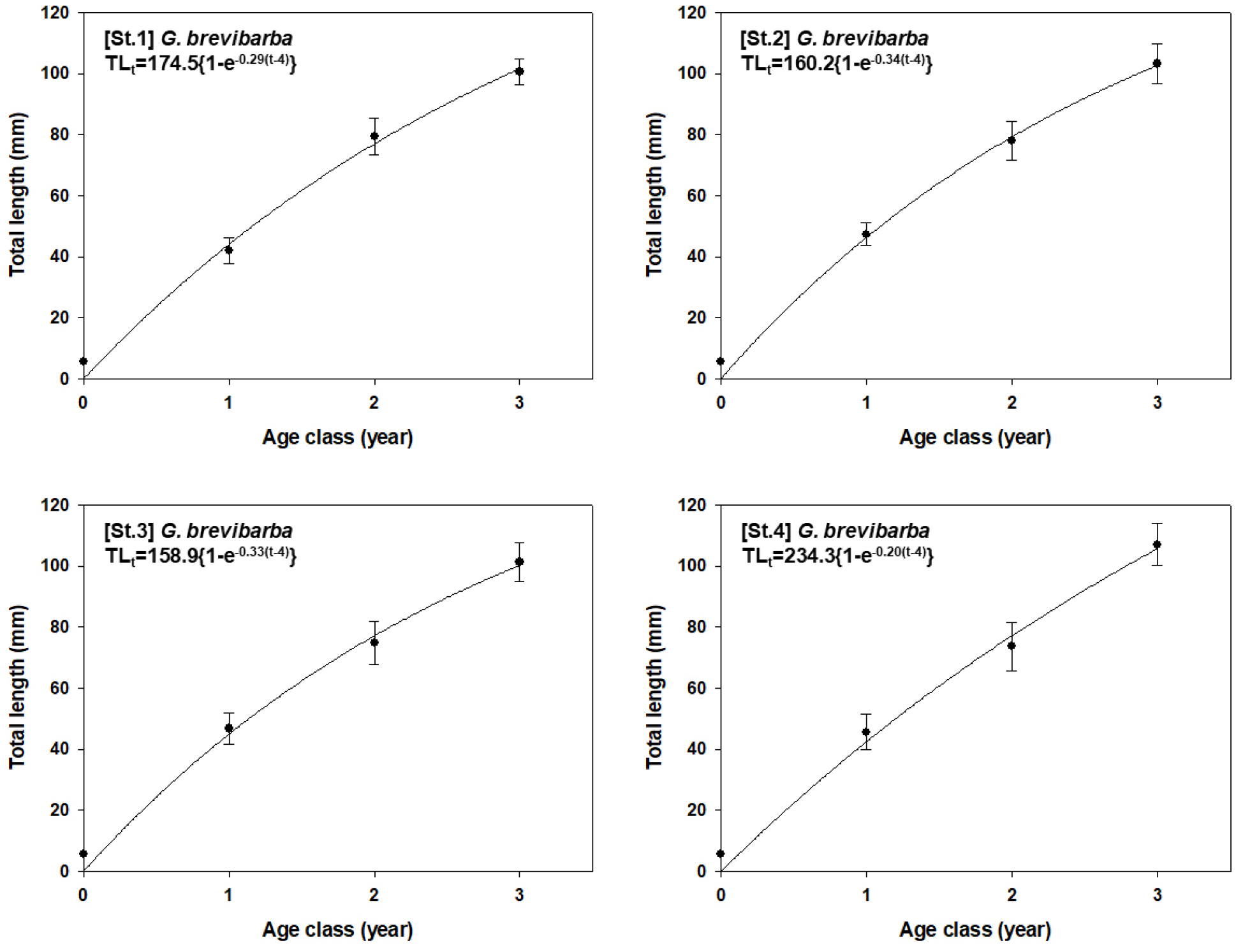
A comparison of the potential growth capability among the sites revealed that St.4 showed the highest potential, with an L∞ of 234.3 mm and a k of 0.20. In contrast, St.3 showed the lowest values, with an L∞ of 158.9 mm and a k of 0.33. St.1 and St.2 showed intermediate values, with St.1 having an L∞ of 174.5 mm and a k of 0.29, and St.2 having an L∞ of 160.2 mm and a k of 0.34 (Fig. 8).
These differences in the potential growth capability among the sites are closely related to the age and size composition of the individuals. Generally, the younger and smaller the individuals, the higher their potential growth capability, which is often attributed to more active feeding and faster growth rates. Accordingly, a high potential growth capability was estimated for St.3 and St.4, where a large number of young individuals were found. Conversely, a relatively low potential growth capability was observed at St.1 and St.2, which had low numbers of young-of-the-year individuals and a dominance of larger individuals (2+ years or older). This further reinforces our previous interpretation that the upstream sections serve as suitable habitats for mature individuals and spawning, whereas the downstream sections function as crucial nursery grounds for juvenile growth and development.
In conclusion, the fish community in the Pyeongchang River, Yeongwol, exhibited high species diversity and a high rate of legally protected species. The particularly high proportion of endemic Korean species clearly indicates that the aquatic environment of this region is well-preserved. The surveyed G. brevibarba population was considered healthy throughout all sections, showing favorable growth and high potential growth capability, which suggests that it maintains a stable population. These results emphasize that the Pyeongchang River is a riverine ecosystem with very high ecological conservation value.
The findings of this study are expected to serve as meaningful baseline data for future research on G. brevibarba populations in other river systems and for the establishment of restoration strategies. Furthermore, to better understand the ecological characteristics of G. brevibarba and to secure foundational data for long-term conservation and restoration, continuous data accumulation through long-term seasonal monitoring is essential. The ongoing collection of quantitative data on key habitat factors such as water temperature, flow velocity, streambed composition, and water quality will provide a more robust basis for effectively managing and conserving G. brevibarba populations in a changing environment.