Introduction
Materials and Methods
Investigation of Cultivation History and Characteristics
Criteria for Characteristics Survey
Statistical Analysis
Results and Discussion
Analysis of Characteristics and Genetic Mechanisms of Interspecific Hybrids
Growth Characteristics of Selected Breeding Combinations
Hormone Treatment for Fruit Set Induction
Fruit Characteristics of Interspecific Hybrids
Introduction
Pumpkins, a major food crop belonging to the Cucurbitaceae family, are classified into five species, including Cucurbita moschata, Cucurbita maxima, and Cucurbita pepo. Among these, C. moschata and C. maxima are primarily cultivated in South Korea. C. maxima was introduced to South Korea via Japan in the 1920s, and its production expanded significantly from the 1960s (Kim et al., 2012). It is characterized by a deep green rind, bright yellow flesh, and high levels of functional components including β-carotene, vitamins, minerals, and fiber (Park et al., 2016). Additionally, its high sugar content exceeding 8° Brix has led to increased consumer preference for these varieties, resulting in a continuous rise in domestic and international consumption (Kim et al., 2012).
Meanwhile, C. moschata, native to Central America and southern Mexico, exhibits high cultivation stability in hot and humid environments. Its low-calorie content (approximately 40 kcal) and high β-carotene content make it highly valued as a health and diet food (Itle and Kabelka, 2009; Olson et al., 2006). β-carotene is known to convert to vitamin A in the body, contributing to vision health (Slaska-Grzywna et al., 2016; Zaccari and Galietta, 2015; Xu et al., 2024). Furthermore, these two species exhibit differences in storage and quality retention: C. maxima shows a decrease in carotenoid content during storage, while C. moschata has been reported to maintain marketability for a certain period (Kim et al., 2012). These differences suggest that the two species possess complementary characteristics, emphasizing the need for breeding research to harness this potential.
C. maxima and C. moschata possess complementary advantages in various aspects, such as growth, functionality, and fruit quality. Therefore, interspecific hybridization between them is considered a crucial breeding strategy for securing novel trait combinations and utilizing hybrid vigor. A representative success case is the ‘Tetsukabuto’ variety developed in 1951, which was reported to have high yields and productivity through the hybridization of two species (Formiga et al., 2019). This case suggests that C. maxima × C. moschata hybridization can make a substantial contribution to breeding trait improvement.
This study aimed to investigate the growth characteristics and fruit traits of hybrid individuals obtained through the interspecific hybridization of C. maxima and C. moschata.
Materials and Methods
Investigation of Cultivation History and Characteristics
In this experiment, we utilized Cucurbita maxima and Cucurbita moschata genetic resources stored in the laboratory. Using 7 C. maxima genetic resources and 10 C. moschata genetic resources, we conducted a total of 15 crossbreeding combinations (Table 1). After the hybridization, seeds were placed in Petri dishes lined with filter paper for 20 days, and germination was induced in a constant-temperature oven maintained at 30°C under dark conditions. Seeds with radicle lengths of 2 mm or more were sown in a 50-hole plug tray (5 × 10) filled with horticultural potting mix and underwent a seedling-raising process for approximately 30 days. Subsequently, seedlings were transplanted into the unheated seedling greenhouse at the Kangwon National University Farm in Sinbuk-eup, Chuncheon-si, Gangwon-do, South Korea, with a spacing of 30 cm between rows.
Table 1
Crossbreeding combinations using C. moschata as the female parent and C. maxima as the male parent
To suppress aphid infestation during pumpkin cultivation, water-dispersible granules Sulfoxaflor (Straight, Dongbang Agro Corporation, Korea) were applied as a foliar spray prior to transplanting. For powdery mildew control, a suspension concentrate (Bibando, FarmHannong Co., Korea) was sprayed. To secure individuals with new traits, interspecific hybridization was conducted. Hybridization was performed at 6 a.m. on the day the female flowers bloomed at the 10th node, using artificial pollination: transferring pollen from C. moschata to female flowers of Cucurbita maxima, and pollen from C. maxima to female flowers of C. moschata.
To overcome the male sterility issue that arises in interspecific crossing between varieties with dissimilar genes, a hormonal treatment agent was utilized. This study referenced the method of Hu et al. (2019) to apply synthetic growth hormones from the auxin group: 2,4-Dichlorophenoxyacetic acid (2,4-D) at concentrations of 50, 100, 200, and 400 ppm, and gibberellin at concentrations of 50 and 100 ppm, each as a single treatment. Additionally, experiments were conducted by mixing 2,4-D and gibberellin in ratios of 50:50, 50:100, 100:50, and 100:100. The fruit set rate and characteristics of set fruit were investigated according to the concentration of the hormone treatment agent.
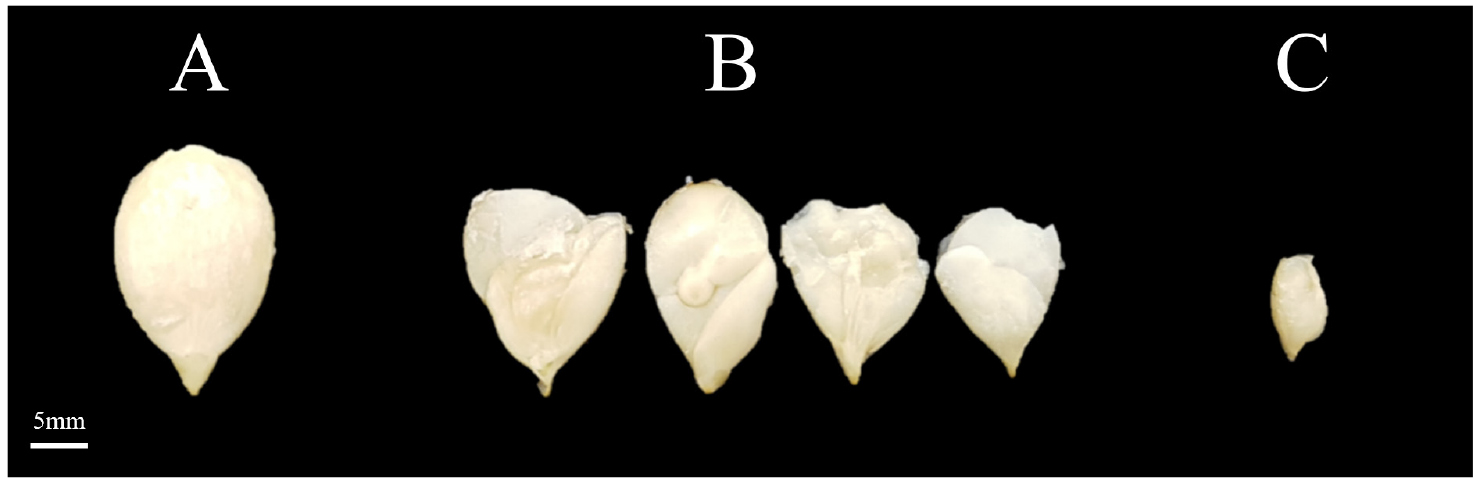
After 45 days had passed since the pollination, the fruit was harvested, and the seeds inside were examined. The seed testing survey was conducted by referring to the hybridization results between C. moschata and C. maxima reported by Karaağaç and Balkaya (2013) and the effects of plant growth regulators on watermelon seed count reported by Jaskani et al. (2005). Survey items included the number of pollinated flowers and fruit set number, fruit set rate, seed morphology, and seed degeneration rate. Seeds were classified into normal seeds and seeds that were completely degenerated during development (Fig. 1). Through this analysis, we selected crossbreeding combinations with a high proportion of normal seeds.
Subsequently, F1 seeds from a crossbreeding combination with a high proportion of normal seeds were cultivated to confirm the development of floral organs and the occurrence of male sterility. This process involved the evaluation of the floral organs developed normally, while determining the physiological characteristics and growth potential of the variety. Subsequently, we comprehensively investigated growth characteristics and fruit traits to select varieties with superior qualities.
Criteria for Characteristics Survey
The growth characteristic survey was conducted using the crop-specific characteristic survey criteria for Cucurbita (C. maxima and C. moschata) of the Korean Seed and Variety Service. Fruit characteristics measured included fruit weight, fruit length, fruit width, sepal length, flesh thickness, sugar content, fruit peel color, and flesh color.
The fruit length and fruit width were analyzed using ImageJ (Ver. Java 1.8.0, National Institutes of Health, Baltimore, MD, USA) by taking the images after arranging them horizontally and vertically at a right angle. The sugar content of the fruit was measured using a digital refractometer (Pocket Refractometer PAL-1, Atago, Japan) after grinding the fruit with a grater and filtering the juice through gauze. The peel and flesh color was measured on the flesh cross-section and peel surface after cutting the central part of the fruit. At this point, the device was calibrated using a standard white plate (L = 97.79, a = -0.38, b = -2.05), and Hunter ‘L*’, ‘a*’, ‘b*’ values were recorded using a colorimeter (CR-400, Minolta Japan).
Statistical Analysis
Statistical analysis was performed using SPSS (Ver. 26, IBM Corp., Armonk, NY, USA). After analyzing the ANOVA results, Duncan’s multiple range test was applied at the 5% level.
Results and Discussion
Analysis of Characteristics and Genetic Mechanisms of Interspecific Hybrids
This study investigated the characteristics of interspecific hybrids from a total of 16 genotype combinations. Genetic resources include C. moschata of 15-21-2S, 15-19-7S, 15-17R-1s, 15-23R-6S, 15-31-1S, 15-18-4S, 15-23R-6S and 15-16-2S along with C. maxima of 5-1-1, 3-8-2, 1-1-1, 4-4-12, and 3-3-1. The crossbreeding combination of C. maxima and C. moschata are 15-21-2S X 5-1-1, 15-19-7S X 5-1-1, 15-19-7S X 3-8-2, 15-17R-1s X 3-8-2, 15-23R-6S X 3-8-2, 15-19-7S X 1-1-1, 15-21-2S X 4-4-12, 15-19-7S X 4-4-12, 15-17R-1s X 4-4-12, 15-31-1S X 4-4-12, 15-18-4S X 4-4-12, 15-16-2S X 4-4-12, 5-1-1 X 15-2-7S, 3-3-1 X 15-11-2S, and 15-2-7S X 15-19-7S, totaling 16 combinations.
The seed characteristics of the interspecific hybrids are shown in Table 2. An examination of the seeds from the investigated interspecific hybrids revealed that pollination had occurred in most cases. However, during development, the embryo often degenerated or failed to fully develop, resulting in numerous seeds exhibiting partially degenerated morphology (Fig. 1). Karaağaç and Balkaya (2013) found that out of 234 crossbreeding combinations in the Cucurbitaceae family, seed production occurred in 79 crossbreeding combinations. Among these, the crossbreeding combination between C. maxima and C. moschata showed relatively better seed production rates. This study reported that in three combinations, the ratio of normal seeds to degenerated seeds was more favorable compared to other combinations, and in most combinations, seeds either failed to develop normally or showed signs of degeneration. In hybrids of the Cucurbitaceae family, seed production is hindered by genetic infertility mechanisms that inhibit normal pollen formation and cause problems during fertilization. This leads to reduced seed survival rates and quality, and overcoming these genetic barriers remains a critical research challenge in enabling hybrids to produce seeds (Tinna, 2019).
Table 2
Seed of interspecies hybridization in genotypes with Cucurbita moschata and Cucurbita maxima combinations
Among the 16 crossbreeding combinations analyzed in this study, the seed degeneration rate was mostly 95±5%. However, relatively low seed degeneration rates were observed in three crossbreeding combinations: -3-1 × 15-11-2S, 19-7S × 3-8-2, and 15-21-2S × 5-1-1. Due to the low seed degeneration rate, seeds develop and remain healthy. For each crossbreeding combination, the number of defective and normal seeds was 76 and 121, respectively, for 15-21-2S × 5-1-1 and 65 and 84, respectively, for 19-7S × 3-8-2. Finally, the number was 128 and 130, respectively, for 3-3-1 × 15-11-2S, which was found to be higher than in other crossbreeding combinations. This is attributed to the relatively close genetic relationship between their crossbreeding combinations. The results of this study align with previous research, which showed that the proportion of normal seeds was higher in crosses between closely related lineages and that the fertilization rate tended to decrease as the genetic distance between lineages increased (Karaağaç and Balkaya, 2013).
The high seed degeneration rates observed in most other combinations in this study are attributed to reproductive sterility and developmental disorders caused by chromosomal mismatches and genetic incompatibility in the polyploid hybrids resulting from interspecific hybridization, as demonstrated in the study by Ramsey and Schemske (1998). These results suggest that hybrid productivity is highly dependent on genetic factors, and future research should focus on methodologies to overcome these genetic barriers.
Upon completing this investigation, the following crossbreeding combinations, which exhibited relatively high proportions of normal seeds in their seed characteristics, were selected: 15-21-2S × 5-1-1, 3-3-1 × 15-11-2S, 15-17R-1S × 3-8-2, 15-21-2S × 4-4-12, 15-19-7S × 3-8-2, and 15-23R-6S × 3-8-2. The F1 seeds from these crossbreeding combinations were sown and cultivated. These breeding combinations have the potential to serve as key foundational data for the development of new varieties in the future.
Growth Characteristics of Selected Breeding Combinations
The results of sowing F1 seeds from the crossbreeding combination with a high proportion of normal seeds and investigating their growth characteristics are shown in Table 3. When examining leaf size by combination, the leaf width and length of the 15-17R-1S × 3-8-2 combination were low at 14.5 cm and 16.5 cm, respectively. In contrast, the 15-21-2S × 4-4-12 combination exhibited the largest leaf morphology at 36.2 cm in width and 37 cm in length compared to other combinations. The 3-3-1 × 15-11-2S and 15-19-7S × 3-8-2 combinations also showed relatively large leaf sizes. This indicates that the genetic variation in the hybrids was reflected in the leaf size trait.
Table 3
Phenotype of the interspecies hybrid cultivar during plant growth
In all combinations, the leaf margin shape and marbling pattern appeared consistently, and the green intensity was also clearly expressed in all combinations, suggesting that these are common characteristics of interspecific hybrid varieties. The length and width of the leaf stalk showed significant variation among combinations. The length of the leaf stalk in the 15-21-2S × 4-4-12 combination was 34.1 cm, indicating a long leaf stalk, while the 15-17R-1S × 3-8-2 combination had a relatively short leaf stalk at 14.6 cm. The width of the leaf stalk was also wider at 1.3 cm for the combinations 3-3-1 × 15-11-2S and 15-21-2S × 4-4-12, while it was narrower at 0.4 cm for the combination 15-17R-1S × 3-8-2.
In plants, broad leaves can absorb more light, which is advantageous for promoting photosynthesis. Long leaf stalk allows leaves to spread more widely, reducing the shade cast by overlapping leaves and maximizing light absorption. Additionally, the long and thick leaf stalk reduces the impact of wind, helping to prevent leaf damage (Filartiga et al., 2022). Therefore, the length and width of leaves and leaf stalks can serve as key indicators for evaluating the structural stability and durability of a plant (Yoshida et al., 2011). The 15-17R-1S × 3-8-2 combination appears to have relatively smaller leaf width and length compared to other combinations, along with shorter leaf stalk length and width, suggesting insufficient support capacity. This is expected to reduce photosynthetic efficiency, potentially impairing plant growth. On the other hand, the 15-21-2S × 4-4-12 and 3-3-1 × 15-11-2S combinations demonstrated superior leaf and leaf stalk traits compared to other combinations, making them a promising candidate for further crossbreeding evaluation.
Differences were observed among combinations in terms of the sepal length and peduncle characteristics in female flowers. The 15-17R-1S × 3-8-2 combination had the longest sepal length at 6.3 cm, while the 3-3-1 × 15-11-2S combination had the shortest at 1 cm. At 4.3 cm, the peduncle length was longest in the 15-23R-6S × 3-8-2 combination, while the peduncle width ranged from 0.6-1.1 cm according to crossbreeding combinations, showing no significant variation. The sepal and peduncle of the male flower also exhibited distinct characteristics depending on the combination. In the 15-19-7S × 3-8-2 combination, the sepal length and peduncle length of the male flower were 2.7 and 7.8 cm, respectively, which were relatively shorter than in other combinations. However, in the combination of 3-3-1 × 15-11-2S, the peduncle length was 30.9 cm, which was considered long, and the peduncle width was 0.7 cm, indicating favorable results.
In certain combinations, the complete form of the male flower could not be observed (Fig. 2A, B). This is considered to be a sterile form of the male flower, likely due to the presence of pollen incapable of fertilization or because the stamen was degenerated and failed to develop fully. When examining the female flowers of the interspecific hybrid (Fig. 2C, D), they exhibited normal morphology. Queiroga et al. (2017) reported that in the hybrid Tetsukabut, created by crossbreeding C. maxima and C. moschata, male and female flowers bloom on the same plant. However, when female flowers appear, male flowers are absent or rare; therefore, the hybrid is considered male sterile. This can result in defects in organ development due to interactions between heterogeneous genes or chromosomal mismatches, or lead to the production of incomplete pollen due to a triploid genetic composition, causing sterility of male flowers. Additionally, infertility may occur due to chromosome fragmentation or bridge formation during cell division. This genetic incompatibility has been reported to act as a major cause of reproduction and infertility in interspecific hybridization (Fishman and Willis, 2006; Sevilleno et al., 2023).
Considering the combined growth characteristics of female and male flowers, hormonal treatment to induce fruit set should be conducted, as the reduced capacity of reproductive organs in interspecific hybridization negatively impacts fruit set. Growth characteristic surveys confirmed that F1 interspecific hybrids exhibit diverse leaf sizes, lengths of leaf stalks, and flower structures, depending on their genetic combinations, indicating genetic variation. Alix et al. (2017) reported that various mutations can arise through gene transfer and recombination, and that increased genetic diversity through mutation can positively influence the adaptability and survival of a species.
Hormone Treatment for Fruit Set Induction
When F1 seeds, with a high proportion of normal seeds derived from 16 genotype combinations, were sown and cultivated, flowers exhibiting male sterility characteristics showed signs of sterility due to the heterogeneous genetic background. As a result, the male flowers lacked reproductive capacity, and artificial pollination was induced by applying hormones to the stigma of the female flowers. Previous studies have reported that treating male sterile fruit with 2,4-Dichlorophenoxyacetic acid (2,4-D), which is an auxin-type hormone treatment agent, at 50 ppm increased fruit set rates and promoted fruit development (Hu et al., 2019). Additionally, MIRANDA (2012) reported that treating Tetsukabuto squash—a hybrid of C. maxima and C. moschata—with 2,4-D (187.5-262.5 mg/L) through a plant hormone inducer yielded superior results in average fruit weight, yield, and growth cycle compared to the naturally pollinated control group. Fruit weight and yield increased with an increase in 2,4-D concentration up to 250 mg/L.
The crossbreeding combination used in this study was 3-3-1 × 15-11-2S. Hormone treatments included 2,4-D and gibberellin (GA), applied both individually and in combination (Table 4). Experimental results showed that fruit set occurred only in the treatment group treated with 2,4-D, while no fruit set occurred in the treatment group treated with GA alone. These results are consistent with previous studies indicating that gibberellin had no direct effect on fruit set or exerted a negative influence in Cucurbitaceae, contrary to the common finding that gibberellin positively affected growth and development in most plants (Watanabe et al., 2008).
Table 4
Fruit characteristics according to the type and density of hormones in interspecies hybrid cultivar (C. moschata × C. maxima combinations)
| Hormone |
Fruit set ratioz |
Fruit weight (g) |
Fruit length (mm) |
Fruit Diameter (mm) |
Flesh thickness (mm) |
Firmnessy (N) | TSSx | |||||
| 2,4D 50 ppm | 3/3 (100%) | 1616.6 ± 138.7 a | 116.6 ± 1.5 a | 165.6 ± 4.0 a | 31.5 ± 0.9 a | 30.2 ± 0.3 a | 11.6 ± 0.8 ab | |||||
| 2,4D 100 ppm | 3/3 (100%) | 1460.0 ± 91.7 a | 119.3 ± 2.0 a | 156.3 ± 5.8 a | 32.2 ± 1.8 a | 27.5 ± 3.5 a | 10.6 ± 1.1 bcd | |||||
| 2,4D 200 ppm | 3/3 (100%) | 1606.6 ± 348.5 a | 123.0 ± 9.5 a | 165.3 ± 9.8 a | 28.4 ± 4.6 a | 28.6 ± 3.7 a | 9.6 ± 0.1 d | |||||
| 2,4D 400 ppm | 4/4 (100%) | 1610.0 ± 190.3 a | 122.0 ± 3.1 a | 163.7 ± 6.6 a | 33.3 ± 2.7 a | 29.5 ± 1.9 a | 10.2 ± 0.5 cd | |||||
| 2,4D50 + GA50 | 3/3 (100%) | 1393.3 ± 137.1 a | 114.3 ± 3.5 a | 154.3 ± 5.8 a | 28.4 ± 2.0 a | 29.6 ± 0.5 a | 10.7 ± 0.8 bcd | |||||
| 2,4D50 + GA100 | 3/3 (100%) | 1523.3 ± 194.9 a | 115.0 ± 4.5 a | 162.0 ± 6.5 a | 32.0 ± 3.0 a | 25.2 ± 4.8 a | 12.1 ± 0.5 a | |||||
| 2,4D100 + GA50 | 3/3 (100%) | 1565.0 ± 204.7 a | 120.3 ± 6.0 a | 163.0 ± 8.8 a | 30.5 ± 1.5 a | 30.7 ± 6.4 a | 11.1 ± 0.5 abc | |||||
| 2,4D100 + GA100 | 3/3 (100%) | 1540.0 ± 164.5 a | 120.6 ± 9.8 a | 159.3 ± 4.7 a | 31.3 ± 2.5 a | 31.2 ± 2.8 a | 11.2 ± 0.5 abc | |||||
| GA50 | 0/3 (0%) | - | - | - | - | - | - | |||||
| GA100 | 0/3 (0%) | - | - | - | - | - | - | |||||
| Hormone | Skin color | Flesh color | ||||||||||
| L* | a* | b* | L* | a* | b* | |||||||
| 2,4D 50 ppm | 23.7 ± 0.9 a | -2.0 ± 0.3 a | 3.0 ± 0.8 a | 63.0 ± 1.5 a | 10.5 ± 2.8 a | 38.2 ± 0.5 a | ||||||
| 2,4D 100 ppm | 24.3 ± 0.4 a | -1.9 ± 0.2 a | 2.8 ± 0.3 a | 66.5 ± 1.3 a | 13.1 ± 2.6 a | 39.5 ± 0.7 a | ||||||
| 2,4D 200 ppm | 23.5 ± 0.5 a | -1.3 ± 0.1 a | 1.8 ± 0.1 a | 65.2 ± 2.9 a | 10.9 ± 1.3 a | 39.0 ± 1.2 a | ||||||
| 2,4D 400 ppm | 24.4 ± 1.9 a | -1.9 ± 0.7 a | 2.9 ± 1.1 a | 64.1 ± 1.3 a | 11.1 ± 1.9 a | 38.7 ± 0.3 a | ||||||
| 2,4D50 + GA50 | 23.6 ± 1.4 a | -1.4 ± 0.4 a | 2.2 ± 0.7 a | 63.8 ± 2.7 a | 12.4 ± 0.1 a | 38.8 ± 1.2 a | ||||||
| 2,4D50 + GA100 | 23.9 ± 0.5 a | -1.9 ± 0.4 a | 2.6 ± 0.6 a | 62.7 ± 6.1 a | 13.4 ± 1.5 a | 38.0 ± 2.9 a | ||||||
| 2,4D100 + GA50 | 23.9 ± 1.6 a | -1.4 ± 0.7 a | 2.2 ± 1.2 a | 64.2 ± 1.3 a | 11.8 ± 2.7 a | 38.7 ± 0.6 a | ||||||
| 2,4D100 + GA100 | 23.2 ± 0.8 a | -1.5 ± 0.3 a | 2.3 ± 0.3 a | 63.1 ± 1.8 a | 11.5 ± 1.6 a | 38.1 ± 0.1 a | ||||||
| GA50 | - | - | - | - | - | - | ||||||
| GA100 | - | - | - | - | - | - | ||||||
2,4-D induced normal fruit set at concentrations of 50 ppm or higher, and no synergistic effect was observed with combined treatment using GA. In the Tetsukabuto squash study, 2,4-D concentration within a certain range positively influenced yield and fruit weight as a means to ensure and increase fruit set in interspecific hybrid pumpkin production, serving as an alternative to pollen use. When concentrations significantly increased up to 250 mg/L, fruit weight and yield significantly increased, whereas higher concentrations negatively impacted fruit development and quality (Queiroga et al., 2017).
Fruit Characteristics of Interspecific Hybrids
In this study, the fruit morphology induced by hormone treatment was found to resemble that of C. maxima (Fig. 3). The fruit characteristics are listed in Table 5. The fruit characteristics survey items were investigated using the Korea Seed and Variety Service’s crop-specific characteristic survey standards, covering fruit weight, fruit length, fruit width, sepal length, flesh thickness, and soluble solids content. Color analysis was conducted on the fruit peel and flesh.
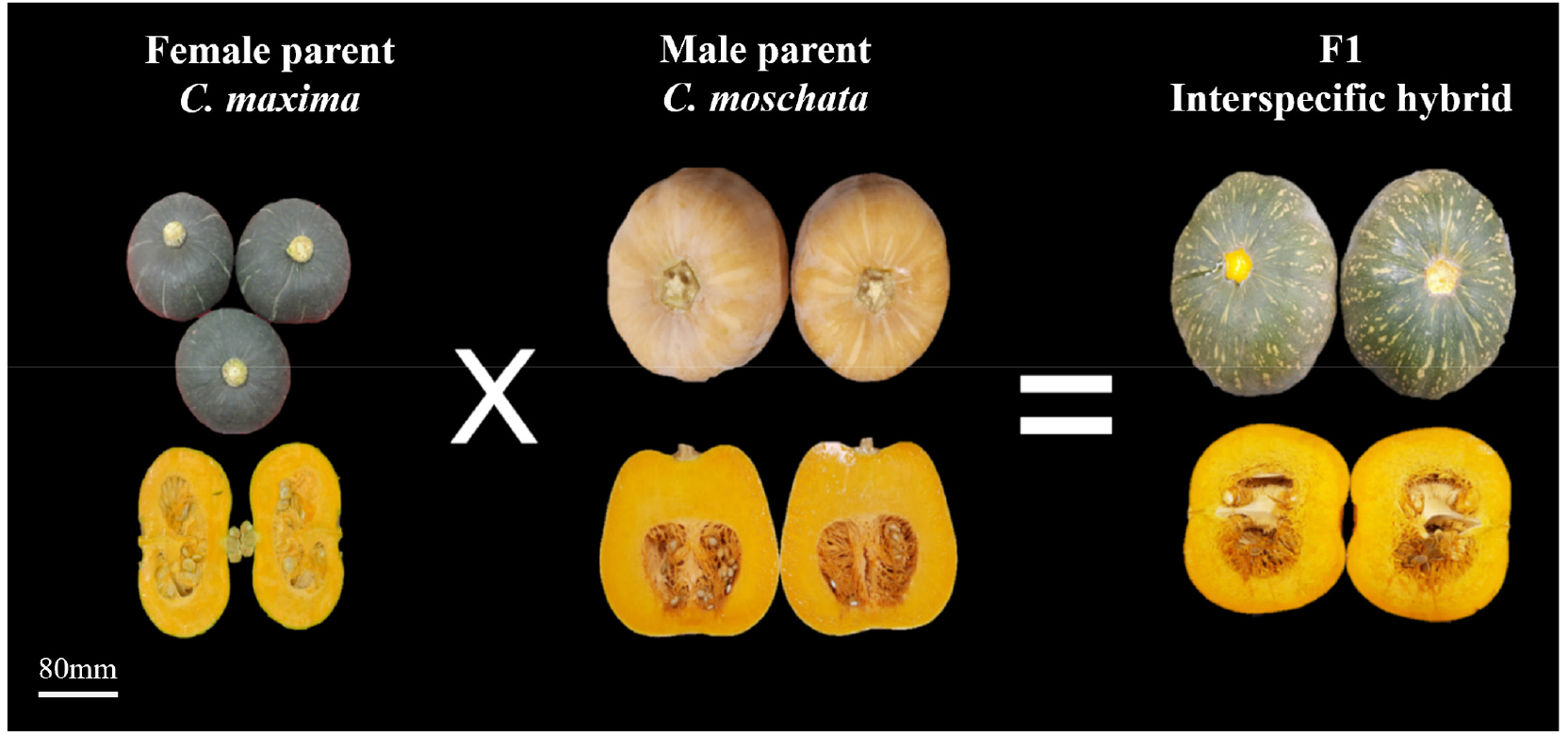
Fig. 3.
Fruit morphology of interspecific hybrids derived from C. maxima (female parent) and C. moschata (male parent). The parental fruits show distinct rind color, size, and internal structure, whereas the F1 interspecific hybrids display intermediate phenotypes in both external and internal fruit characteristics.
Table 5
Fruit characteristics of interspecies hybridization in genotypes with C. moschata and C. maxima combinations
In terms of fruit weight, the 15-19-7S × 3-8-2 combination was the heaviest at 1.97 kg, while the 15-23R-6S × 3-8-2 combination was the lightest at 1.39 kg. As for fruit length, the 15-19-7S × 3-8-2 combination was the longest at 170.08 mm, while the 3-3-1 × 15-11-2S combination was the shortest at 137.53 mm. Regarding fruit width, the combination 15-19-7S × 3-8-2, which showed the heaviest fruit weight, recorded the largest width at 167.00 mm, while the combination 15-21-2S × 5-1-1 had the smallest width at 109.79 mm. Monforte et al. (2014) emphasized that fruit morphology, including the length-to-width ratio, plays a crucial role in market classification, and consumer preferences often favor smaller fruits, making size and shape important selection criteria in breeding.
According to a survey by the Korea Rural Economic Institute, consumers prefer small to medium-sized pumpkins suitable for home use, weighing between 1.5 and 2.5 kg. For these reasons, the 3-3-1 × 15-11-2S combination is expected to be more favorable. The 3-3-1 × 15-11-2S combination had the longest sepal length at 74.22 mm, while the 15-21-2S × 5-1-1 combination, which had a smaller fruit width diameter, had the shortest at 47.57 mm. The fruit flesh thickness was thickest at 33.22 mm for the 15-19-7S × 3-8-2 combination, which also produced the heaviest and longest fruit weight and width. Meanwhile, the 15-23R-6S × 3-8-2 combination was found to be the thinnest at 29.88 mm and also the lightest in weight. This indicates that fruit flesh thickness is directly related to fruit weight accumulation and inversely proportional to the fruit’s internal structure. In other words, it aligns with research findings that thicker flesh leads to greater accumulation of fruit mass, while thinner flesh results in larger internal spaces (Gonçalves et al., 2014).
Soluble solids content is used as an indirect indicator of sugar content, but since soluble solids also include various substances such as vitamins, organic acids, phenols, pectin, and pigments that dissolve into the cells, it does not accurately represent the actual sugar content. However, since sugar constitutes approximately 90% of the total as the primary component of soluble solids, it can serve as an indicator that indirectly reflects sugar content (Chitarra and Alves, 2001). When examining the total soluble solids content, the 3-3-1 × 15-11-2S combination showed the highest value at 16.28, while the 15-23R-6S × 3-8-2 combination exhibited the lowest Brix value at 12.2.
In peel color, the 15-21-2S × 5-1-1 combination showed the highest L* value at 61.71, while the 15-19-7S × 3-8-2 combination had the lowest a* value at 16.1, best revealing the green intensity of the peel of C. maxima. The b* values showed no significant difference. In flesh color, the L* value showed high brightness at 47.7 for the 15-21-2S × 5-1-1 combination, the a* value was highest at 0.57 for the combination 15-21-2S × 5-1-1, while the b* value was highest at 16.94 for the 15-21-2S × 5-1-1 combination.
The color of the skin and flesh of C. maxima is an important indicator of quality. Generally, C. maxima has a dark green or bluish-green skin when underripe, and as it ripens, it changes to a brighter green or reddish-orange. The flesh is typically a deep orange color, tending to become more vivid as it ripens. Based on the overall fruit skin color results, the 3-3-1 × 15-11-2S combination was found to maintain a green intensity well, suggesting freshness. The flesh color appears more yellow as the b* value increases, and this yellow color is associated with carotenoids, particularly beta-carotene. Therefore, higher b* values tend to indicate higher carotenoid content, and the 15-21-2S × 5-1-1 combination appears to be superior (Itle and Kabelka, 2009).
Based on these results, the 3-3-1 × 15-11-2S crossbreeding combination is judged to exhibit superior characteristics in fruit weight, size, color, and soluble solids content, aligning with consumer preferences. Therefore, the 3-3-1 × 15-11-2S combination proposed in this study is expected to be a promising choice for future C. maxima cultivation. However, when cultivating this variety, male sterility is caused by the heterogeneous gene combination resulting from interspecific hybridization. Therefore, treatment of the flowering stigma with 2,4-D at a concentration of 50 ppm or higher appears to be of critical importance.