Introduction
Materials and methods
Experimental materials
Measurement of the gallic acid content
Measurement of total phenolic and total flavonoid contents
DPPH radical scavenging assay
ABTS radical scavenging assay
Reducing power
MTT cell viability assay
Tyrosinase inhibition activity
Antimicrobial activity assay
Measurement of nitric oxide (NO) production
Statistical analysis
Results and Discussion
Gallic acid content
Total polyphenol and flavonoid contents
DPPH radical scavenging activity
ABTS
Reducing power
Cell viability
Tyrosinase inhibition
Nitric oxide (NO) production
Antimicrobial activity
Correlation between the antioxidant content and antioxidant activity
Summary
Introduction
Rhus chinensis, a plant found throughout Korea, is known for the galls formed on its leaves by the parasitic aphid Schlechtendalia chinensis, a member of the order Hemiptera. The main bioactive compounds identified in this plant include gallic acid and methyl gallate, which exhibit antibacterial, anti-inflammatory, and antioxidant activities (Oh et al., 2003). It contains tannins (50-60%), which confer astringency and contribute to antidiarrheal, hemostatic, antibacterial, and sweat-inhibitory effects (Cha et al., 2000). R. chinensis contains gallotannins and triterpenoids, which have antioxidant, antithrombotic, antiviral, and melanin biosynthesis-inhibitory activities (Bae et al., 2012; Chen et al., 2009; Kurokawa et al., 1999; Song et al., 2002). Additionally, studies have reported the preservative effects of R. chinensis extracts on cosmetic formulations (Cho et al., 2008). In traditional medicine, the species is used to treat diarrhea, colitis, dysentery, gastric bleeding, hemoptysis, night sweats, spontaneous sweating, external wounds, abscesses, eczema, impetigo, and chronic cough (Lee et al., 2018). Numerous phenolic compounds and flavonoids present in natural materials are known for their antioxidant, whitening, anti-wrinkle, and moisturizing effects (Garg et al., 2017; Panzella and Napolitano, 2019).
Given the growing interest in health and natural functional substances, research on bioactive compounds derived from medicinal plants and natural extracts is increasing rapidly (Joung et al., 2007). These bioactive substances are considered high-value materials with broad applications and are the focus of continuous research and new compound development (Tabance et al., 2001). Furthermore, interest in antioxidants and anti-aging has significantly influenced consumer trends in cosmetic purchases, driving research on free radical-based mechanisms of aging (Harman, 1992).
Supercritical fluids are incompressible at temperatures and pressures above the critical point, exhibiting unique properties not found in conventional organic solvents. Supercritical fluids possess high densities (similar to liquids), low viscosity (comparable to gases), low surface tension, and high diffusion coefficients, making them ideal for the efficient extraction of bioactive compounds (Coelho et al., 2003; Kim et al., 2019). Supercritical fluids can be continuously adjusted from low-density gas-like states to high-density liquid-like states, allowing control over various properties, such as the equilibrium, transport properties, and molecular aggregation (Cho et al., 2004). Owing to the non-toxic, non-flammable, non-reactive, and non-corrosive nature, carbon dioxide (CO2) is commonly used as an extraction solvent, with applications spanning food, cosmetics, and pharmaceuticals (Hwang et al., 2017). However, research on the bioactivities of supercritical extracts from R. chinensis is limited. Thus, this study aimed to analyze the components and activities of R. chinensis extracts obtained using supercritical fluid extraction, providing a basis for assessing their value for use in cosmetics and applications in the cosmetic industry.
Materials and methods
Experimental materials
R. chinensis was purchased from Daekwang Yakupsa (Chuncheon, Gangwon Province, Korea). Supercritical fluid extraction was performed using an SC-CO2 extraction system (Ilshin Autoclave, Daejeon, Korea) installed at the Nano-Bio Center in Jangseong County under the conditions listed in Table 1. Dried R. chinensis was ground using a pulverizer (KSP-25, KOREAMEDI Co., Daegu, Korea) for 1 minute and passed through a 100-mesh sieve. The resulting powder was refrigerated before extraction. For extraction, 500 g of R. chinensis was loaded into the reactor and extracted under the following conditions for 4 hours: pressure of 400 bar, temperature of 50°C, and an ethanol co-solvent flow rate of 0.7 mL/min. The separation vessel pressure was set at 50 bar, with a temperature of 40°C and a CO2 flow rate of 60 mL/min (Kim and Lee, 2021).
Table 1.
Operating conditions for CO2 extraction
| Extractor | Ethanol | Separator | CO2 | Chiller | Run-Time | ||||||
| (bar) | (°C) | (mL/min) | (min) | (bar) | (°C) | (mL/min) | (°C) | (min) | |||
| 400 | 50 | 0.7 | 240 | 50 | 40 | 60 | 5 | 240 | |||
Measurement of the gallic acid content
The gallic acid content was measured using a modified high-performance liquid chromatography (HPLC) method based on Cho et al. (2016). A total of 50 mg of extract was added to a 10 mL volumetric flask, dissolved in MeOH, and filtered through a 0.45 µm membrane filter. A 10 µL aliquot of the filtered extract was injected into the HPLC system. All reagents were of HPLC grade. The gallic acid used as a marker compound was purchased from Sigma-Aldrich (St. Louis, MO, USA). The analytical conditions for gallic acid detection are detailed in Table 2.
Table 2.
HPLC conditions for the analysis of gallic acid
| Instrument | Conditions | ||
| Column | Shiseido C18 (5.0 µm, 4.6 mm × 250 mm) | ||
| Column temp. | 40 | ||
|
Mobile phase (Gradient) | Time (min) | Solvent A1) | Solvent B2) |
| 0 | 8 | 92 | |
| 23 | 8 | 92 | |
| 26 | 15 | 85 | |
| 36 | 30 | 70 | |
| 40 | 45 | 55 | |
| 43 | 45 | 55 | |
| 45 | 8 | 92 | |
| Detector | Waters 996 Photodiode Array Detector (280 nm) | ||
| Flow rate | 1.0 mL/min | ||
| Injection volume | 10 µL | ||
| Run time | 45 min | ||
Measurement of total phenolic and total flavonoid contents
Total phenolic content was determined using the Folin-Denis method (Folin and Denis, 1912). In 100 µL of Folin-Ciocalteu phenol reagent, 100 µL of the sample solution diluted to various concentrations was added and reacted at room temperature for 3 minutes. Then, 100 µL of Na2CO3 solution was added, and absorbance at 760 nm was measured using an enzyme-linked immunosorbent assay (ELISA) reader. The average phenolic content was quantified using a standard curve prepared with gallic acid. The total flavonoid content was measured following the methods of Kim et al. (2011), with modifications. Each R. chinensis sample (0.5 mL) was mixed with 1.5 mL of 95% ethanol, 0.1 mL of 10% aluminum nitrate, 0.1 mL of 1 M potassium acetate, and 2.8 mL of distilled water and then reacted at room temperature for 30 minutes. Absorbance at 415 nm was measured using a microplate reader, and the total flavonoid content was calculated using a rutin standard curve.
DPPH radical scavenging assay
DPPH radical scavenging activity was measured to evaluate antioxidant potential based on electron transfer. This method is widely used for screening antioxidants from natural sources; it is simple with a short turnaround time. The principle involves the reduction of purple DPPH radicals to a stable, colorless molecule when they receive electrons or hydrogen atoms from antioxidants. The reaction mixture contained 1 mL of ethanol, 10 µL of extract, and 990 µL of 100 mM sodium acetate buffer (pH 5.5) in a test tube, to which 0.5 mL of 0.5 mM DPPH solution was added. The mixture was incubated in the dark for 5 minutes, and the absorbance of the remaining radicals was measured at 517 nm using a UV spectrophotometer (Lee and Lee, 2008).
DPPH radical scavenging capacity (%) = (1-absorbance with sample/absorbance without sample) × 100
ABTS radical scavenging assay
The ABTS assay is a method for assessing hydrogen ion donation capability; it measures antioxidant efficacy based on the decolorization of the ABTS radical from turquoise to light green upon interaction with antioxidants generated through the reaction of ABTS with potassium persulfate (Natella et al., 2014). The assay is conducted by reacting ABTS radicals with antioxidants in a sample, leading to the reduction of the characteristic color of the radical (Cho et al., 2013). The radicals were generated by incubating a 7 mM ABTS solution with 2.45 mM potassium persulfate for 16 hours in the dark. For the assay, 10 µL of the sample and 290 µL of ABTS solution were mixed in a 96-well microplate and incubated in the dark for 6 minutes. Absorbance was read at 734 nm (Re et al., 1999), and ABTS radical scavenging activity was calculated.
ABTS radical scavenging activity (%) = (1-absorbance of the experimental group/absorbance of the control group) × 100
Reducing power
To assess the reducing power, 0.1 mL of the sample was mixed with 0.5 mL of sodium phosphate buffer (0.2 M) and 0.5 mL of 1% potassium ferricyanide, followed by incubation at 50°C for 20 minutes. Next, 0.5 mL of 10% trichloroacetic acid was added, and the mixture was centrifuged for 10 minutes. The supernatant (0.5 mL) was combined with 0.5 mL of distilled water and 0.1 mL of 0.1% iron(III) chloride. The reaction mixture was left at room temperature for 10 minutes, and absorbance was measured at 700 nm using a microplate reader to compare the reducing power (Cho et al., 2011).
MTT cell viability assay
An MTT assay was conducted to determine the cytotoxicity of supercritical fluid extracts from R. chinensis on RAW264.7 cells and to establish experimental treatment concentrations. RAW264.7 cells were cultured in Dulbecco’s modified Eagle medium at a density of 5 × 104 cells/mL. The cells were seeded at a density of 1 × 104 cells/mL into 96-well plates (180 µL per well) and incubated for 2 hours to allow attachment. The attached cells were then treated with 20 µL of each extract for 24 hours. After incubation, the medium was replaced with fresh medium, and MTT solution was added to achieve a final concentration of 0.5 mg/mL. The amount of purple formazan was quantified (Mosmann, 1983).
Tyrosinase inhibition activity
The activity of mushroom tyrosinase (150 units, Sigma-Aldrich) was assessed using 150 µL of an enzyme solution mixed with 225 µL of 2.5 mM l-tyrosine, 225 µL of 0.4 M Hepes buffer (pH 6.8), and 300 µL of ethanol. Absorbance before and after the reaction was measured at 475 nm (Kim et al., 2006), and tyrosinase inhibition was calculated as follows:
Tyrosinase inhibition (%) = × 100
A and B: Absorbance of sample-treated solution before and after incubation.
C and D: Absorbance of control solution (no sample) before and after incubation.
Antimicrobial activity assay
Antibacterial activity was assessed using the paper disc method (Davidson and Parish, 1989). Microbial strains used for the assay were obtained from the Biological Resource Center (KCTC/BRC, Jeongeup, Korea) and the Korean Collection for Type Cultures (KCCM, Seoul, Korea). The species and strain numbers are listed in Table 3. The cultured microorganisms were adjusted to 1.0 × 106 CFU/mL. Each strain was inoculated onto agar plates (100 µL per plate), and filter paper discs (8 mm in diameter, Roshi Kaisha Ltd., Tokyo, Japan) were impregnated with 40 µL of extract solutions at concentrations ranging from 0.5 to 10 mg/mL. The plates were incubated at 37°C for 24 hours. Ethanol was used as a control, and antibacterial activity was determined by measuring the diameter of the inhibition zone (clear zone) around each disc.
Table 3.
List of strains and cultivation conditions used for antimicrobial experiments
Measurement of nitric oxide (NO) production
RAW264.7 cells were seeded in a 96-well plate at a density of 6 × 104 cells/well and incubated in a 37°C, 5% CO2 cell incubator for 24 hours to allow cells to adhere to the plate. Cells were then treated with the sample at the specified concentrations and incubated for another 24 hours. To prepare the Griess reagent, phosphoric acid was dissolved in distilled water at a concentration of 5%, followed by the addition of sulfanilamide at 1% to the phosphoric acid solution. Additionally, N-(1-naphthyl)ethylenediamine dihydrochloride was dissolved in distilled water at 0.1%. These prepared solutions were mixed at a 1:1 ratio to prepare the Griess reagent. After incubation, the cell culture supernatant was mixed with the Griess reagent at a 1:1 ratio, and absorbance was measured at 540 nm using an ELISA reader (Green et al., 1982). The amount of NO was determined using a standard curve generated from sodium nitrite. The final NO concentration was normalized to the protein content and compared with that in the negative control group. The inhibition of NO production was calculated as the difference between NO levels induced by lipopolysaccharide (LPS) stimulation and NO levels in the extract-treated samples.
Statistical analysis
Statistical analyses were performed using SAS version 9.3 (SAS Institute Inc., Cary, NC, USA). One-way ANOVA was used to assess statistical significance, followed by Duncan’s multiple range tests for post hoc analyses, with a significance level of P < 0.05. Correlations between antioxidant components and antioxidant activity were analyzed using Pearson correlation coefficients, with P < 0.01 indicating significance.
Results and Discussion
Gallic acid content
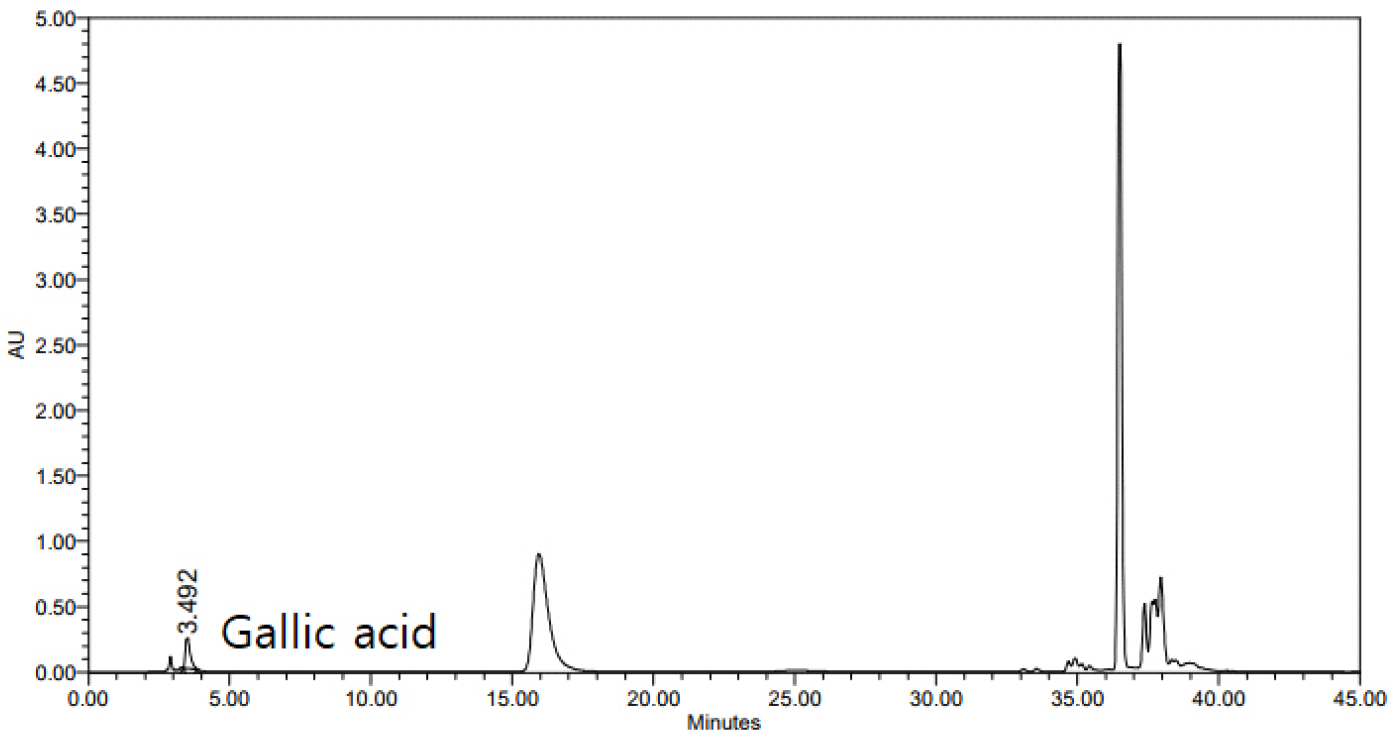
The gallic acid content of the R. chinensis extract was high, i.e., 15.22 mg/g (Table 4 and Fig. 1). This amount was higher than those reported for methanolic extracts by Park and Choi (2022). Gallic acid is a water-soluble compound found in plants either freely or bound as gallotannin. It was present in greater quantities than those of various chestnut cultivars, which range from 0.15% to 1.3% (Jeon et al., 2020).
Table 4.
Contents of gallic acid in Rhus chinensis CO2 extracts
| Sample | Contents (mg/g) |
| RCM1) | 15.22 ± 0.32 |
Total polyphenol and flavonoid contents
Phenolic compounds found in plant-based foods have diverse functions, including antioxidant, anti-obesity, and anti-inflammatory effects (Cho et al., 2007). As polyphenolic substances, flavonoids are categorized based on their chemical structures into various types, such as flavonols, flavones, catechins, and isoflavones. These compounds exhibit different solubilities in water and ethanol, and their biochemical activities, such as the inhibition of lipid peroxidation, are influenced by structural variation (Middleton and Kandaswami, 1994). The total phenol and flavonoid contents of the supercritical R. chinensis extract are presented in Table 5. The total phenol content was 268.10 ± 27.11 mg GAE/g and total flavonoid content was 68.21 ± 0.28 mg RE/g. These values were notably higher than the values of 5.98-8.12 wt% reported for hot water extracts of traditional medicinal herbs, such as Epimedium koreanum, Pueraria lobata, Acanthopanax sessiliflorus, and Kalopanax septemlobus (Kim et al., 2004).
Table 5.
Total polyphenol and flavonoid contents of Rhus chinensis CO2 extracts
| Sample | RCM1) |
| Total Phenol contents (GAE2) mg/g) | 268.10 ± 27.11 |
| Total Flavonoid contents (RE3) mg/g) | 68.21 ± 0.28 |
DPPH radical scavenging activity
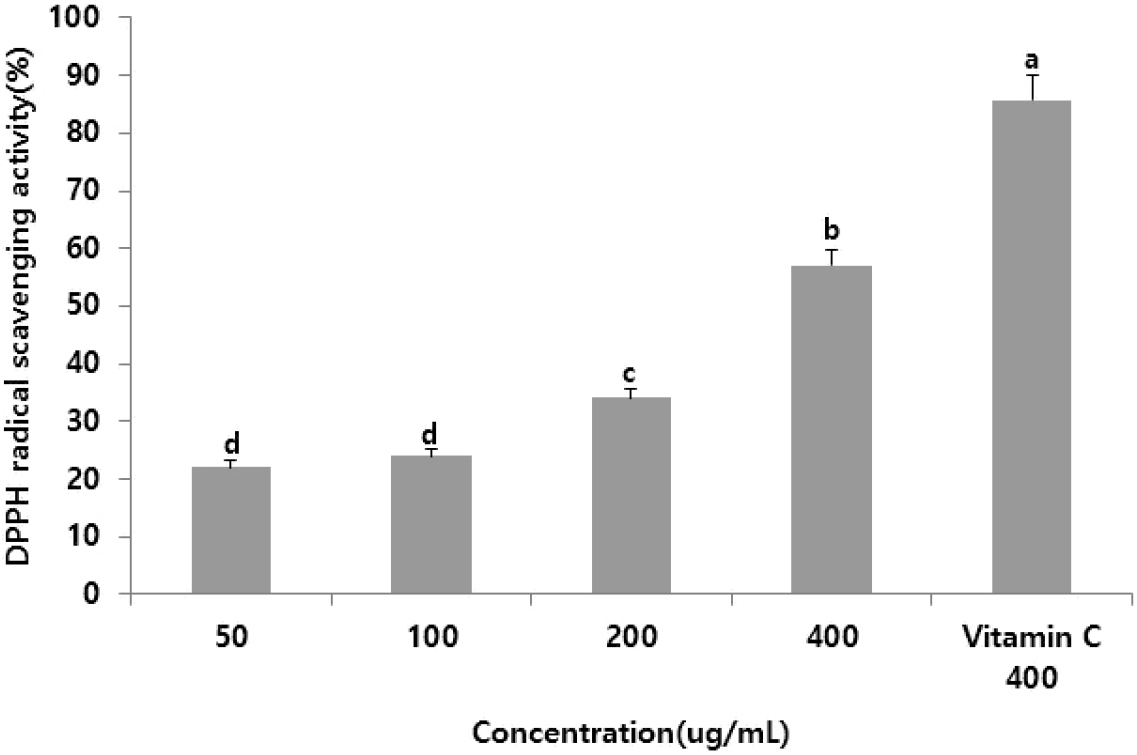
Various methods have been developed to assess the antioxidant activity of specific substances. The DPPH free radical scavenging assay is among the most widely used owing to its simplicity, suitability for large-scale testing, and rapid measurement (Que et al., 2006). The DPPH scavenging activity of the supercritical R. chinensis extract increased with increasing concentrations, with estimates of 23.81%, 25.88%, 31.21%, and 60.01% at 50.0, 100.0, 200.0, and 400.0 µg/mL, respectively (Fig. 2). These results are consistent with those for the supercritical Anemarrhena asphodeloides extract (An et al., 2018), demonstrating the antioxidant potential of the extract and suggesting its efficacy in inhibiting free radical-induced aging.
ABTS
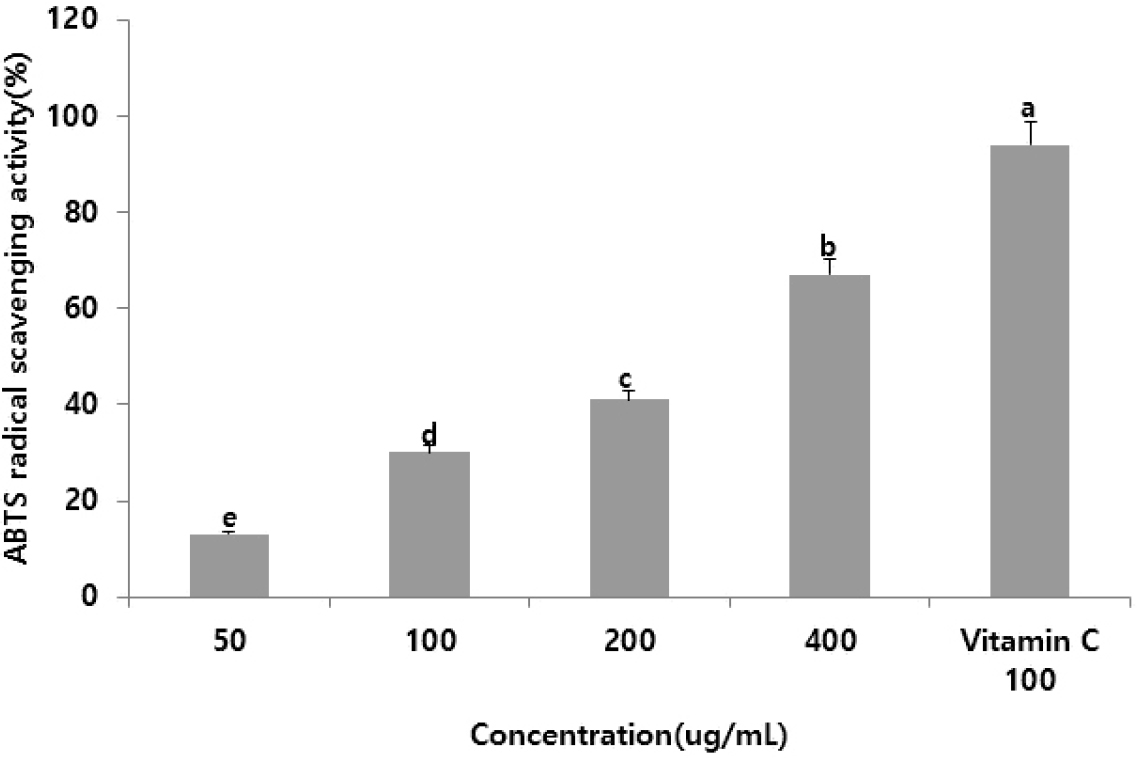
ABTS radicals are produced by the reaction of ABTS with potassium persulfate, resulting in a peroxide radical that turns a deep teal color. Antioxidant activity is measured based on absorbance, indicating the extent to which the teal color fades when antioxidants neutralize the radicals. Unlike DPPH radical scavenging, which involves neutralizing free radicals, ABTS radical scavenging targets cationic radicals (Miller et al., 1993). The ABTS radical scavenging activity of supercritical R. chinensis extracts is shown in Fig. 3. Similar to the results for ginger supercritical extracts (Lee et al., 2011), the ABTS scavenging activity increased with higher extract concentrations. These findings suggest that R. chinensis extracts are a potential new natural antioxidant for the prevention and treatment of skin aging and chronic inflammatory skin conditions induced by oxidative stress.
Reducing power
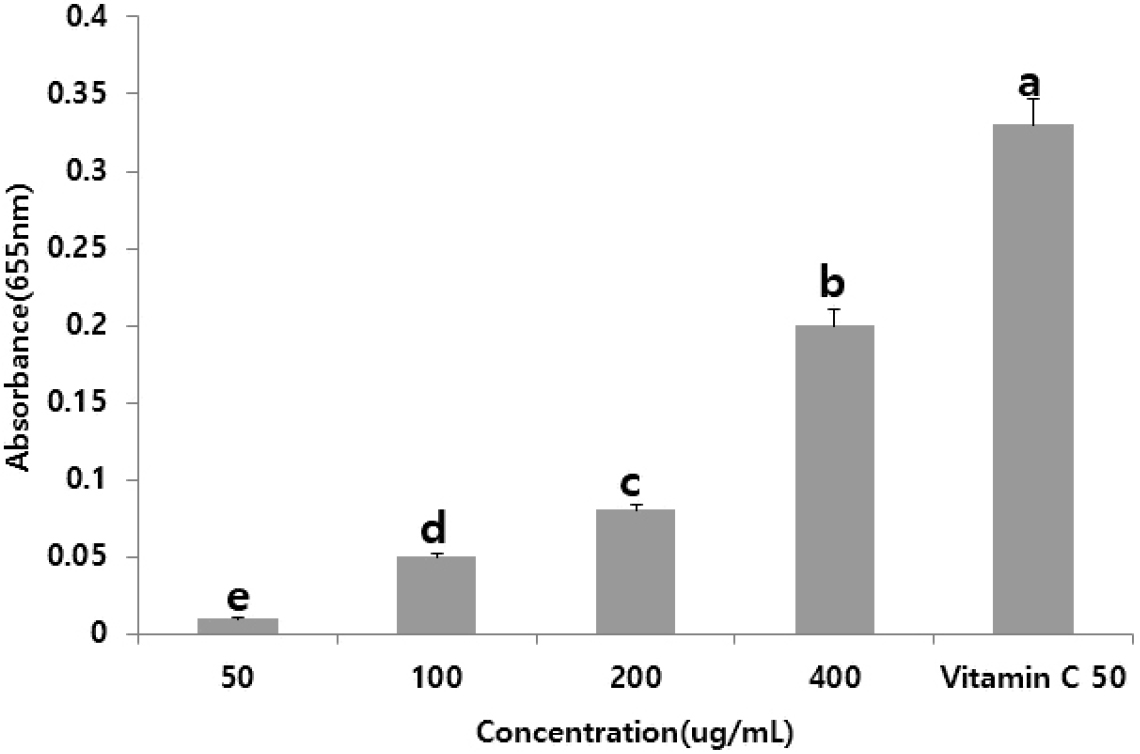
The reducing power assay measures the electron-donating ability of a sample, which stabilizes radicals by reducing Fe3+ to Fe2+. The absorbance values at 655 nm for concentrations of 50.0, 100.0, 200.0, and 400.0 µg/mL were 0.29, 0.40, 0.55, and 0.60, respectively, indicating a dose-dependent increase in reducing power (Fig. 4). These findings are consistent with those of studies on lotus leaf methanol extracts, which showed strong antioxidant activity, including hydroxyl and free radical scavenging (Wu et al., 2003).
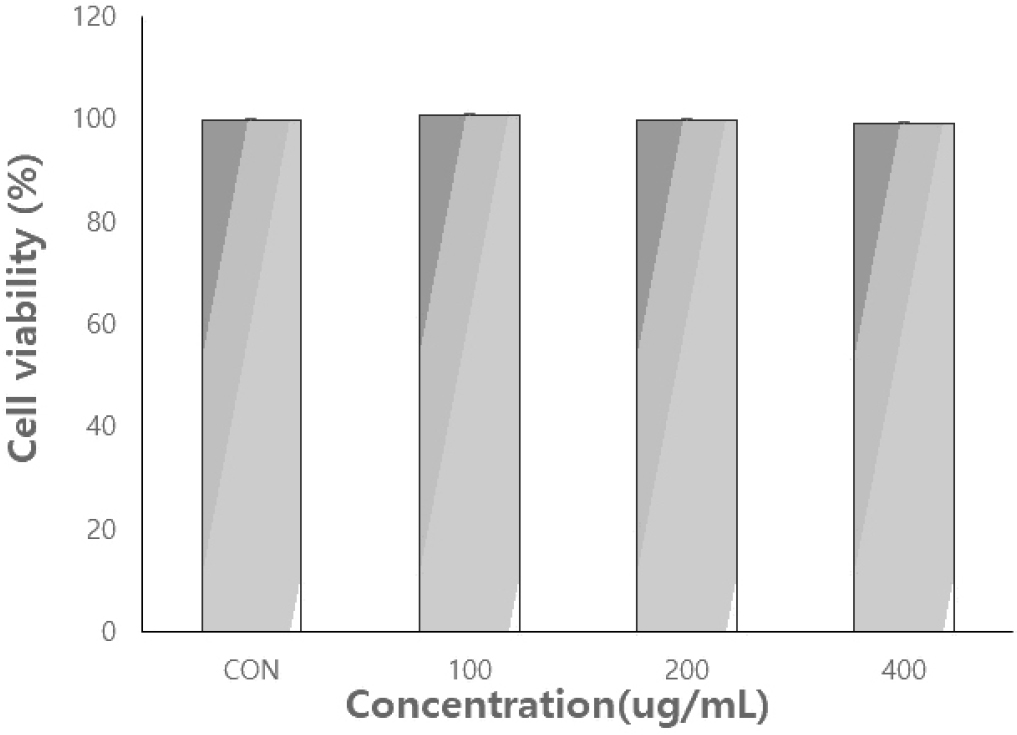
Cell viability
The MTT assay is a widely used method for assessing in vitro cell proliferation and viability. This assay was used to assess the viability of RAW264.7 cells treated with R. chinensis extracts. As shown in Fig. 5, cell viability was greater than 100% at concentrations of 100.0, 200.0, and 400.0 µg/mL.
Tyrosinase inhibition
Tyrosinase is a critical enzyme in melanin synthesis within the skin; it catalyzes the conversion of the amino acid tyrosine into 3,4-dihydroxy phenylalanine (DOPA), DOPA quinone, and eventually dihydroxyindole (DHI) to form melanin (Lee et al., 2012). The inhibitory activity of the R. chinensis supercritical fluid extract against tyrosinase was analyzed. The results indicated a concentration-dependent inhibitory effect (Table 6). A study of supercritical fluid extracts from Angelica gigas reported a tyrosinase inhibition rate of 45.04%, suggesting its potential as a skin-whitening agent (Park et al., 2021).
Table 6.
Effects of Rhus chinensis CO2 extracts on tyrosinase activity (%)
| Control | RCM1) (µg/mL) | ||
| 100 | 200 | 400 | |
| 100 ± 1.82a) | 99.20 ± 1.14a) | 98.02 ± 3.53b) | 92.12 ± 4.53c) |
Nitric oxide (NO) production
Inflammatory responses are essential defense mechanisms against external threats. NO is a well-known inflammatory mediator; however, excessive production can lead to inflammation and, in severe cases, autoimmune disorders (Vlassara, 2005). The inhibitory effect of the R. chinensis supercritical fluid extract on NO production was evaluated. NO synthesis was induced in cells using LPS, and the concentration of NO in the cell culture supernatant was measured using Griess reagent. NO inhibition was calculated as the difference between the NO level induced by LPS and the NO level with the extract. NO production increased by 90% with LPS stimulation; however, 20 µg/mL extract reduced NO production by 65%, and dose-dependent inhibition was observed at higher concentrations. These findings suggest that the R. chinensis supercritical fluid extract has significant anti-inflammatory properties.
Antimicrobial activity
Excessive proliferation of skin-resident bacteria can trigger or worsen skin inflammation. In inflammatory skin conditions, the skin’s natural defense against harmful microorganisms weakens, and a compromised skin barrier increases vulnerability to infections, exacerbating chronic inflammatory skin diseases (Lee et al., 2016). The R. chinensis supercritical fluid extract demonstrated antimicrobial activity across all tested concentrations. The extract showed particularly strong activity against Escherichia coli, with the largest clear zone observed at a concentration of 2 mg/mL. The clear zone size increased in a concentration-dependent manner.
Table 7.
Antimicrobial activity of the supercritical extract of Rhus chinensis
|
Control (mg/mL) | Microorganisms | |||
| E. coli | S. aureus | P. aeruginosa | P. acnes | |
| Clear zone diameter (mm) | ||||
| 0.5 | 9 | 9 | 0 | 5 |
| 5 | 11 | 10 | 11 | 7 |
| 10 | 15 | 11 | 13 | 10 |
Correlation between the antioxidant content and antioxidant activity
Antioxidant activity is commonly assessed using five in vitro methods based on metal ion chelation, hydrogen donation, antioxidant enzyme activity, oxygen scavenging, and singlet oxygen quenching (Lee et al., 2012; Yang et al., 2011). In this study, the antioxidant components (total phenolic and flavonoid contents) and antioxidant activities (DPPH radical scavenging, ABTS radical scavenging, and reducing power) were evaluated for various concentrations of R. chinensis extracts. Correlation coefficients (R2) for the relationships between these components and activities were obtained (Table 8). For all antioxidant assays, R2 values were close to 1, indicating a significant positive correlation between the phenolic and flavonoid contents and antioxidant activity. These findings suggest that supercritical R. chinensis extracts possess strong radical-scavenging and redox capabilities, indicating that they are potentially effective natural antioxidants.
Table 8.
Correlation coefficients (R2) for relationships between the antioxidant activity and antioxidant compounds of Rhus chinensis CO2 extracts
| Parameter | DPPH | ABTS | Reducing power | TPC1) | TFC2) |
| DPPH | 1.000 | ||||
| ABTS | 0.892 | 1.000 | |||
| Reducing power | 0.958 | 0.851 | 1.000 | ||
| TPC1) | 0.952 | 0.967 | 0.998 | 1.000 | |
| TFC2) | 0.978 | 0.985 | 0.961 | 0.978 | 1.000 |
Summary
In this study, R. chinensis extracts were obtained using the supercritical carbon dioxide extraction method and assessed with respect to the gallic acid content, total polyphenol content, and antioxidant, whitening, and antimicrobial activities. The extract had a high gallic acid content of 15.22 mg/g, total polyphenol content of 268.10 mg/g, and flavonoid content of 68.21 mg/g. The DPPH radical scavenging activity was 60.01% at 400 µg/mL, ABTS radical scavenging activity was 67.51%, and reducing power was 0.21. Additionally, tyrosinase inhibition reached 92.12% at 400 µg/mL. Antimicrobial activity was detected across all concentrations (0.5-10 mg/mL), and the extract effectively suppressed inflammatory responses. These results indicate that R. chinensis extracts exhibit antioxidant activity through radical scavenging and redox mechanisms, suggesting significant potential as natural antioxidants and effective whitening agents. In future research, we will identify the optimal supercritical carbon dioxide extraction conditions and further evaluate the association between bioactivity and the gallic acid content.