Introduction
Materials and Methods
Materials and reagents
Preparation of extracts
Measurement of total polyphenol and total flavonoid contents
Measurement of ABTS (2,2’-azinobis-[3-ethylbenzothiazoline-6-sulfonic acid]) radical scavenging activity
Measurement of DPPH radical scavenging activity
Measurement of oxygen radical absorbance capacity (ORAC) activity
Measurement of ferric-reducing antioxidant power (FRAP)
Statistical analyses
Results and Discussion
Total polyphenol and total flavonoid contents
ABTS radical scavenging
Measurement of DPPH radical scavenging activity
ORAC assay using fluorescein
FRAP assay
Correlation between active components and antioxidant potency
Conclusions
Introduction
Aceriphyllum rossii Engler is a perennial herbaceous plant belonging to the Saxifragaceae family that grows along valleys in moist, rocky areas, with rhizomes spreading through crevices to facilitate growth and reproduction (Ahn et al., 2008; Lim et al., 2010). The leaves typically emerge wrapped in protective scales near the tips of rhizomes, giving the appearance of multiple leaves originating from the same point. The flowers of A. rossii are white or pale pink and bloom in May, growing up to 30 cm tall (Lee, 2006). In Korea, it is also called “baunari” (Yoo, 2007), and another scientific name, Mukdenia rossii (Oliv.) Koidz (Zheng et al., 2010), is also used.
Aceriphyllum rossii is distributed in the northern regions of Korea and China. Traditionally, its young leaves and stems have been consumed in Korea as a health food (Cui et al., 2012; Lee, 1996), whereas in China, they have been used as a cardiac stimulant and diuretic (Zheng et al., 2010). The major constituents of the plant are reported to be pentacyclic triterpenoids (Han et al., 2002) and flavonol glycosides (Han et al., 2004), and its anticancer activity has been reported using flavonoid compounds isolated from the plant (Ahn et al., 2008). Other properties associated with this plant and its extracts have also been researched, including anti-inflammatory (Trang et al., 2014), anti-complement (Min, 2012), anti-wrinkle (Ha et al., 2015), and anti-obesity (Park et al., 2011) properties. Although its antioxidant activity has been reported to be superior to that of ascorbic acid (Ha et al., 2015), studies evaluating the effects of different extraction solvents on its antioxidant activity have not been conducted.
Generally, the extraction method for natural materials must be selected based on the affinity of the solvent for plant-based natural compounds, the cost of the solvent, and other practical considerations. Extraction solvents are mainly hot water or organic solvents such as ethanol and methanol (Kim et al., 2008). Hot water extraction increases the contents of phenolic compounds and other bioactive substances in sweet corn (Dewanto et al., 2002). However, hot water extraction has been shown to lead to substance loss and relatively low yield for Toona sinensis (Oh and Yoon, 2017). Ethanol extraction has been reported to efficiently elute insoluble substances from Scutellaria baicalensis roots (Lim et al., 2019). A study examining bioactivity in T. sinensis extracts obtained using various enzyme treatments and solvents showed that ethanolic extract exhibited the highest bioactivity (Oh and Yoon, 2017).
In this study, we investigate antioxidant activity in different extracts of A. rossii leaves obtained using water and ethanol mixes of various concentrations and measure the total flavonoid and polyphenol contents in these extracts.
Materials and Methods
Materials and reagents
Aceriphyllum rossii leaves used in this study were cultivated in Yanggu, Gangwon Province, Korea. The leaves harvested and ground in June 2021 were used for the extraction. Reagents used in the experiments included 2,2-diphenyl-1-picrylhydrazyl (DPPH), methanol, ascorbic acid, 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), Folin-Ciocalteu phenol reagent, sodium carbonate (Na2CO3), tannic acid, sodium nitrite (NaNO2), aluminum chloride (AlCl3), sodium hydroxide (NaOH), and rutin hydrate, all purchased from Sigma-Aldrich Co. (St. Louis, MO, USA). Dimethyl sulfoxide (DMSO) was obtained from Junsei Chemical Co. (Tokyo, Japan); 300 mM sodium acetate buffer (pH 3.6) from Biosolution Co. (Seoul, Korea); ferric chloride from Kanto Chemical Co. (Tokyo, Japan); and 2,4,6-tripyridyl-s-triazine (TPTZ) from Merck & Co. (St. Louis, MO, USA). Other reagents and solvents were analytical grade (Merck & Co., USA).
Preparation of extracts
Aceriphyllum rossii leaves were dried for 2 days at 40°C in a cold-air dehumidifier (TJHP-1003; Daegu, Korea), then ground to a 40-mesh powder for extraction. For the water extract, 20 g of the powdered sample was mixed with 200 mL of distilled water and subjected to ultrasonic extraction at 60°C for 6 h using an ultrasonic extractor (8510R-DTH; Bransonic, Danbury, CT, USA), with the process repeated. The extract was filtered twice under vacuum using a Buchner funnel with Whatman No. 2 filter paper (Whatman, Maidstone, England) and concentrated using a rotary vacuum evaporator (N-21NS; EYELA, Tokyo, Japan). The concentrated extract was dissolved in 10 mL of distilled water and freeze-dried (LP-100; Ilsin Lab, Yangju, Korea) for storage at -20°C until use.
For ethanol extracts, 20 g of the powdered sample was mixed with 200 mL of ethanol solutions diluted to 10%, 30%, 50%, and 70% with distilled water. Extraction was performed twice over 12 h at 120 rpm using an orbital shaker (SK71; JeioTech, Daejeon, Korea). The ethanol extracts were prepared in the same way as the water extract. Before experiments, the stored samples were dissolved in 30% DMSO and diluted to the required concentrations.
Measurement of total polyphenol and total flavonoid contents
The total polyphenol content was determined using a modified Folin and Denis (1912), which measures the blue color produced when phenolic compounds react with phosphomolybdic acid. In a 96-well plate, 10 µL of the sample (1 mg/mL concentration) was mixed with 90 µL of distilled water, and then 10 µL of Folin-Ciocalteu phenol reagent was added. After a 3 min reaction at room temperature, 70 µL of distilled water and 20 µL of a Na2CO3 saturated solution pre-dissolved at 70°C were added. The mix was allowed to react for an hour at room temperature, and absorbance was measured at 725 nm. Tannic acid was used as the standard to generate a calibration curve, and total polyphenol content was expressed in milligrams of tannic acid equivalent (TAE) per gram, i.e., mg TAE/g.
The total flavonoid content was measured by mixing 25 µL of the sample (1 mg/mL concentration) with 100 µL of distilled water, and then 7.5 µL of 5% NaNO2 was added. The mix was reacted at room temperature for 5 min, following which 7.5 µL of 10% AlCl3 was added and allowed to react for another 5 min. Subsequently, 50 µL of 1 M NaOH was added and allowed to react for 1 min. The mix was diluted with 60 µL of distilled water, mixed well, and absorbance was measured at 510 nm. Rutin hydrate was used as the standard, and total flavonoid content was expressed in milligrams of rutin hydrate equivalent (RHE) per gram, i.e., mg RHE/g.
Measurement of ABTS (2,2’-azinobis-[3-ethylbenzothiazoline-6-sulfonic acid]) radical scavenging activity
ABTS radical scavenging activity was measured using the method of Re et al. (1999) with a few modifications. The ABTS solution (7.4 mM) was mixed with 2.6 mM potassium persulfate and allowed to react in the dark at room temperature for 12-15 h until the absorbance reached 0.7 ± 0.03 at 734 nm. Subsequently, 300 µL of the ABTS solution was mixed with 20 µL of the sample. After allowing the mix to stand at room temperature for 20 min, absorbance was measured at 734 nm. ABTS radical scavenging activity was calculated as a percentage using the formula (1 - absorbance of sample / absorbance of control) × 100, and the half maximal inhibitory concentration (IC50) value was determined to quantify antioxidant activity.
Measurement of DPPH radical scavenging activity
DPPH radical scavenging activity was assessed using the method proposed by Blois (1958) with a few modifications. Briefly, a 24-well plate was used to mix 200 µL of the sample with 800 µL of 0.2 mM DPPH solution. The mix was left to stand at room temperature for 30 min, and absorbance was measured at 517 nm. Samples were prepared at various concentrations to represent the concentration for 50% scavenging of DPPH radicals. The scavenging activity was calculated as (1 - absorbance with sample added / absorbance with no sample added) as a percentage. In addition, antioxidant activity was quantitatively evaluated by calculating the IC50 value of DPPH radical scavenging activity measured at each concentration.
Measurement of oxygen radical absorbance capacity (ORAC) activity
ORAC activity was measured using an ORAC assay kit (Cell Biolabs Inc., CA, USA). In a 96-well plate, 25 µL of sample and 150 µL of 1X fluorescein solution were added and reacted for 30 min at 37°C. Subsequently, 25 µL of free radical initiator solution was added, and fluorescence decay was measured every 3 min for 60 min using a fluorescence detector (FLUOstar Omega; BMG Labtech, Germany) with excitation at 480 nm and emission at 520 nm. Results were compared with a Trolox calibration curve and expressed as µmol Trolox equivalent (TE) per 100 g.
Measurement of ferric-reducing antioxidant power (FRAP)
FRAP was measured based on the method proposed by Benzie and Strain (1996). The reaction mix consisted of 300 mM sodium acetate buffer (pH 3.6), 10.0 mM TPTZ, and 20.0 mM FeCl3 mixed in a 10:1:1 ratio and was reacted for 15 min. Subsequently, 50 µL of the sample was mixed with 150 µL of distilled water and 1.5 mL of FRAP reagent, followed by a 15-min reaction. Absorbance was then measured at 593 nm. The reducing power was calculated using a Trolox standard curve and expressed as mg TE per 100 g.
Statistical analyses
All results are presented as the mean of three replicates. Statistical significance was determined using the analysis of variance with SAS version 9.4 (Statistical Analysis System, SAS Institute Inc., Cary, NC, USA), and Duncan’s multiple range test was used to evaluate differences at a significance level of p < 0.05.
Results and Discussion
Total polyphenol and total flavonoid contents
The extraction yields were 29.27%, 29.70%, 31.33%, 29.37%, and 31.07% for the water extract and 10%, 30%, 50%, and 70% ethanolic extracts, respectively. Although the yields varied depending on the extraction solvent because of differences in extracted components (Cha et al., 2006; Kim et al., 1995), the variation was not substantial in this study.
Polyphenolic compounds are secondary metabolites believed to help inhibit reactive oxygen species-induced damage at the DNA or cellular level (Vaya et al., 1997). Phenolic compounds are known to increase bioactivity in the body, and high levels of phenolic compounds are associated with high antioxidant activity (Um et al., 2017). Table 1 summarizes the extraction yields and the total polyphenol and flavonoid contents of A. rossii leaf extracts obtained using different solvent compositions of water and ethanol. The extract obtained using 50% ethanol exhibited the highest polyphenol content at 381.27 mg/g and a high flavonoid content of 176.53 mg/g. In contrast, the water extract showed lower polyphenol and flavonoid contents at 231.10 and 99.93 mg/g, respectively. The higher antioxidant activity and flavonoid and polyphenol contents observed in the ethanol extracts than in water extracts are likely because of the efficiency of ethanol in eluting key antioxidant components from A. rossii leaves. Unlike water, ethanol exhibits a high capacity to solubilize both lipophilic and hydrophilic compounds, making it relatively more effective for extracting antioxidants, such as flavonoids and polyphenols (Shahidi and Ambigaipalan, 2015; Lee et al., 2007). According to Kim et al. (2018), ethanol extraction increases the yield of antioxidant substances compared with that obtained with water extraction, presumably because the solubility of various antioxidant compounds changes with ethanol concentration. The high polyphenol content observed in the 50% ethanol extract in this study aligns with this trend and may be attributed to the strong affinity between ethanol and polyphenolic and flavonoid compounds (Antolovich et al., 2002; Vaya et al., 1997). Additionally, water-based extraction may negatively impact the stability of certain antioxidant compounds compared with that in organic solvent extraction. During high-temperature water extraction, certain antioxidant compounds may degrade because of heat exposure (Ha et al., 2015; Kim et al., 2008). Shahidi and Ambigaipalan (2015) reported that heat can damage the structure of phenolic compounds.
Table 1.
Total polyphenol and flavonoid contents in Aceriphyllum rossii leaf extracts
| Solvent used | Yields (%) |
Total phenolic content (mg TAE1)/g of dry extract) |
Total flavonoid content (mg RHE2)/g of dry extract) |
| Distilled water | 29.27 ± 0.21 | 231.10 ± 2.59d | 99.93 ± 3.50c |
| 10% Ethanol | 29.70 ± 0.04 | 293.47 ± 8.59c | 147.20 ± 2.64b |
| 30% Ethanol | 31.33 ± 0.18 | 342.17 ± 23.65b | 174.66 ± 5.28a |
| 50% Ethanol | 29.37 ± 0.18 | 381.27 ± 36.98a | 176.53 ± 12.68a |
| 70% Ethanol | 31.07 ± 0.21 | 306.40 ± 6.00bc | 180.80 ± 3.96a |
ABTS radical scavenging
Considering the association of reactive oxygen species generated during human metabolism with various human diseases, research on developing natural antioxidants has been actively pursued (Seong et al., 2018). In this study, the ABTS radical scavenging assay revealed that the water extract exhibited the highest IC50 value at 1.026 mg/mL (Fig. 1). The ethanol extracts exhibited varying IC50 values depending on the A. rossii leaf extract concentrations of 0.684, 0.592, 0.599, and 0.644 mg/mL for 10%, 30%, 50%, and 70% ethanol extracts, i.e., E10, E30, E50, and E70, respectively. The 30% ethanol extract demonstrated the lowest IC50 value, indicating the strongest radical scavenging activity. Overall, the ethanol extracts had lower IC50 values than exhibited by the water extract, suggesting that ethanol extraction may be relatively more effective in enhancing the antioxidant content of A. rossii leaves. Other studies have also shown that the methanol-extracted fractions of A. rossii leaves exhibit significant ABTS radical scavenging activity compared with that in control fractions. This high activity is likely attributed to compounds, such as astragalin and kaempferol 3-O-α-L-rhamnopyranosyl (1→6)-β-D-glucopyranoside, present in A. rossii leaves (Han et al., 2003).
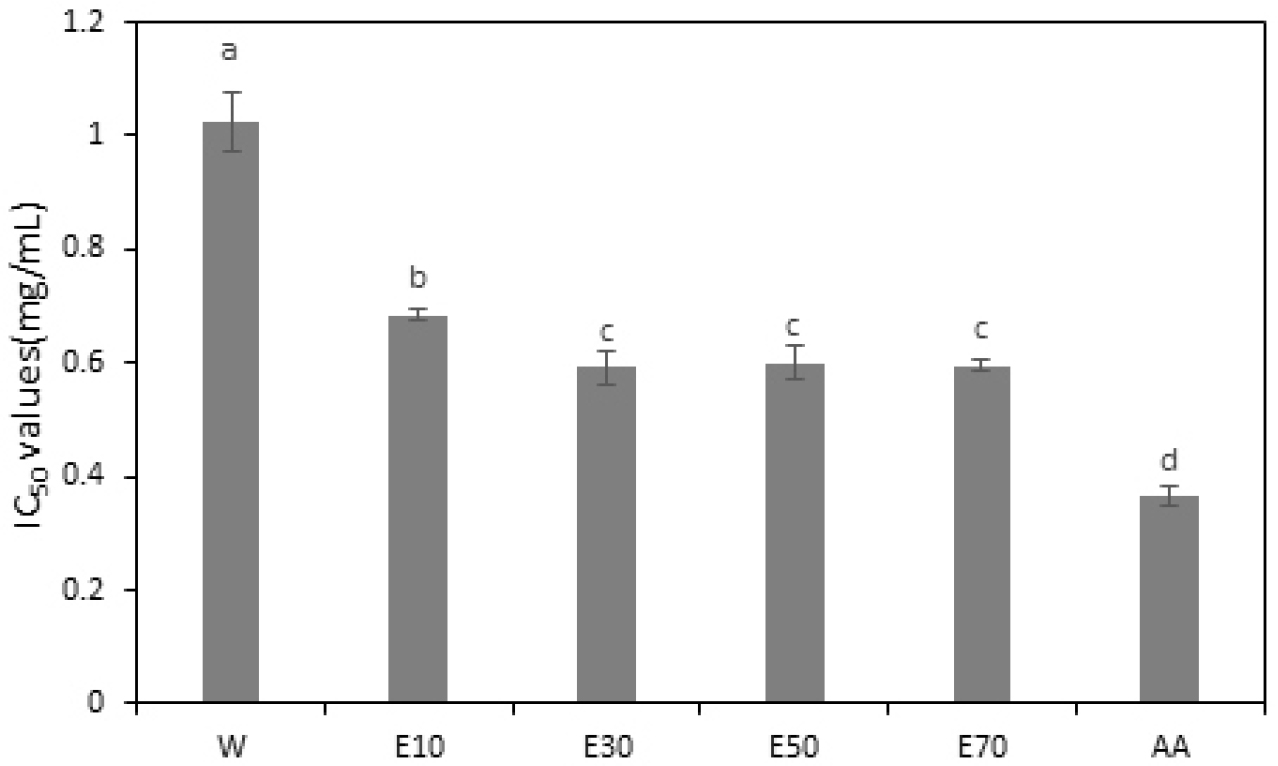
Fig. 1.
Half maximal inhibitory concentration (IC50) values of the ABTS ((2,2’-azinobis-[3-ethylbenzothiazoline-6-sulfonic acid]) radical scavenging abilities of Aceriphyllum rossii leaf (ARL) extracts. W, water extract of ARL; E10, E30, E50, and E70; 10%, 30%, 50%, and 70% ethanol extracts of ARL, respectively; AA, control, L-ascorbic acid. IC50 values represent the concentrations required for 50% ABTS radical scavenging activity. All values represent mean ± SD of three independent measurements, and different letters of superscripts on the respective bars are significantly different at p < 0.05 based on Duncan’s multiple range test (a > b > c > d).
Measurement of DPPH radical scavenging activity
The DPPH radical scavenging assay, which measures the activity of antioxidants based on hydrogen atom donation to stabilize nitrogen-based radicals, is an established method for assessing antioxidant capacity (Teixeira et al., 2013). This method is widely used to measure lipid peroxidation inhibition and determine the anti-aging effects of various components to counter oxidative stress (Hyun et al., 2007; Lee et al., 2007). The DPPH radical scavenging activity assay revealed that the IC50 values of A. rossii leaf extracts varied based on the extraction solvents in the following order: water extract (96.2 µg/mL) >10% ethanol extract (70.367 µg/mL) >30% ethanol extract (62.467 µg/mL) >50% ethanol extract (59.567 µg/mL) >70% ethanol extract (59.433 µg/mL) (Fig. 2). The 30%, 50%, and 70% ethanol extracts showed the highest scavenging activities without significant differences between their values, and all ethanol extracts demonstrated higher activities than those of the water extract. The superior DPPH radical scavenging activity of the ethanol extracts can be attributed to the ability of ethanol to extract both hydrophilic and lipophilic compounds, particularly its ability to extract lipophilic antioxidants. Although water extraction is generally suitable for hydrophilic components, ethanol can extract a broader range of antioxidant compounds (Dai and Mumper, 2010), likely contributing to the higher antioxidant activity in the ethanolic extracts than in the water extract. The increase in antioxidant activity observed with increasing ethanol concentration is consistent with the results reported in previous studies. Vongsak et al. (2013) reported an improved concentration of antioxidants and increased antioxidant potency when high-concentration ethanol extracts were used. The increase in DPPH radical scavenging capacity with increasing ethanol concentration observed in this study is consistent with this mechanism.
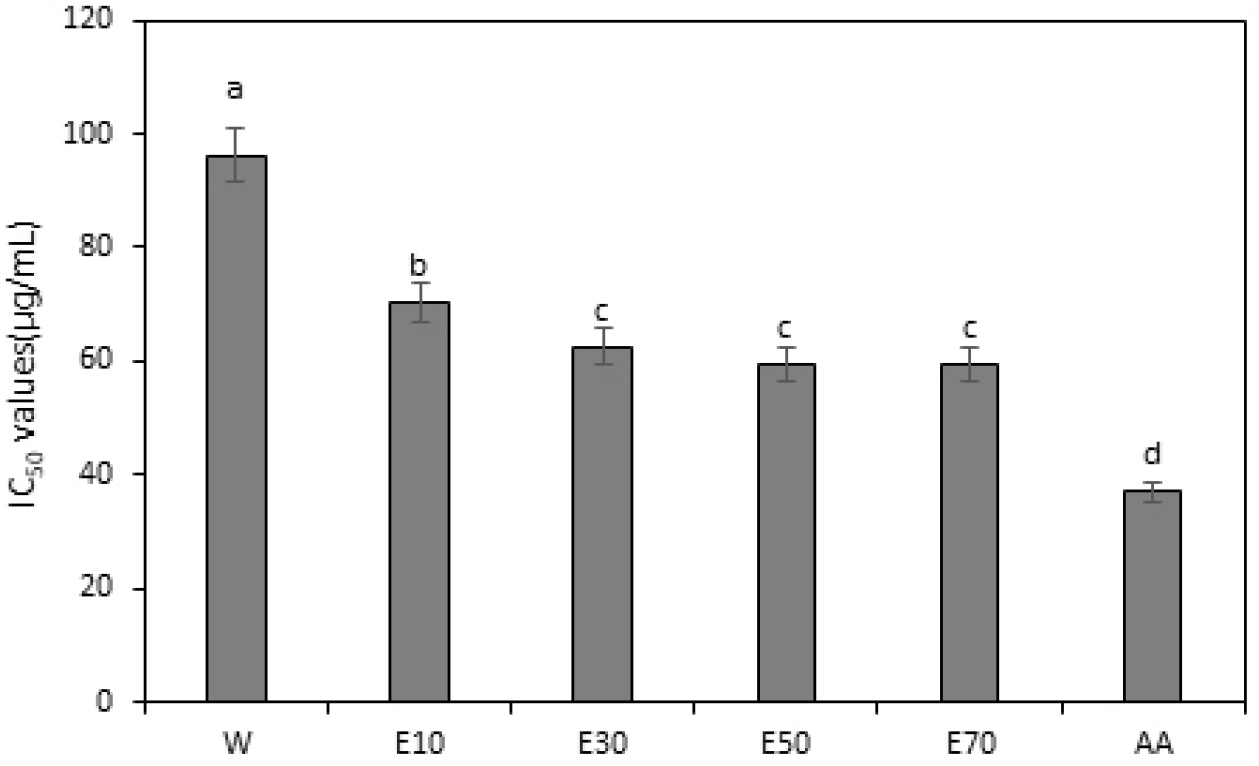
Fig. 2.
Half maximal inhibitory concentration (IC50) values of the DPPH radical scavenging abilities of Aceriphyllum rossii leaf. IC50 values represent the concentrations required for 50% DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activity. AA, control, L-ascorbic acid. All values represent mean ± SD of three independent measurements, and different letters of superscripts on the respective bars are significantly different at p < 0.05 based on Duncan’s multiple range test (a > b > c > d).
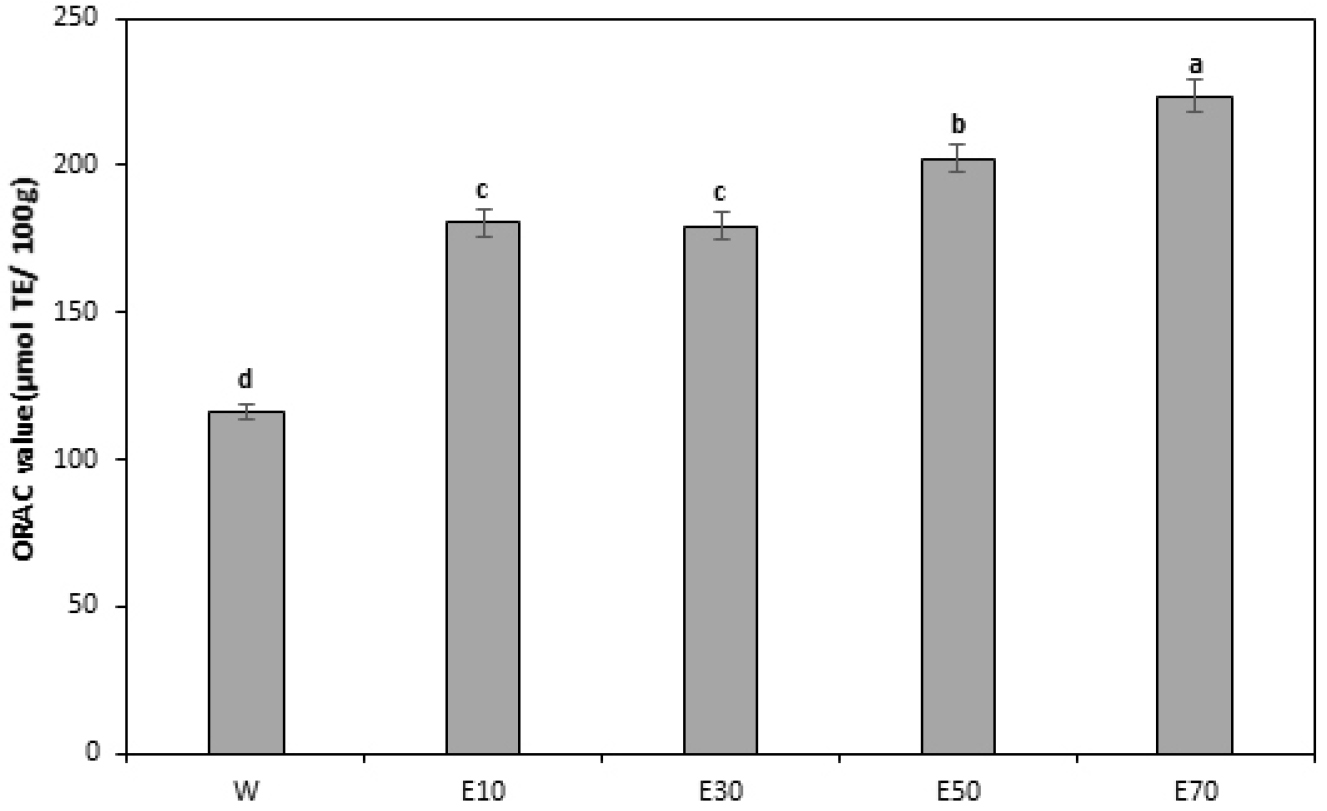
ORAC assay using fluorescein
The ORAC assay quantifies antioxidant capacity by measuring the rate of fluorescence decay towing to peroxy radical formation and destruction (Kim et al., 2007). Fig. 3 shows the ORAC values of A. rossii leaf extracts based on the extraction solvent used. At a 10.0 µg/mL concentration of A. rossii leaf extract, ORAC was the highest for the 70% ethanol extract, followed by that for the 50%, 10%, and 30% ethanol extracts and water extract in that order. The 70% ethanol extract exhibited the highest ORAC value at 223.44 µmol TE/100 g, supporting the trend that higher ethanol concentrations yield greater antioxidant activity. Dai and Mumper (2010) also highlighted the efficacy of ethanol in extracting antioxidant compounds, which, therefore, is relatively more effective for preparing extracts with high ORAC values. The lower ORAC value of the water extract than those of the ethanol extracts suggests that antioxidant components dissolve relatively more efficiently in ethanol. Lee et al. (2014) reported that antioxidant compounds in plants, such as polyphenols and flavonoids, generally exhibit higher solubility in ethanol than in water; therefore, ethanolic extracts exhibit higher antioxidant activities than observed in water extracts. The results obtained in the present study are consistent with these reports and suggest that ethanol extraction may be a relatively more effective way to extract antioxidant activity.
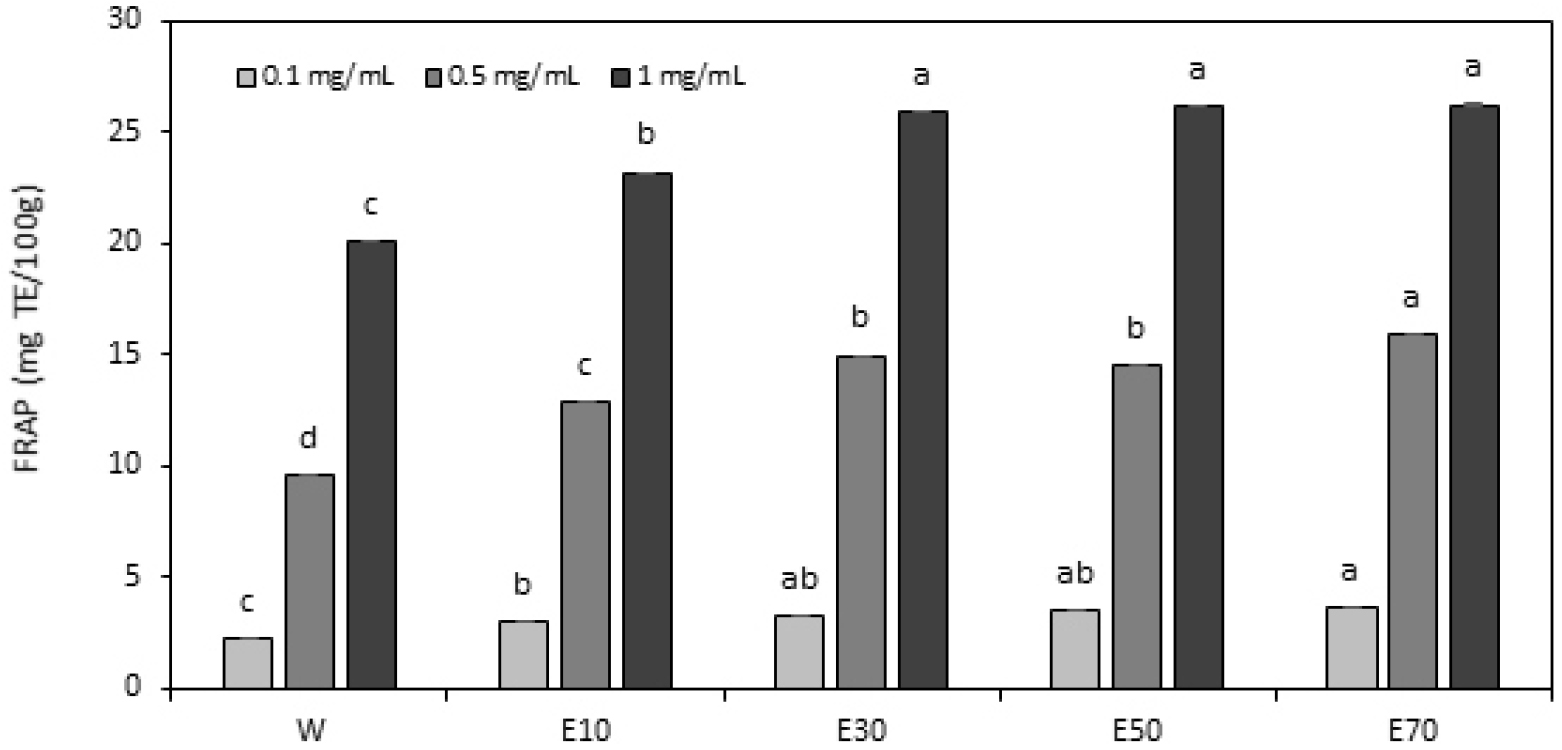
FRAP assay
The FRAP assay is a classical electron transfer-based method that measures the reduction of the ferric-TPTZ (Fe3+-TPTZ) complex to ferrous-TPTZ (Fe2+-TPTZ) complex, which produces an intense blue color (Antolovich et al., 2002). This method emphasizes an increase in redox potential to drive changes in reaction mechanisms. Experiments are conducted under acidic pH conditions to maintain iron solubility and facilitate effective electron transfer (Shahidi and Ambigaipalan, 2015). Higher absorbance values of the Fe2+-TPTZ complex indicate greater reducing power, signifying stronger antioxidant capacity (Jang et al., 2018). The FRAP measurements for the A. rossii leaf extracts obtained using different solvents are shown in Fig. 4. Across all solvents, FRAP values increased with increasing concentrations of A. rossii leaf extracts. At all concentrations, the ethanol extracts showed higher reducing power than that recorded for the water extract, with the strongest antioxidant activity at a high concentration of 1 mg/mL. At 1 mg/mL concentration, the highest FRAP value was recorded for the 30% ethanol extract, followed by that for the 50% and 70% ethanol extracts in that order, indicating that ethanol is relatively more effective in extracting antioxidant components from A. rossii leaves. This observation aligns with the findings of Lee and Shibamoto (2001), who reported that ethanol is an efficient solvent for plant-based polyphenolic and flavonoid compounds. The electron-donating ability of phenolic compounds contributes to their increased reducing power, and the higher antioxidant capacity of the ethanol extracts can be attributed to the enhanced extraction of these compounds. The results of this study validate that ethanol extracts demonstrate a higher reducing power than observed for water extracts, likely because of the amphiphilic nature of ethanol, which enhances its antioxidant extraction efficiency and positively impacts FRAP values.
Correlation between active components and antioxidant potency
The results of the correlation analysis between the total polyphenol and flavonoid contents and antioxidant efficacy recorded in this study are represented as a heat map (Fig. 5). The strongest correlation was observed between the total flavonoid content and DPPH radical scavenging activity, indicating that the total flavonoid content exerts a significant impact on the antioxidant activity of A. rossii leaves, which suggests that flavonoids play a crucial role in the antioxidant activity of A. rossii leaves. The mechanism through which polyphenols and flavonoids contribute to antioxidant activity involves their ability to scavenge free radicals. Polyphenols stabilize radicals through their phenolic hydroxyl groups, whereas flavonoids are known to reduce oxidative stress through various mechanisms, including enhancing antioxidant enzyme activity, chelating metal ions, and inhibiting signaling pathways (Heim et al., 2002; Pietta, 2000; Rice-Evans et al., 1997).
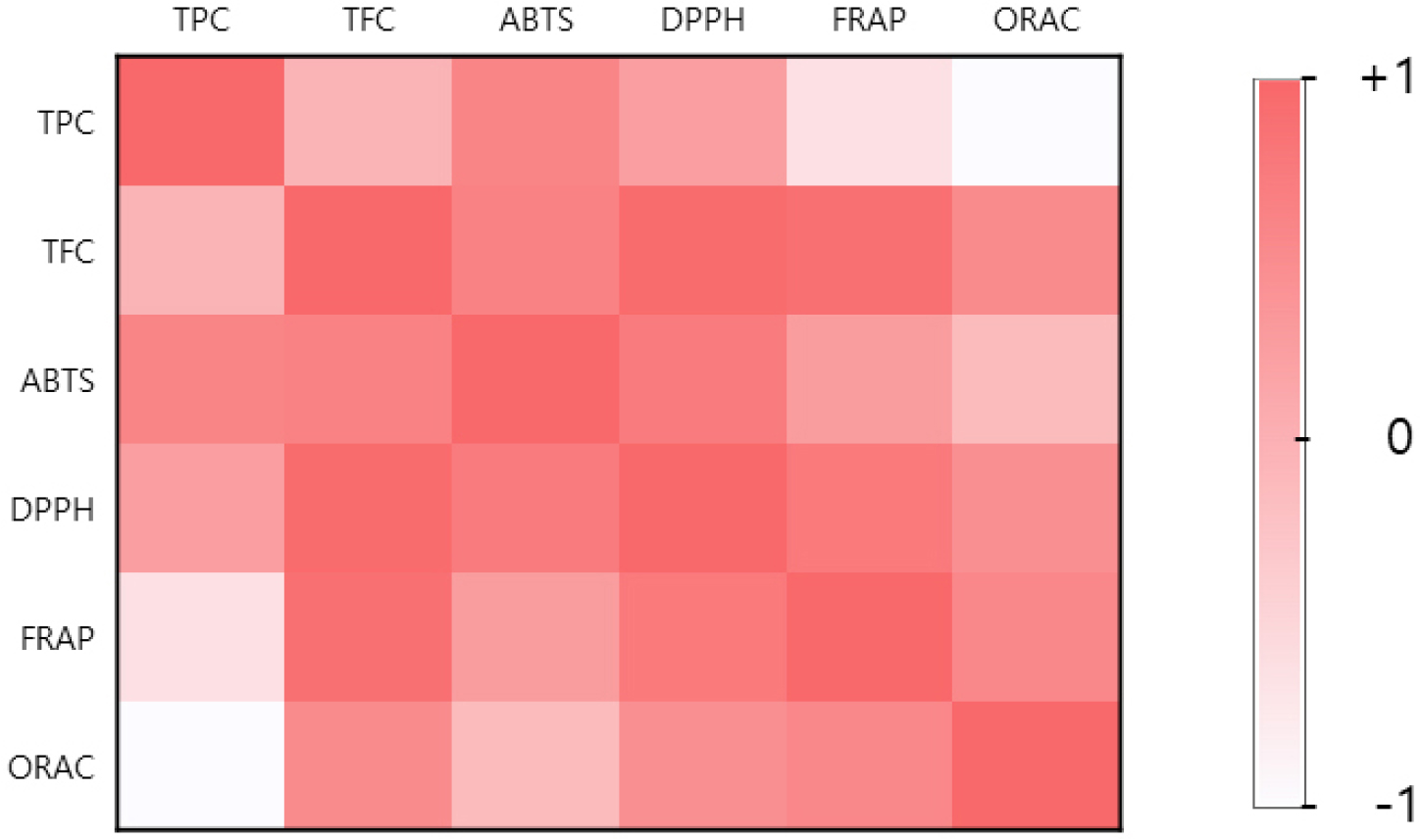
Fig. 5.
Heat map of correlation between the analyzed antioxidant parameters. TPC, total phenolic content; TFC, total flavonoid content; ABTS, ABTS (2,2‘-azinobis-[3-ethylbenzothiazoline-6-sulfonic acid]) radical scavenging activity (1.0 mg/mL); DPPH, DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activity (0.1 mg/mL); FRAP, ferric-reducing antioxidant power; ORAC, oxygen radical absorbance capacity.
Conclusions
The antioxidant activity experiments on A. rossii leaf extracts obtained using different extraction solvents revealed that the ethanol extracts exhibited higher antioxidant compound content and activity than those in the water extracts. In particular, the 30%, 50%, and 70% ethanol extracts showed higher polyphenol and flavonoid contents and also exhibited better antioxidant effects than those of the water extract and other ethanol extracts. However, from an industrial application perspective, 30% ethanol extraction is likely the most economically efficient. Using 30% ethanol reduces solvent costs and has advantages in large-scale production, such as relatively low ethanol recovery expenses. Therefore, incorporating 30% ethanol extraction in industrial processes could maximize economic benefits while preserving antioxidant efficacy. The A. rossii leaf extract prepared with 30% ethanol exhibited substantial potential as a natural antioxidant source for use in functional foods and cosmetics. Future studies must focus on examining the in vivo efficacy and safety of the ethanol extracts of A. rossii leaves and investigating additional solvents and extraction conditions.